What is the AI Clinical Trial Optimization Market Size?
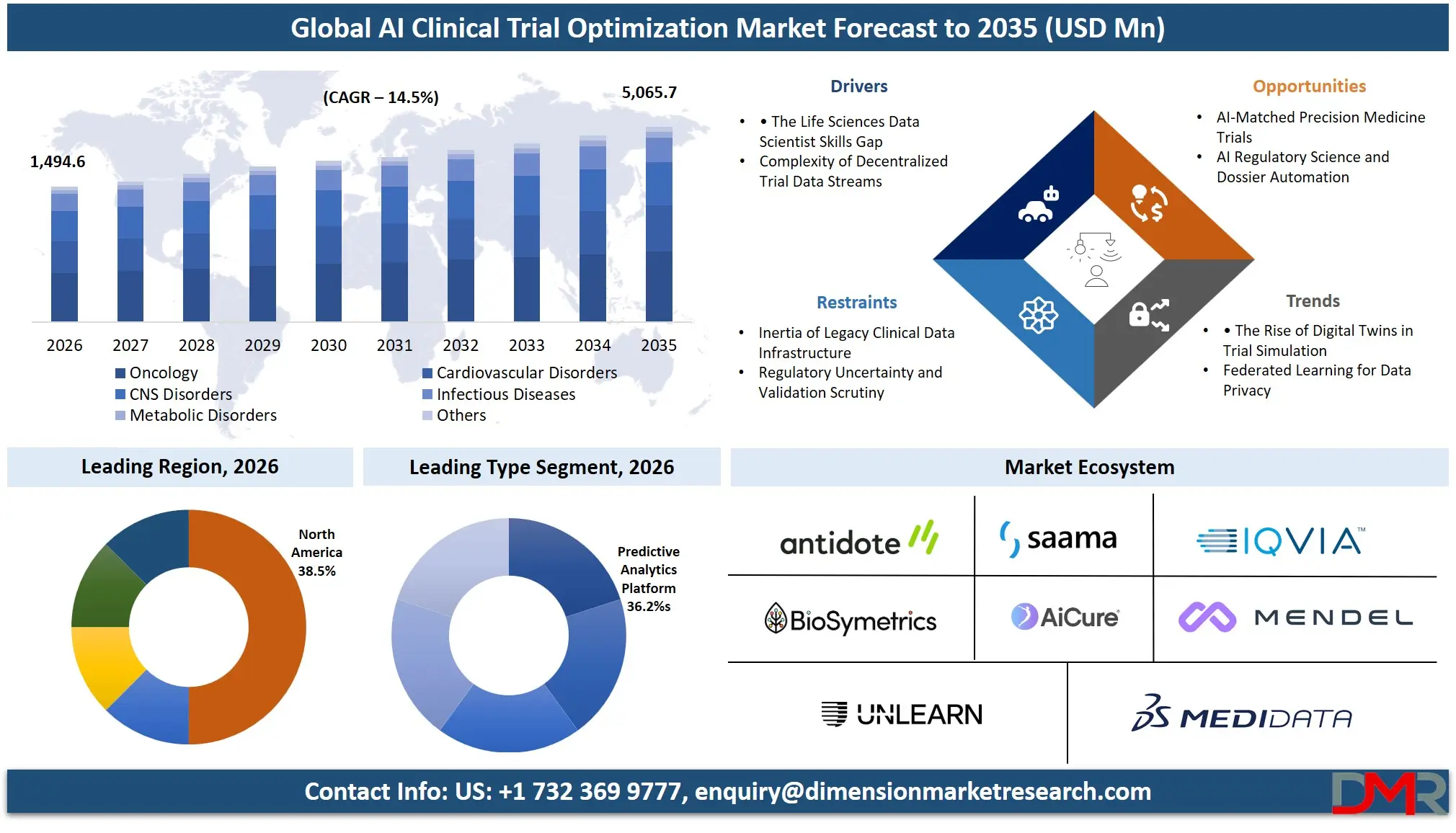
The Global AI Clinical Trial Optimization Market is expected to reach a value of USD 1,494.6 million in 2026, and it is further anticipated to reach USD 5,065.7 million by 2035, growing at a CAGR of 14.5% during the forecast period.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The AI clinical trial optimization market showed a high rate of growth and the life sciences industry is under the pressure never experienced to reduce the cycle time and increase the chances of success in drug development. The market includes the solutions of predictive analytics, patient recruitment, data management and safety assessment that can help organizations to streamline their clinical processes throughout all stages of research.
