Market Overview
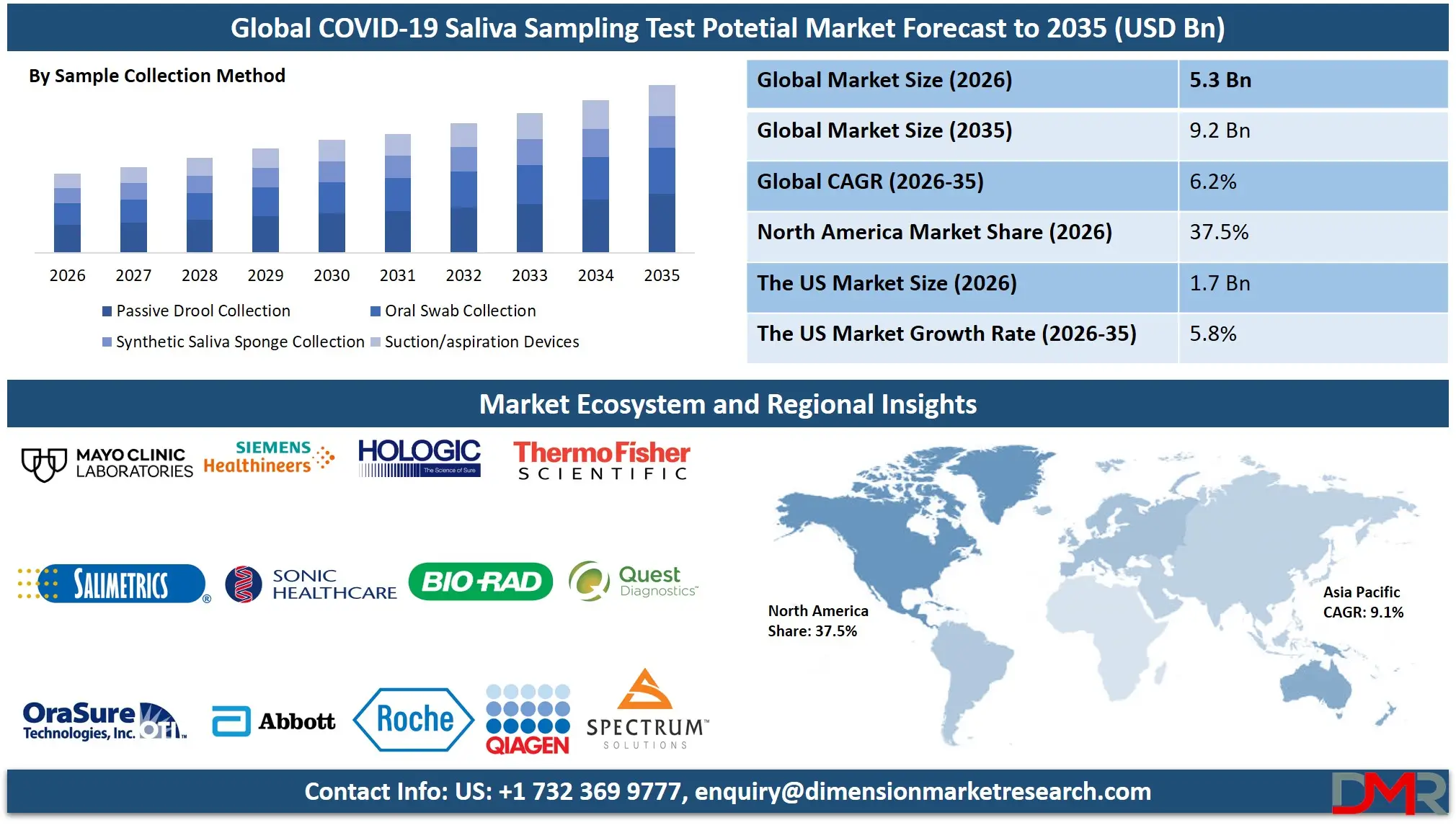
The Global COVID-19 Saliva Sampling Test Potential Market is set for substantial expansion, reaching an estimated USD 5.3 billion in 2026 and projected to grow at a moderate CAGR of 6.2% from 2026 to 2035, to the market value of USD 9.2 billion by 2035. This growth trajectory is fueled by the sustained need for non-invasive diagnostic solutions, rapid antigen testing kits, RT-PCR saliva-based assays, and at-home collection devices across healthcare systems.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Rising incidences of seasonal COVID-19 variants, influenza co-infections, and the growing emphasis on decentralized healthcare are compelling governments and private healthcare providers to maintain accessible and scalable testing infrastructure. The widespread adoption of telehealth services, remote patient monitoring, and workplace health screening programs is significantly increasing demand for reliable saliva sampling solutions.