The growing intricacy of the trial protocols, the trend toward decentralized clinical trials (DCTs), and the necessity to utilize the real-world evidence are all contributing to the need to have specific AI-driven tools. The most common adopters are pharmaceutical and biotechnology companies where cloud-based solutions are the most popular due to their scalability and capability to combine dissimilar data sources. The oncology, CNS disorders, and infectious disease therapeutic areas are the major stakeholders since they demand sophisticated analytics to handle the complex datasets and determine responsive patient subpopulations.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US AI Clinical Trial Optimization Market
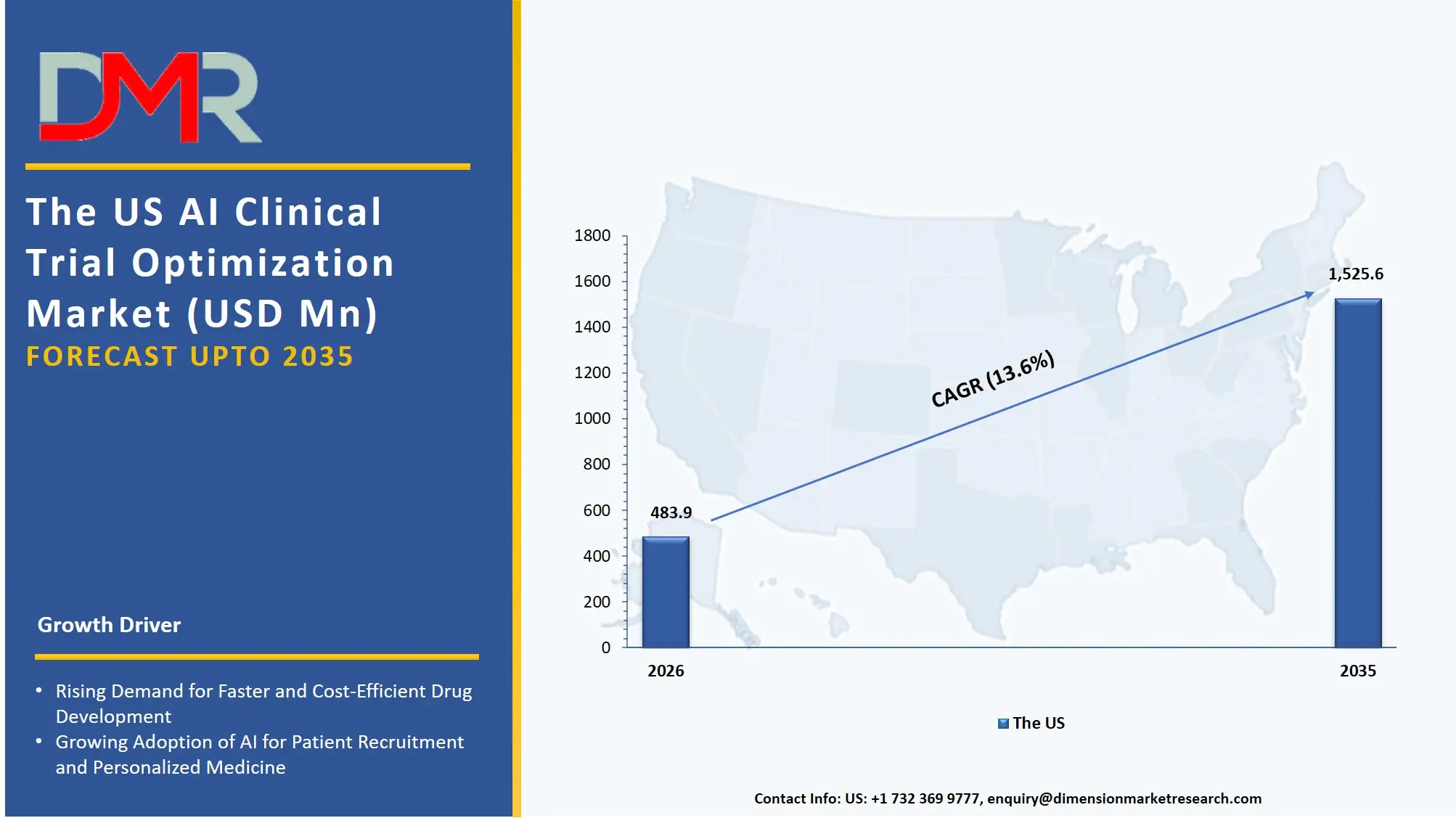
The US AI Clinical Trial Optimization Market is projected to reach USD 483.9 million in 2026 at a compound annual growth rate of 13.6% over its forecast period, which is further poised to reach USD 1,525.6 million by 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US is still the biggest and most advanced market in AI clinical trial optimization because of the high concentration of pharmaceutical R&D headquarters and the pushy nature of the FDA to modernize and promote the use of digital health tools. The market has been characterized by a high demand for predictive analytics platforms, whereby sponsors are targeted to predict the performance of the site and the time of patient enrollment more accurately. In addition, the trial workflow automation with natural language processing (NLP) Tools is generating an analogous requirement in Data Management and Analysis services to automate the curation of unstructured electronic health records (EHR) and medical literature.
The Europe AI Clinical Trial Optimization Market
The Europe AI Clinical Trial Optimization Market is estimated to be valued at USD 432.8 million in 2026 and is further anticipated to reach USD 1,490.2 million by 2035 at a CAGR of 14.8%. The regulatory frameworks, such as the European Clinical Trials Regulation (CTR) and EMA guidelines on AI in the medicines lifecycle, are influential to the European market and lead to the necessity to use AI-powered Decision Support Tools and Real-World Evidence (RWE) Platforms. The region is also experiencing accelerated growth of Hybrid deployment models with pharmaceutical companies in Germany and France attempting to find a balance between patient data sovereignty and the requirement to access cloud-based computational power. Besides this, there are initiatives like the European Health Data Space (EHDS) that are trying to encourage service providers to develop specific Data Integration and Management Solutions to offer data residency and interoperability across the European healthcare ecosystems.
The Japan AI Clinical Trial Optimization Market
The Japan AI Clinical Trial Optimization Market is projected to be valued at USD 147.5 million in 2026. It is further expected to witness robust growth, holding USD 478.8 million in 2035 at a CAGR of 14.1%. The Japanese market is distinct, whereby a corporate and governmental initiative exists to solve the drug lag and a rapidly ageing population in need of novel therapies in CNS Disorders and Oncology. A substantial portion of the expenditure goes to machine learning algorithms of trial design optimization and site selection, and monitoring services as pharmaceutical companies transition to AI-based adaptive trial design models, rather than their traditional siloed data analysis. A high level of necessity is also to integrate in depth with the local market in order to close the gaps between old hospital information systems and the new SaaS solutions, which develop a niche in Computer Vision Systems of medical imaging analysis and automated adverse event signal detection.
Key Takeaways
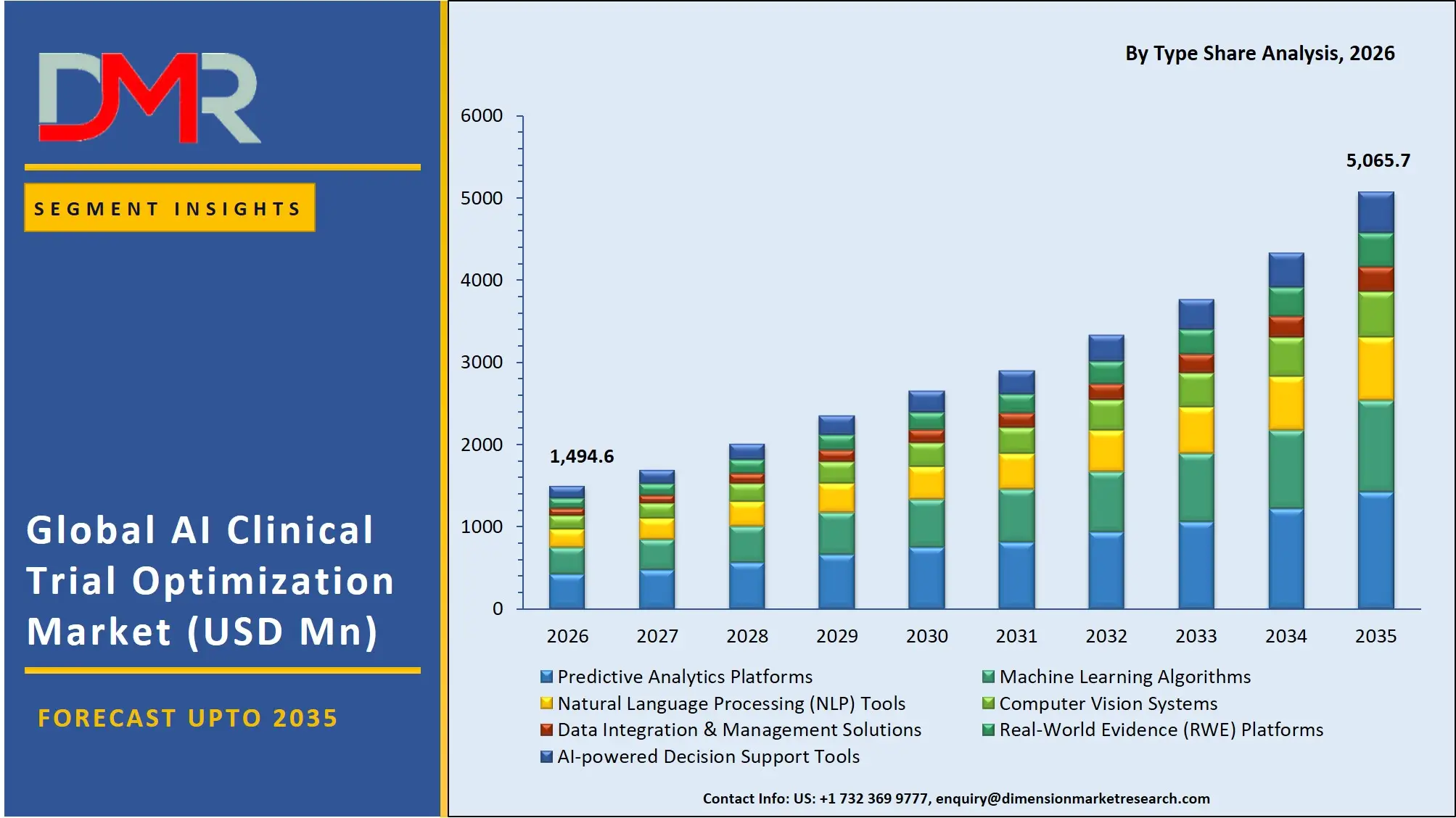
- Market Size & Forecast: The Global AI Clinical Trial Optimization market is projected to reach USD 1,494.6 million in 2026, expanding dramatically to USD 5,065.7 million by 2035, driven by the complexity of clinical trials and the imperative to cut down the high cost of drug development failures.
- Growth Rate & Outlook: Global market growth is expected at a CAGR of 14.5%, due to the severe lack of skilled data scientists in the biopharma sector, as well as the increasing complexity in handling multi-modal patient data generated by wearables, genomics, and imaging.
- Primary Growth Drivers: The broad adoption of non-traditional site-centric trials to decentralized and hybrid models, the urgency to develop AI-based Decision Support Tools to streamline protocol design and prevent expensive amendments, and the adoption of real-world evidence (RWE) platforms with specialized data management expertise are key drivers.
- Key Market Trends: Major trends, including the emergence of AI-controlled synthetic control weapons, NLP Tools to automate the screening of patients on a large scale, and the transition to Predictive Analytics Platforms as stakeholders focus on site feasibility and enrollment projections to avoid delays in trials.