Additionally, ongoing public health preparedness initiatives related to pandemic surveillance, school safety protocols, travel requirements, and corporate health mandates are further accelerating market adoption. Healthcare institutions are increasingly integrating saliva-based PCR tests, rapid molecular assays, and high-throughput automated collection systems to enhance diagnostic efficiency and reduce dependency on invasive nasal swabs.
Direct-to-consumer distribution models, pharmacy-based test kits, and scalable manufacturing capabilities are enabling diagnostic companies and public health authorities to implement cost-effective and widely accessible COVID-19 testing services. As respiratory illness surveillance becomes embedded in global public health infrastructure and population health monitoring continues to expand worldwide, the COVID-19 Saliva Sampling Test Potential Market is expected to witness sustained single-digit growth through 2035, driven by ongoing variant emergence and diagnostic preparedness initiatives.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Growing government investments in biosecurity frameworks, infectious disease rapid response systems, and cross-border health compliance verification further accelerate global adoption. However, barriers such as variable reimbursement policies, sensitivity variations compared to nasopharyngeal swabs, cold chain requirements for certain test kits, and the cyclical nature of pandemic waves remain. Despite these limitations, the convergence of molecular diagnostics, microfluidics, and digital health platforms positions saliva sampling as a cornerstone of future respiratory disease surveillance through 2035.
The US COVID-19 Saliva Sampling Test Potential Market
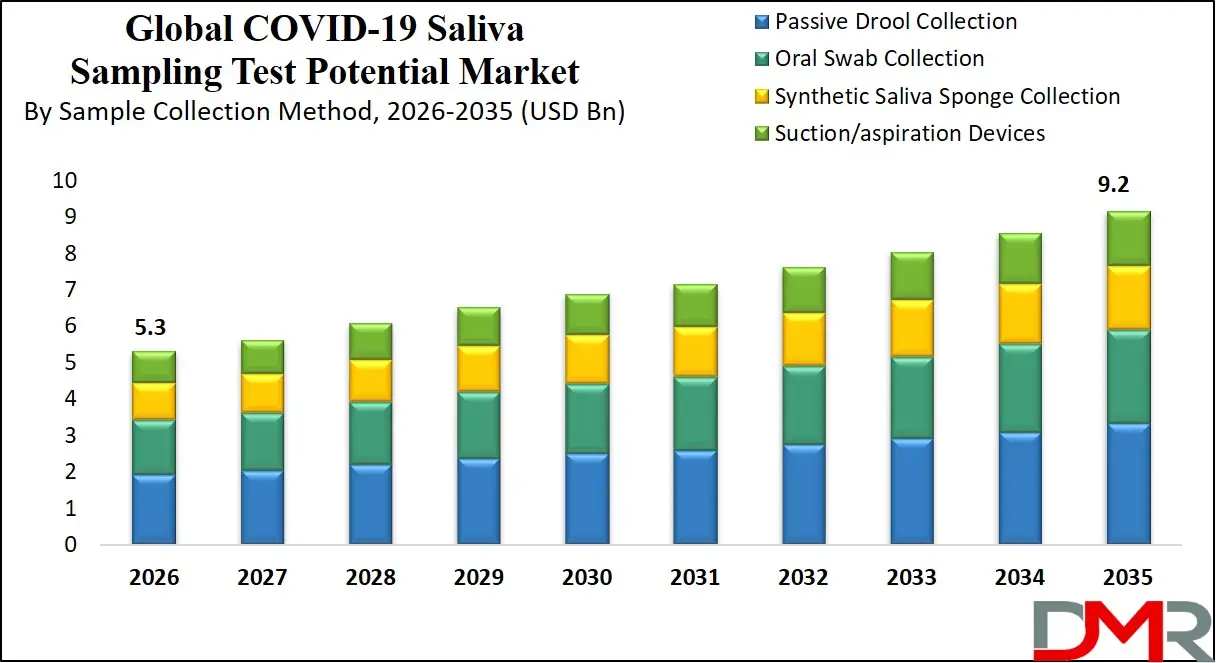
The U.S. COVID-19 Saliva Sampling Test Potential Market is projected to reach USD 1.7 billion in 2026 and grow at a CAGR of 5.8%, reaching USD 2.8 billion by 2035. The U.S. leads global adoption due to its well-established healthcare infrastructure, widespread insurance coverage for diagnostic testing, and strong public-private partnerships in pandemic response.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The proliferation of workplace testing programs in corporate America, university surveillance systems, and school district safety protocols, coupled with consumers' preference for painless collection methods, fuels demand for reliable saliva sampling solutions. Major diagnostic manufacturers, pharmacy chains, and healthcare providers such as Quest Diagnostics, CVS Health, and Kaiser Permanente are integrating advanced saliva collection technologies to support ongoing community testing needs.
U.S. regulatory support through FDA Emergency Use Authorizations (EUA) and CLIA laboratory standards, alongside frameworks like the Public Health Preparedness and Response Capabilities, encourages continued investment in saliva-based diagnostics. The market is witnessing a shift toward multiplex respiratory panels that include COVID-19 alongside influenza and RSV, enhancing clinical utility and year-round relevance. The emergence of new variants as a persistent public health concern has further intensified the focus on scalable, user-friendly collection methods, positioning the U.S. as a critical innovator in this space.