- By Type Analysis: Predictive analytics platforms are projected to dominate because they predict outcomes, optimize protocols, and decrease the failure rates. They manipulate data sets, recognize patient trends, and optimize decision-making, and are highly embraced in pharmaceutical and biotech firms.
- By Deployment Analysis: Cloud-based solutions is poised to dominate deployment because of scalability, flexibility and cost efficiency. They facilitate immediate access to data, smooth cooperation, and connectivity with clinical teams. The trend toward increasing decentralized trials and remote administration requires more and more support for cloud deployment.
- Regional Leadership: North America is poised to dominate this market with a 38.3% of market share of the global market in 2026 because it has already established a robust technological ecosystem, is highly concentrated having much pharmaceutical R&D investment, and has a favorable regulatory outlook on AI implementation.
What is the AI Clinical Trial Optimization?
AI Clinical Trial Optimization refers to the dedicated software platforms and analytical tools, which are provided by technology vendors, CROs, and specialized consultancies to support life sciences organizations through the entire clinical development lifecycle. The tools, compared to the traditional clinical trial management system (CTMS), are connected to the how of creating insights and forecasting outcomes. This includes Predictive Analytics to anticipate site performance and patient dropout rates, Natural Language Processing (NLP) Tools to process the unstructured clinical notes in large volumes so that they can be matched to patients, and Data Integration and Management Solutions to make sure that the different data sources, such as EHRs, labs, and imaging archives, can communicate effectively with each other. As more than 80.0% of clinical trials fail to meet their enrollment schedules, AI optimization programs are required to realize patient-centered trial designs, proactive safety signal identification, and speedy time-to-market, so that R&D investments are monetized into a real competitive advantage, rather than sunk cost.
Use Cases
- Oncology Patient Matching in Phase II Trials: NLP Tools and Predictive Analytics Platforms are used by sponsors to search thousands of unstructured pathology reports, and earmark patients with targeted genetic mutations (e.g., KRAS G12C) as targets of targeted therapy, eliminating the need to activate a site and to move a patient to first-patient-in.
- RWE-Driven Synthetic Control Arms: Pharmaceutical firms are finding applications of Real-World Evidence (RWE) Platforms as a supplement or alternative to conventional placebo arms by using historical patient data found in electronic medical records to simulate disease progression, accelerating trials, and reducing the workload associated with patient recruiting.
- Automated Adverse Event Detection in Pharmacovigilance: Regulatory authorities and sponsor safety teams can scan clinical data and published literature continuously to automatically detect and flag potential safety signals much faster than manual review, ensuring patient safety and regulatory compliance.
- Predictive Site Selection for Global Phase III Trials: Large CROs apply AI-driven decision support tools and computer vision systems to review historical site performance, live satellite data on population density, and competitor trial activity, resulting in ranked lists of the best investigator sites to avoid expensive non-performers.
How AI is Transforming the AI Clinical Trial Optimization?
AI is transforming clinical trials by speeding up the patient recruitment process and increasing the predictive capability of trial design. AI-based simulation tools can be used in trial design optimization to automatically simulate different protocol scenarios over historical datasets, reducing significantly the risk of non-viability at protocol endpoints and requiring amendments to the protocol mid-study. In the meantime, AI-driven capabilities in data management and analysis enable sponsors to have a more precise control over data quality by identifying anomalies, forecasting the future attrition rates based on the engagement trends of patients, and recommending remedial measures, such as retention campaigns to strengthen patient compliance.
AI is also becoming the center of attention of risk-based monitoring and safety pharmacovigilance projects. Intelligent computer vision is applied in the field of safety and risk assessment, where medical imaging data undergoes continuous review on multi-site trials and protocol deviations and possible efficacy signals are identified without the variability of intra-reader. In addition, Drug Efficacy Prediction is being supplanted by Generative AI assistants that are simulating molecular interactions and modelling patient-specific responses to provide researchers with a visual representation of the potential success of a compound before investing in costly late-stage trials.
Market Dynamics
Key Drivers in the Global AI Clinical Trial Optimization Market
The Life Sciences Data Scientist Skills Gap
Pharmaceutical companies across the world are struggling to recruit experienced personnel, who understand modern AI/ML models such as PyTorch, federated learning, and time-series analysis of clinical data. The quantity of skilled labor required is increasing at a rate higher than the supply of trained talents and hence a structural scarcity in the labor market. It is causing a shift into a trend of companies outsourcing AI-enabled CROs and purchasing SaaS-based AI solutions instead of relying on developing their own models. These off-the-shelf solutions help in the key procedures such as automated patient screening, predictive enrollment, and harmonization of data, reducing the possibility of trial delays caused by a lack of in-house data science abilities.
Complexity of Decentralized Trial Data Streams
The large size of sponsors is also likely to have a variety of data sources such as wearables, eCOA, telemedicine visits, and standard site assessments so that patient-centricity and real-world data can be captured. However, multi-modal trial data environment is extremely difficult to integrate. Organizations should harmonize data standards, semantic interoperability, cybersecurity, and privacy policies on several technology platforms. This complication can lead to data silos, overlooked safety indicators, and redundant risk without the assistance of expert AI tools. Therefore, the demand of Data Integration and Management Solutions to help enterprises work in such a complicated ecosystem increases.
Restraints in the Global AI Clinical Trial Optimization Market
Inertia of Legacy Clinical Data Infrastructure
The majority of pharmaceutical organizations continue to use outdated clinical data management systems that had a long history of development and are extremely siloed across therapeutic area. Such old systems are a major barrier to the use of AI, even though AI tools could provide greater agility and data-driven insights. Moving large and tested GxP-compliant databases and sophisticated workflows to a new cloud-based AI architecture can be expensive and risky. Decades of trial data require careful mapping, validation, and regulatory documentation in case of data migration. Organizations are afraid of business disruption, lack of data lineage, and audit findings during transition.