The Europe COVID-19 Saliva Sampling Test Potential Market
The Europe COVID-19 Saliva Sampling Test Potential Market is projected to be valued at approximately USD 1.1 billion in 2026 and is projected to reach around USD 1.6 billion by 2035, growing at a CAGR of about 4.1% from 2026 to 2035. Europe's steady demand is anchored by its integrated public health systems, cross-border travel policies, and the European Centre for Disease Prevention and Control (ECDC) surveillance recommendations.
Countries such as Germany, the U.K., France, and Italy are maintaining testing capacity through national health services, driven by a high emphasis on vulnerable population protection and variant monitoring. The U.K.'s UKHSA surveillance programs and the EU's HERA (Health Emergency Preparedness and Response Authority) initiatives further necessitate ongoing diagnostic readiness across member states.
Europe's universal healthcare coverage, aging population requiring continued protection, and established diagnostic supply chains drive the demand for accessible, validated saliva testing solutions. Funding for pan-European diagnostic preparedness and standardization of testing protocols across member states encourages cross-border data sharing and harmonized quality requirements. With a sophisticated public health infrastructure and a regulatory landscape that prioritizes patient safety and accessibility, Europe remains a highly advanced and essential region for saliva sampling services.
The Japan COVID-19 Saliva Sampling Test Potential Market
The Japan COVID-19 Saliva Sampling Test Potential Market is anticipated to be valued at approximately USD 280 million in 2026 and is expected to attain nearly USD 410 million by 2035, expanding at a CAGR of about 4.3% during the forecast period. Japan's aging population and a government push for "Community-based Integrated Care Systems" are driving the adoption of accessible and non-invasive diagnostic methods, making saliva testing a cornerstone of geriatric infection control.
The Ministry of Health, Labour and Welfare (MHLW) actively supports the development of streamlined diagnostic infrastructure, promoting the use of saliva collection for both clinical and home settings. Japan's leadership in precision manufacturing and high-quality healthcare standards provides a robust foundation for advanced collection devices, including automated systems and user-friendly kits.
Japan's concept of "Sustainable Healthcare for Super-Aged Society," driven by major healthcare corporations and prefectural health departments, integrates saliva testing into everything from nursing home surveillance to outpatient clinics. Companies are deploying enhanced saliva collection technologies to combat outbreaks in long-term care facilities and ensure the integrity of workplace testing programs in Tokyo's corporate sector and regional public health offices. Japan's cultural emphasis on precision and reliability positions it as a quality-focused market for diagnostic sampling solutions.
Global COVID-19 Saliva Sampling Test Potential Market: Key Takeaways
- Stable Global Market Growth Outlook: The Global COVID-19 Saliva Sampling Test Potential Market is expected to be valued at USD 5.3 billion in 2026 and is projected to reach USD 9.2 billion by 2035, showcasing steady demand supported by ongoing public health surveillance needs.
- Moderate CAGR Driven by Endemic Disease Management: The market is expected to grow at a CAGR of 6.2% from 2026 to 2035, fueled by sustained variant monitoring, integration with respiratory panel testing, and decentralized healthcare expansion.
- Steady Growth Trajectory in the United States: The U.S. COVID-19 Saliva Sampling Test Potential Market stands at USD 1.7 billion in 2026 and is projected to reach USD 2.8 billion by 2035, expanding at a CAGR of 5.8% due to established healthcare coverage and workplace safety programs.
- Regional Dominance: North America is expected to capture approximately 37.5% of the global market share in 2026, supported by robust healthcare spending, widespread insurance reimbursement, and advanced diagnostic infrastructure.
- Rapid Advancement in Collection Technologies: Innovations including microfluidic collection devices, ambient-temperature stable transport media, integrated lysis tubes, and digital health platform connectivity are significantly enhancing accuracy, ease-of-use, and scalability of saliva sampling services.
- Ongoing Pandemic Preparedness Boosts Adoption: Sustained global vigilance regarding emerging variants, seasonal surges, and the need for frictionless population screening is driving continued demand for reliable, non-invasive saliva testing solutions.
Global COVID-19 Saliva Sampling Test Potential Market: Use Cases
- Workplace Health Surveillance: Corporations maintain regular testing programs using saliva-based assays to ensure workforce safety, reduce absenteeism, and comply with occupational health guidelines.
- School and University Screening: Educational institutions implement routine saliva testing to detect asymptomatic cases, prevent outbreaks, and maintain in-person learning environments.
- Long-Term Care Facility Protection: Nursing homes and assisted living facilities utilize frequent saliva testing to protect vulnerable elderly populations from COVID-19 introductions.
- Travel and Border Control: Airports and travel authorities use rapid saliva tests for pre-departure and arrival screening, facilitating safe international movement.
- Telehealth-Integrated Home Testing: Patients consult with healthcare providers remotely and receive mail-in saliva collection kits for laboratory analysis, expanding diagnostic access.
Global COVID-19 Saliva Sampling Test Potential Market: Stats & Facts
World Health Organization (WHO)
- Over 95% of WHO member states have established ongoing respiratory pathogen surveillance systems post-pandemic.
- Saliva-based testing is recommended by WHO as an alternative collection method for COVID-19 diagnosis.
- More than 150 countries have incorporated saliva sampling into national COVID-19 testing guidelines.
U.S. Food and Drug Administration (FDA)
- Over 40 saliva-based COVID-19 tests have received Emergency Use Authorization since 2020.
- Saliva tests demonstrate 85-98% sensitivity compared to nasopharyngeal swabs in clinical studies.
Centers for Disease Control and Prevention (CDC)
- Approximately 35% of all COVID-19 tests administered in 2023 utilized saliva-based collection methods.
- Workplace testing programs account for an estimated 25 million annual test administrations in the U.S.
National Institutes of Health (NIH) - RADx Program
- The Rapid Acceleration of Diagnostics (RADx) initiative invested over USD 1.5 billion in innovative testing technologies, including saliva-based platforms.
- At-home saliva testing adoption increased by 300% during the Omicron variant surge.
European Centre for Disease Prevention and Control (ECDC)
- EU/EEA countries conduct over 10 million COVID-19 tests monthly during active surveillance periods.
- Saliva testing represents 20-30% of total diagnostic volume in several member states.
UK Health Security Agency (UKHSA)
- The U.K. processed over 500 million COVID-19 tests during the pandemic, with saliva sampling playing an increasing role in later phases.
- Winter respiratory surveillance programs include routine saliva testing in community settings.
Ministry of Health, Labour and Welfare (MHLW), Japan
- Japan authorized over 15 saliva-based COVID-19 test kits for clinical and home use.
- Saliva testing adoption in nursing homes reduced outbreak response time by 40%.
Global COVID-19 Saliva Sampling Test Potential Market: Market Dynamic
Driving Factors in the Global COVID-19 Saliva Sampling Test Potential Market
Sustained Need for Non-Invasive Testing Solutions
The growing preference for painless, self-administered collection methods among patients and healthcare providers is a major driver for saliva sampling tests. Nasopharyngeal swabs cause significant discomfort and require trained personnel, limiting scalability. Saliva collection eliminates these barriers, enabling broader testing access, higher compliance rates, and frequent repeat testing essential for outbreak control. This is particularly valuable in pediatric populations, elderly care facilities, and workplace screening programs.
Integration with Multiplex Respiratory Panels
Saliva-based testing benefits significantly from the shift toward comprehensive respiratory diagnostics that detect COVID-19, influenza A/B, and RSV simultaneously. Healthcare systems prefer multiplex assays for efficient patient triage and treatment decisions, particularly during respiratory season. Saliva's compatibility with these advanced molecular platforms extends its utility beyond pandemic-specific applications, ensuring sustained demand as part of routine diagnostic offerings.
Restraints in the Global COVID-19 Saliva Sampling Test Potential Market
Variable Sensitivity and Collection Consistency
The significant challenge of ensuring consistent sample quality across diverse self-collection scenarios creates quality control concerns. Factors such as timing since last food/drink intake, adequate saliva volume, and proper device handling can affect test sensitivity. Achieving accuracy comparable to healthcare worker-collected nasopharyngeal swabs requires rigorous user instructions and device design optimization, which can be difficult to standardize across global populations.
Reimbursement and Funding Uncertainty
Healthcare reimbursement policies for COVID-19 testing vary significantly across countries and are subject to change as public health emergency declarations expire. In many regions, the transition from government-funded free testing to commercial insurance or out-of-pocket payment models has reduced testing volumes. This funding unpredictability creates market volatility and challenges for manufacturers planning long-term production capacity.
Opportunities in the Global COVID-19 Saliva Sampling Test Potential Market
Expansion into Routine Respiratory Illness Surveillance
The integration of saliva testing into seasonal respiratory illness monitoring programs represents major growth opportunities. Healthcare systems require ongoing surveillance infrastructure for early detection of outbreaks, variant tracking, and vulnerable population protection. Saliva-based multiplex panels that simultaneously detect multiple pathogens offer clinical and economic advantages, positioning them for permanent placement in diagnostic formularies.
Direct-to-Consumer and Pharmacy Channel Growth
The expansion of over-the-counter test kit availability through pharmacy chains, e-commerce platforms, and direct-to-consumer channels creates new market opportunities. Consumers value the convenience of home testing with laboratory-confirmed results through mail-in models. Partnerships with retail pharmacy giants, telehealth providers, and corporate wellness programs can drive volume while reducing reliance on government contracts.