Regulatory Uncertainty and Validation Scrutiny
Changing regulatory environments and the absence of particular FDA or EMA precedents that adaptive AI models follow have made organizations more reluctant to invest in advanced AI tools to confirmatory tests. Although optimization of AI is a strategic priority, clinical leads are under pressure to show model explainability and prevent black-box decision-making that can result in a trial rejection. Validation expenses and extended engagement frameworks of implementing AI-driven Decision Support Tools are especially likely to fall under enhanced examination. Businesses have switched to pilot programs in early-stage or operational applications that yield quick wins, including patient recruitment, before it dedicates itself to AI primary endpoint analysis. This transformation is forcing AI vendors to be more open and validation-based.
Growth Opportunities in the Global AI Clinical Trial Optimization Market
AI-Matched Precision Medicine Trials
Helping the pharmaceutical firms to develop secure AI-based tools of matching patients to the biomarker-defined sub-studies is one of the major growth opportunities in the market. Many companies have outgrown general chemotherapy trials and now would like basket and umbrella trial designs that involve dynamic screening of patients against complicated genetic backgrounds. These complex environments are developed with specific NLP skills to process genomic reports, and machine learning algorithms to assign patients to cohorts in real-time, and RWE integration to balance cohorts. The providers of AI solutions can help sponsors to create scalable, adaptable trial ecosystems that can make the process of enrolling into a trial and making decisions faster. The region is capable of generating demand of highly specialized analysis and implementation services.
AI Regulatory Science and Dossier Automation
The increasing demand to incorporate technical AI expertise and knowledge of international regulatory requirements is contributing to the rise of AI solutions as the agencies are modernizing their review procedures. These are medical-centric AI systems to automatically write clinical study report (CSR)s, safety narrative, and compile electronic common technical document (eCTD) modules. The pharmaceutical and biotech firms must meet rigorous submission guidelines and quick response to queries. Therefore, they need AI partners who will understand the machine learning technology and the clinical regulatory compliance system. In order to add value, AI platform providers can think of integrating NLP Tools with the already existing document management systems in accordance with the ICH guidelines, and customizing the automated narrative workflows.
Trends in the Global AI Clinical Trial Optimization Market
The Rise of Digital Twins in Trial Simulation
Digital twin technology is gaining more acceptance as an alternative to conventional, expensive trial-and-error protocol design. Instead of basing decisions on past experiences, enterprises are building AI-generated virtual patient cohorts that mimic drug response. These platforms allow the dynamic simulation of dose changes, endpoint sensitivity, and dropout rates. In response to this, AI vendors are offering experience in generative AI, pharmacokinetic/pharmacodynamic (PK/PD) modeling, and causal inference.
Federated Learning for Data Privacy
The privacy of data is also becoming a central theme in the training of AI models as firms are being pressurized to cooperate and share research on rare diseases without having sensitive patient-level data sent to central servers. Companies are now considering AI models, which are based on distributed datasets without data movement. This has ushered in the necessity of federated learning consulting and deployment services. AI solution vendors have aided in helping companies to find interoperable node setups, resource optimization and HIPAA/GDPR compliance, and minimize data breach risk and expedite cross-institutional research.
Research Scope and Analysis
The AI Clinical Trial Optimization Market is witnessing strong growth driven by increasing adoption of predictive analytics, cloud-based platforms, and AI-powered patient recruitment solutions. Rising oncology trials, expanding Phase III studies, and growing investments by pharmaceutical and biotechnology companies continue to accelerate market demand globally.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
By Type Analysis
Predictive analytics platforms is projected to dominate this segment because they are capable of predicting the outcome of the trial, optimizing protocols and lowering the rate of failure. These technologies allow sponsors to process vast amounts of data, determine patient trends, and enhance decision-making processes. NLP tools are also becoming increasingly popular in deriving insights from unstructured clinical data, where real-world evidence systems are becoming increasingly important. Nonetheless, predictive and ML-driven solutions continue to dominate since their effect is directly on cost reduction, trial speed, and probability of success, which makes them the most prevalent among pharmaceutical and biotech organizations.
By Deployment Mode Analysis
Cloud-based solutions is expected to dominate the deployment segment as they are more scalable, flexible and economical. They provide real-time access to data, smooth cross-geographical collaboration, and quicker connection with clinical systems. Big data processing is also facilitated by cloud platforms, and is needed in AI-based trials. Data-sensitive environments are still managed using on-premises solutions, but the latter are losing their popularity. The new hybrid models are taking shape as a compromise whereby they provide security and scalability. Nevertheless, the growing requirement of remote trial management and decentralized trials persists in pushing robust leadership of cloud deployments.