Trends in the Global COVID-19 Saliva Sampling Test Potential Market
Digital Health Integration
The integration of saliva testing with smartphone apps, QR code-based result delivery, and digital health pass platforms is gaining significant traction. This trend enables seamless reporting to public health authorities, employers, and schools while providing users with convenient access to verified results. Digital connectivity also supports epidemiological surveillance and real-time outbreak monitoring.
Sustainable and Ambient-Temperature Stable Products
Manufacturers are developing collection devices that eliminate cold chain requirements and reduce plastic waste. Ambient-temperature stable transport media simplifies logistics, reduces costs, and expands access in resource-limited settings. Eco-friendly materials and reduced packaging are increasingly important differentiators in procurement decisions.
Global COVID-19 Saliva Sampling Test Potential Market: Research Scope and Analysis
By Sample Collection Method Analysis
The passive drool collection segment is projected to dominate the Global COVID-19 Saliva Sampling Test Potential Market, accounting for the largest revenue share compared to swab-based or sponge-based collection devices. This dominance is primarily driven by the simplicity, cost-effectiveness, and established clinical validation of direct saliva collection methods. Healthcare providers and diagnostic laboratories across public health systems, hospital networks, and commercial lab chains require collection systems that ensure sample integrity while minimizing complexity.
Modern passive drool collection devices provide advanced features such as integrated stabilization buffers, volume indication markings, and leak-proof transport containers. These features significantly reduce pre-analytical errors and ensure reliable molecular testing results. Diagnostic manufacturers and laboratory service providers prefer standardized collection systems that offer compatibility with high-throughput automated platforms, enabling efficient processing of large sample volumes.
Additionally, the expansion of at-home collection programs and mail-in testing services has increased reliance on user-friendly passive drool devices. Subscription-based testing models and direct-to-consumer distribution further strengthen the passive drool segment's dominance, as companies can deploy solutions without extensive user training.
While oral swab and synthetic sponge collection methods offer certain advantages in specific pediatric or geriatric applications, they typically serve niche segments rather than act as primary revenue generators. As respiratory surveillance becomes embedded in global public health infrastructure and laboratories prioritize operational efficiency, the passive drool collection segment is expected to maintain its leadership position due to proven reliability, user acceptance, and manufacturing scalability.
By Test Type Analysis
RT-PCR (Reverse Transcription Polymerase Chain Reaction) molecular testing is expected to dominate the test type segment due to its gold-standard accuracy, regulatory acceptance, and suitability for confirmatory diagnosis. Healthcare systems and public health authorities worldwide continue to prioritize PCR-based methods for symptomatic testing, outbreak investigation, and vulnerable population screening.
RT-PCR-based saliva testing remains popular because it combines the accuracy of molecular diagnostics with the convenience of non-invasive collection. It is extensively used by hospital laboratories, reference labs, public health departments, and employer testing programs. The high sensitivity and specificity of PCR methods are essential for detecting low viral loads in presymptomatic or asymptomatic individuals.
Regulatory requirements in healthcare reimbursement and international travel frameworks mandate PCR confirmation in many contexts, further driving sustained adoption. As multiplex panels incorporating influenza and RSV become standard, PCR platforms offer the flexibility to expand test menus without changing collection methods.
Although rapid antigen tests are gaining traction for screening applications due to speed and cost advantages, RT-PCR continues to dominate in clinical decision-making, outbreak confirmation, and regulatory compliance contexts. Its established role in diagnostic algorithms and laboratory infrastructure ensures continued leadership in this segment.