By Clinical Trial Phase Analysis
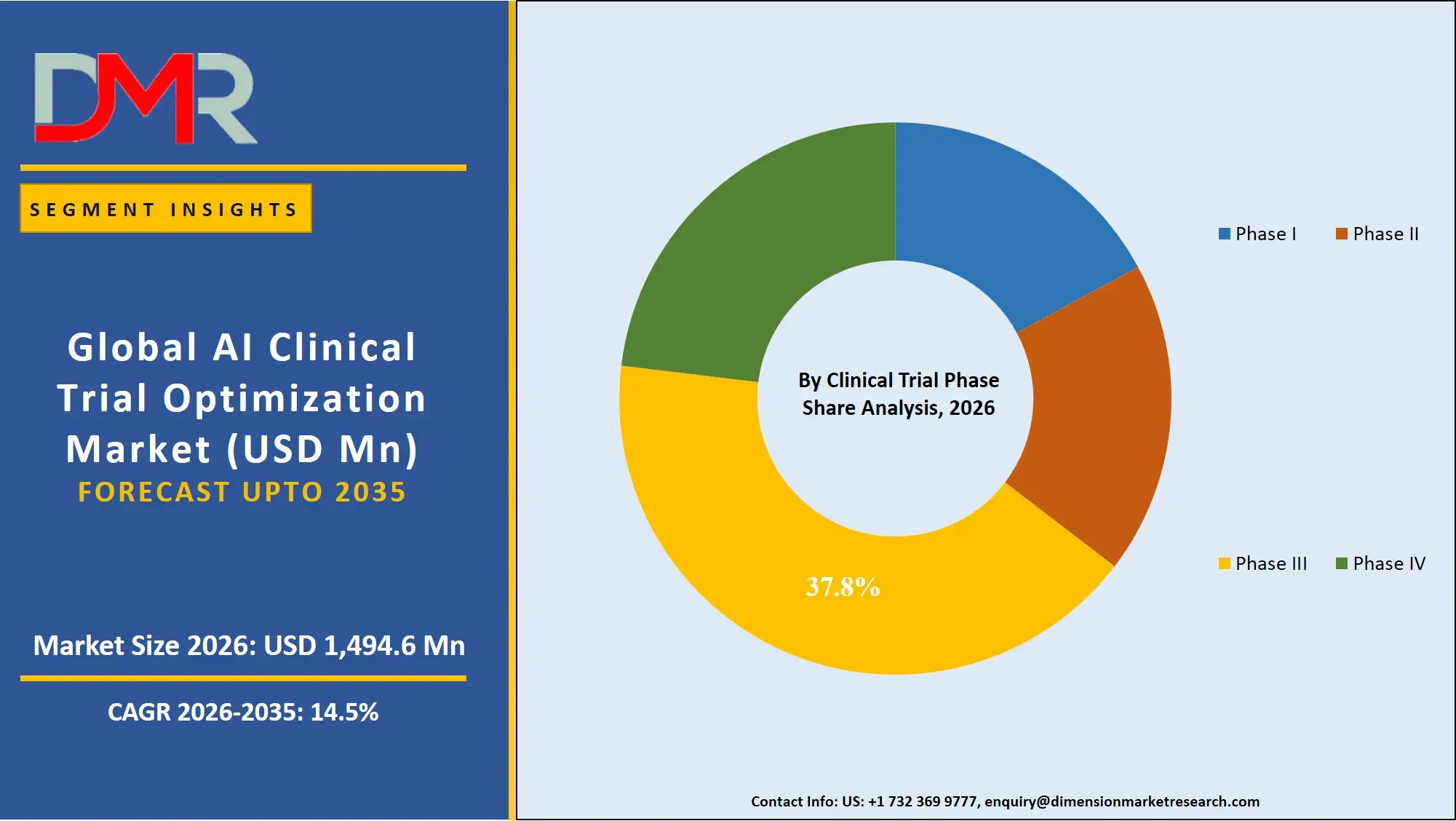
Phase III is expected to dominate this segment because it entails the highest patient population, the highest cost, and the most complicated data analysis needs. At this point, AI is important to maximize the selection of patients, track the progress of the trial, and forecast the results. Phase III is the most crucial phase of AI adoption as pharmaceutical companies invest a lot of money to guarantee the success of regulatory approval. Phase I and II, utilising AI in the safety and efficacy analysis segment, and Phase IV, in the post-market surveillance segment, the magnitude and financial interests of Phase III make it the leader.
By Therapeutic Area Analysis
Oncology is expected to dominate the therapeutic area segment as there are a large number of clinical trials, the rising cancer incidence, and the necessity to find innovative ways to treat it. In oncology studies, AI assists in the discovery of biomarkers, patient stratification, and customized treatment strategies. The intricacy of cancer studies, and the necessity of precision medicine, also contribute to the use of AI in this field. Although other fields such as cardiovascular, CNS, and infectious disease are also using AI, oncology is the most targeted given the high investment, regulatory interest, and pipeline of targeted therapies.
By Application Analysis
This segment is projected to be dominated by patient recruitment and retention because they deal with one of the most critical issues in clinical trials recruitment and retention of suitable participants. AI allows identifying potential patients in a big amount of data, enhancing the quality of matching, and shortening the recruitment process. This greatly reduces delays and costs of trials. Also of interest are trial design optimization and data management, however recruitment is the bottleneck of the greatest importance. Thus, patient enrollment efficiency solutions remain on the market as the most adopted and valuable.
By End User Analysis
Pharmaceutical and biotechnology companies are poised to dominate this segment as they have invested heavily in R&D and have been early adopters of AI technologies. Such organizations are extensively dependent on AI in order to speed up the drug development process, lower costs, and enhance success rates in trials. The other major users are contract research organizations (CROs), which use AI to improve their services. Research and data-generation are also produced in academic institutions and hospitals but the adoption there is relatively low. AI is slowly being incorporated in regulatory bodies. Nevertheless, pharma and biotech companies are the driving force of demand and innovation in this market.
The Global AI Clinical Trial Optimization Market Report is segmented on the basis of the following:
By Type
- Predictive Analytics Platforms
- Machine Learning Algorithms
- Natural Language Processing (NLP) Tools
- Computer Vision Systems
- Data Integration & Management Solutions
- Real-World Evidence (RWE) Platforms
- AI-powered Decision Support Tools
By Deployment Mode
- Cloud-Based Solutions
- On-Premises Solutions
- Hybrid Models
By Clinical Trial Phase
- Phase I
- Phase II
- Phase III
- Phase IV
By Therapeutic Area
- Oncology
- Cardiovascular Disorders
- CNS Disorders
- Infectious Diseases
- Metabolic Disorders
- Others
By Application
- Patient Recruitment & Retention
- Trial Design Optimization
- Site Selection & Monitoring
- Data Management & Analysis
- Safety & Risk Assessment
- Adverse Event Detection
- Drug Efficacy Prediction
By End User
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations (CROs)
- Academic & Research Institutions
- Hospitals & Clinics
- Regulatory Authorities
Regional Analysis
Leading Region by Market Share
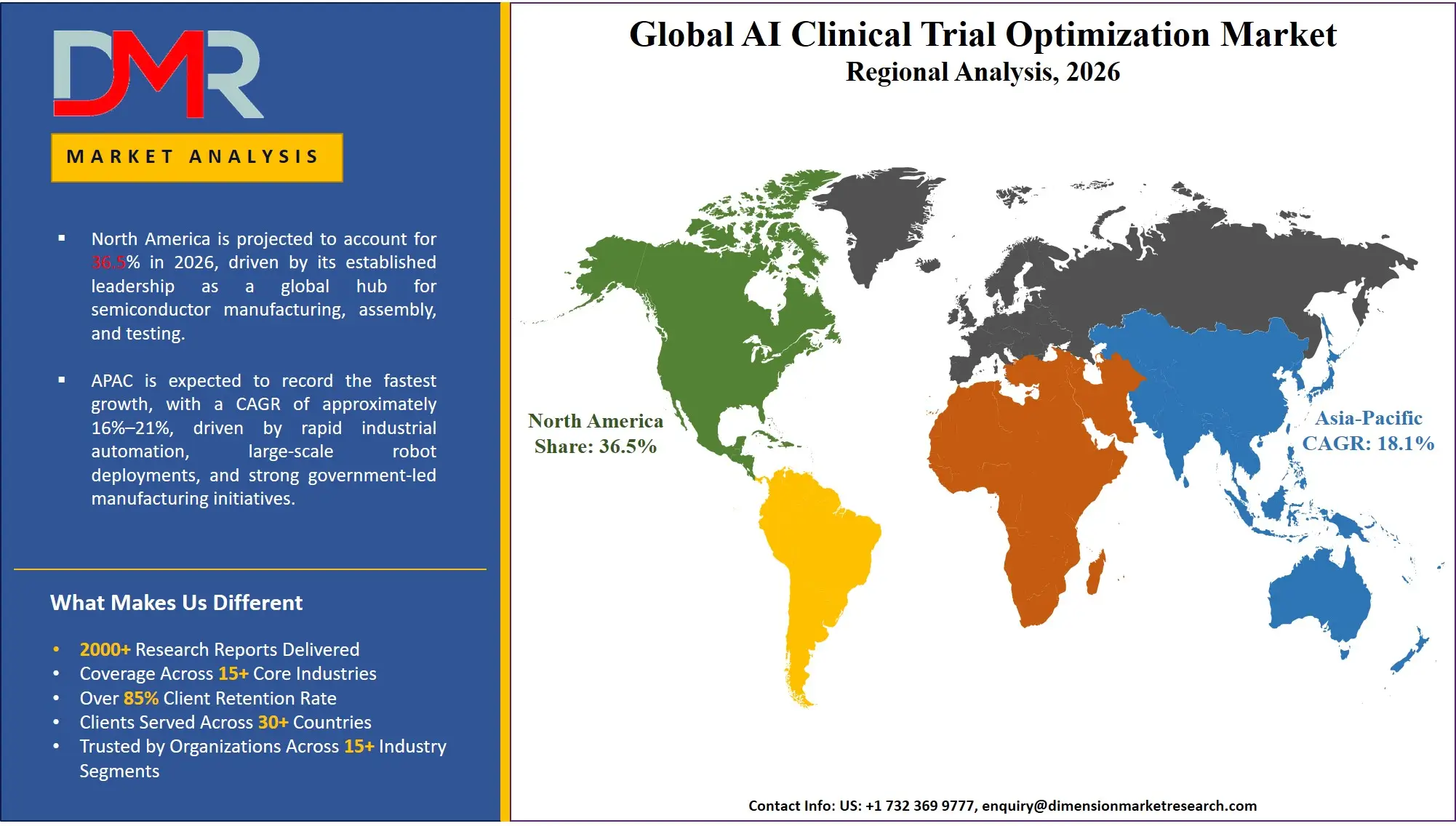
North America is poised to dominate the global AI clinical trial optimization market as it is projected to hold 38.5% of the global market by 2026. The largest stake in this market is held by the United States, which controls North America due to the unparalleled concentration of pharmaceutical R&D headquarters, the proactive position of the FDA on adopting the real-life evidence, and the vigorous digital transformation agendas of the top 20 pharmaceutical firms. It has an ecosystem of global CROs, AI biotech startups, and has a deep talent base in clinical data science and bioinformatics. Precision oncology, advanced CNS biomarkers, and the general conversion to decentralized trial models drive enterprise investment in predictive analytics and NLP tools, as well as RWE platforms, to continue being in demand. In addition, a strong venture capital ecosystem continues to fund new biotechs, which require specialized AI to get fast clinical proof-of-concept and safety compliance.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Fastest-Growing Regional Market
The Asia-Pacific region will be the fastest-growing AI clinical trial optimization market due to the government-led health data sweeps across India, China, Japan, and South Korea. The accelerated economic development, the emergence of a generic-to-innovator pharmaceutical shift, and the dynamic growth of the digital health economy are forcing traditional conglomerates and local innovators to abandon manual processes that are inefficient. Patient Recruitment and Retention tools based on AI are being sought after to enable these large organizations to tap into the gigantic, treatment-naive patient bases of the region. A critical shortage of skilled data scientists and AI-competent clinical operations experts in the area also necessitates the outsourcing of SaaS-based optimization solutions to CROs and vendors of digital health to close the talent gap and allow conducting clinical trials faster and with greater data-sensitivity.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The global AI clinical trial optimization market has evolved into an extremely dynamic competitive landscape with a diverse mix of global AI platform vendors, specialist CRO digital divisions, and specialized analytics consultancies. The success will be based on the deep strategic relationships with leading cloud hyperscalers and EHR data partners because they will create the required co-selling opportunities and access to longitudinal, organized patient data. The trend towards market consolidation is fast accelerating whereby the traditional clinical technology companies are buying NLP and predictive analytics specialty boutiques in order to survive. Validated prognostic algorithms and disease-specific patient-finding models are increasingly becoming proprietary intellectual property, rather than generic dashboard reporting or simple approaches to statistical programming.