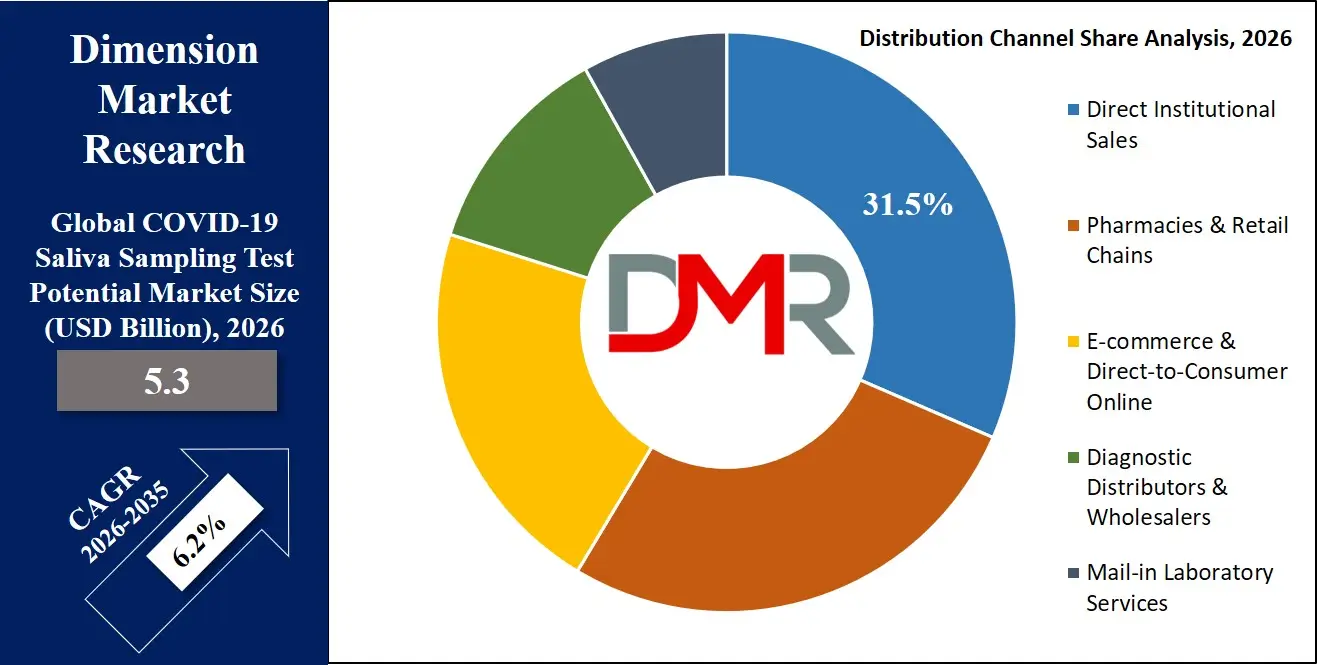
By Distribution Channel Analysis
Direct institutional sales is forecasted to dominate the distribution channel segment, driven by procurement practices of large healthcare systems, government health agencies, and corporate testing programs. Public health departments, hospital networks, and large employers purchase testing supplies through established vendor relationships and competitive bidding processes.
Government procurement for national stockpiles, pandemic preparedness programs, and public health surveillance represents a significant revenue stream. Diagnostic manufacturers maintain dedicated sales teams and distributor networks to serve these institutional customers, offering volume pricing, technical support, and supply chain reliability.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Pharmacy and retail channels have grown significantly with the expansion of over-the-counter testing, but institutional purchasing continues to dominate in terms of revenue due to higher volumes and contract values. Government tenders and healthcare system contracts provide predictable, multi-year demand that supports manufacturing investment.
While e-commerce and direct-to-consumer channels are expanding, particularly for repeat purchasers and convenience buyers, they typically serve individual consumers rather than large-scale programs. Consequently, direct institutional sales remain the primary distribution channel and are expected to maintain dominance over the forecast period.
By End-User Analysis
Hospitals and diagnostic laboratories is projected to dominate the end-user segment due to high testing volumes, regulatory testing requirements, and established laboratory infrastructure. Hospital systems, reference laboratories, and public health labs process millions of diagnostic tests annually, requiring reliable and scalable saliva collection systems.
These institutions prioritize accuracy, regulatory compliance, and operational efficiency. Regulatory mandates for reportable disease surveillance, infection control protocols, and transplant/immunocompromised patient management require robust testing capabilities, prompting healthcare facilities to maintain comprehensive diagnostic offerings. Additionally, hospital networks often serve as reference centers for surrounding communities, necessitating collection systems capable of serving diverse patient populations.
Hospitals and laboratories also adopt automated extraction and PCR platforms compatible with saliva samples, driving demand for validated collection devices. Their purchasing volume and quality requirements make them anchor customers for diagnostic manufacturers.
While home testing and workplace screening are growing segments due to decentralization trends, their testing volumes and clinical confirmation requirements remain lower compared to institutional healthcare settings. Consequently, hospitals and diagnostic laboratories continue to generate the majority of market revenue and are expected to sustain their dominant position.
The Global COVID-19 Saliva Sampling Test Potential Market Report is segmented on the basis of the following:
By Sample Collection Method
- Passive Drool Collection
- Oral Swab Collection
- Synthetic Saliva Sponge Collection
- Suction/aspiration Devices
By Test Type
- RT-PCR (Real-Time Polymerase Chain Reaction)
- Rapid Antigen Testing
- LAMP (Loop-Mediated Isothermal Amplification)
- Next-Generation Sequencing (for variant surveillance)
By Distribution Channel
- Direct Institutional Sales
- Pharmacies & Retail Chains
- E-commerce & Direct-to-Consumer Online
- Diagnostic Distributors & Wholesalers
- Mail-in Laboratory Services
By End-User
- Hospitals & Clinics
- Diagnostic Laboratories
- Home Testing/Self-Collection
- Corporate & Workplace Testing Programs
- Academic & Research Institutes
- Government & Public Health Laboratories
- Travel & Hospitality Sector
Impact of Artificial Intelligence in the Global COVID-19 Saliva Sampling Test Potential Market
- AI for Test Utilization Optimization: AI analyzes epidemiological data, seasonal patterns, and regional outbreak signals to optimize testing resource allocation across healthcare systems, ensuring supplies reach highest-need areas.