Some of the prominent players in the Global AI Clinical Trial Optimization Market are:
- IQVIA
- Medidata
- Unlearn.ai
- Owkin
- Saama Technologies
- AiCure
- Deep 6 AI
- Exscientia
- Antidote Technologies
- BioSymetrics
- Euretos
- Ardigen
- Mendel AI
- Phesi
- ICON plc
- Parexel
- Syneos Health
- nference
- ConcertAI
- SAS Institute
- Other Key Players
Recent Developments
- January 2026: Medidata has declared it will launch a significant scale-up of its AI-based Synthetic Control Arm solution, a professional services and platform program to support oncology and rare disease sponsors to develop proprietary RWE models using its Data Integration and Management solutions and Trial Design Optimization expertise.
- November 2025: IQVIA enhanced its partnership with a major biopharma firm and launched a dedicated practice called AI-powered Site Selection & Monitoring and Patient Recruitment to help their clients transition to an adaptive Phase III neurology trial and remain compliant with other international data privacy laws.
- October 2025: Saama Technologies purchased a European NLP company to expand its AI-based Decision Support System and Data Management products to automate regulatory dossier, to serve the complex needs of pharmaceutical companies with fast compliance to EMA clinical study report submissions.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 1,494.6 Mn |
| Forecast Value (2035) |
USD 5,065.7 Mn |
| CAGR (2026–2035) |
14.5% |
| The US Market Size (2026) |
USD 483.9 Mn |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Segments Covered |
By Type, By Deployment Mode, By Clinical Trial Phase, By Therapeutic Area, By Application, and By End User |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
Frequently Asked Questions
How big is the Global AI Clinical Trial Optimization Market?
▾ The Global AI Clinical Trial Optimization market is poised to be valued at USD 1,494.6 million in 2026 and is projected to reach USD 5,065.7 million by 2035, driven by the universal need for specialized tools in patient recruitment, trial design, and real-world data integration.
What is the CAGR of the Global AI Clinical Trial Optimization Market from 2026 to 2035?
▾ The market is expected to grow at a CAGR of 14.5% from 2026 to 2035, reflecting the accelerating complexity of clinical protocols and the persistent shortage of internal data science talent in the life sciences industry.
What factors are driving the growth of the Global AI Clinical Trial Optimization Market?
▾ Key drivers include the life sciences data skills gap, the imperative to reduce clinical trial cycle times, the management complexity of multi-modal and decentralized trial data, and the surge in demand for predictive analytics amid evolving FDA and EMA guidance on digital health technologies.
Which region held the largest share of the AI Clinical Trial Optimization Market in 2026?
▾ North America is projected to held a substantial share of the market in 2026, driven by a mature pharmaceutical R&D ecosystem and aggressive enterprise investment in AI-driven patient recruitment and RWE platforms.
Which region is expected to grow the fastest in the AI Clinical Trial Optimization Market?
▾ The Asia-Pacific region is expected to grow the fastest, fueled by rapid digital transformation in Japan, China, and India, where AI-powered Patient Recruitment & Retention tools are critical for accessing large, diverse patient populations efficiently.
What are the major trends in the Global AI Clinical Trial Optimization Market?
▾ Major trends include the integration of Generative AI into trial simulation and dossier writing, the rise of federated learning for privacy-preserving data collaboration, the demand for therapeutic-area-specific AI solutions, and the focus on digital twin technology to simulate trial outcomes before execution.
Who are the key players in the Global AI Clinical Trial Optimization Market?
▾ Key players include AI-native firms like Saama Technologies, Unlearn.AI, and Deep 6 AI, as well as the digital analytics divisions of large CROs like IQVIA and Medidata, alongside specialized pure-play AI consultancies and tech giants like Verily.
How is the Global AI Clinical Trial Optimization Market segmented?
▾ The market is segmented by Type, Deployment Mode, Clinical Trial Phase, Therapeutic Area, Application, End User.