- AI-Driven Result Interpretation: Machine learning algorithms assist in interpreting complex amplification curves and antigen test results, reducing false positives/negatives and supporting clinical decision-making.
- Predictive Demand Forecasting: AI-powered systems continuously learn from variant spread patterns, travel data, and social mobility metrics to predict testing demand surges, enabling manufacturers to optimize production and distribution.
- AI-Based Supply Chain Management: AI analyzes real-time inventory levels, transportation logistics, and expiration date tracking to ensure reliable test kit availability while minimizing waste.
- Learning from Global Surveillance Data: AI systems aggregate and anonymize testing data across global networks to identify emerging variant hotspots and changing transmission patterns, enabling proactive public health response.
Global COVID-19 Saliva Sampling Test Potential Market: Regional Analysis
Region with the Largest Revenue Share
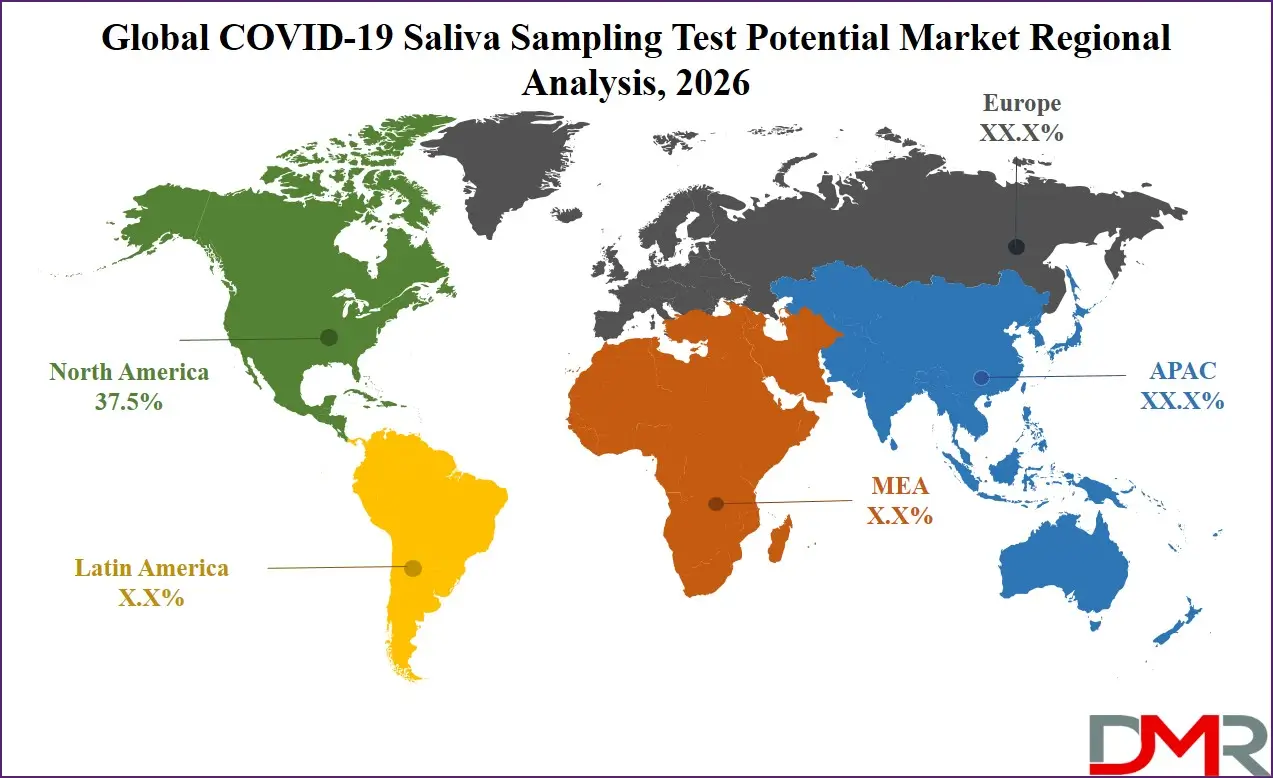
North America is projected to dominate the regional segment with the highest market share as it is anticipated to hold 37.5% of the total market revenue by the end of 2026, due to advanced healthcare infrastructure, widespread insurance coverage, and sustained public health surveillance programs. The region is home to leading diagnostic manufacturers and laboratory service providers, driving technological advancements in saliva collection systems. Strong healthcare spending and regulatory frameworks regarding laboratory standards compel continued investment in quality diagnostics. High public awareness regarding respiratory illness prevention further strengthens demand.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The United States, in particular, accounts for the largest share within North America due to its large healthcare system and established private insurance reimbursement. Although Asia Pacific is the fastest-growing region, North America continues to hold the largest revenue share due to early adoption of saliva-based methods and strong public-private partnerships.
Region with the Highest CAGR
Asia-Pacific holds the highest CAGR and is poised to achieve rapid market share growth due to its massive population base, expanding middle class, and significant government investments in public health infrastructure. Countries like India, China, Indonesia, and Vietnam are strengthening their diagnostic capabilities following pandemic experiences. India's Pradhan Mantri Swasthya Suraksha Yojana (PMSSY) and similar health security initiatives across Southeast Asia are creating fertile ground for diagnostic adoption. The region's price sensitivity is being addressed through local manufacturing, cost-optimized collection devices, and partnerships between global diagnostic companies and regional distributors. This, combined with immense population health needs, positions APAC as the fastest-growing market for saliva sampling test systems.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Global COVID-19 Saliva Sampling Test Potential Market: Competitive Landscape
The Global COVID-19 Saliva Sampling Test Potential Market is moderately fragmented, featuring a mix of global diagnostic manufacturers, molecular diagnostics specialists, and collection device innovators. Leading players like Quest Diagnostics, Laboratory Corporation of America, and Sonic Healthcare leverage their extensive laboratory networks and patient access to offer saliva testing as part of comprehensive diagnostic services. Pure-play molecular diagnostics companies such as Thermo Fisher Scientific, Roche Diagnostics, and Hologic are driving market dynamics with advanced PCR platforms and automated collection systems.
Diagnostic manufacturers like Abbott Laboratories, Becton Dickinson, and Danaher Corporation (Cepheid) play crucial roles as integrated solution providers, while specialized collection device companies such as Spectrum Solutions, DNA Genotek, and Salimetrics are innovating in sample integrity and user experience. Regional diagnostic distributors and public health laboratory consortia are also active, forming partnerships to ensure local manufacturing and supply chain resilience.
Some of the prominent players in the Global COVID-19 Saliva Sampling Test Potential Market are:
- Quest Diagnostics Incorporated
- Laboratory Corporation of America Holdings
- Thermo Fisher Scientific Inc.
- Roche Diagnostics (F. Hoffmann-La Roche Ltd)
- Abbott Laboratories
- Danaher Corporation (Cepheid)
- Hologic, Inc.
- Becton, Dickinson and Company
- Bio-Rad Laboratories, Inc.
- Qiagen N.V.
- Siemens Healthineers AG
- Sonic Healthcare Limited
- Spectrum Solutions LLC
- DNA Genotek Inc. (OraSure Technologies)
- Salimetrics, LLC
- Lucira Health (Vault Health)
- Clinical Reference Laboratory, Inc.
- Eurofins Scientific SE
- ARUP Laboratories
- Mayo Clinic Laboratories
- Other Key Players
Recent Developments in the Global COVID-19 Saliva Sampling Test Potential Market
- November 2025: Thermo Fisher Scientific launched its next-generation saliva collection kit featuring ambient-temperature stable transport media and integrated QR code tracking for digital health platform connectivity.
- October 2025: Quest Diagnostics expanded its at-home collection portfolio with a new multiplex saliva test detecting COVID-19, influenza A/B, and RSV simultaneously, enhancing respiratory illness management.
- September 2025: Abbott Laboratories announced a partnership with a leading pharmacy chain to distribute rapid molecular saliva tests through 10,000+ retail locations nationwide.
- August 2025: Roche Diagnostics completed the acquisition of a microfluidics technology firm to enhance its saliva testing platform's automation capabilities and sample processing efficiency.
- July 2025: A Bill & Melinda Gates Foundation grant was awarded to develop low-cost saliva collection devices for resource-limited settings, expanding global diagnostic access.
- June 2025: Spectrum Solutions partnered with a major Asian public health agency to provide saliva collection systems for nationwide respiratory surveillance programs.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 5.3 Bn |
| Forecast Value (2035) |
USD 9.2 Bn |
| CAGR (2026–2035) |
6.2% |
| The US Market Size (2026) |
USD 1.7 Bn |
| Historical Data |
2020 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Sample Collection Method (Passive Drool Collection, Oral Swab Collection, Synthetic Saliva Sponge Collection, Suction/Aspiration Devices), By Test Type (RT-PCR (Real-Time Polymerase Chain Reaction), Rapid Antigen Testing, LAMP (Loop-Mediated Isothermal Amplification), Next-Generation Sequencing), By Distribution Channel (Direct Institutional Sales, Pharmacies & Retail Chains, E-commerce & Direct-to-Consumer Online, Diagnostic Distributors & Wholesalers, Mail-in Laboratory Services), By End-User (Hospitals & Clinics, Diagnostic Laboratories, Home Testing/Self-Collection, Corporate & Workplace Testing Programs, Academic & Research Institutes, Government & Public Health Laboratories, Travel & Hospitality Sector) |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
| Prominent Players |
Quest Diagnostics Incorporated, Laboratory Corporation of America Holdings, Thermo Fisher Scientific Inc., Roche Diagnostics (F. Hoffmann-La Roche Ltd), Abbott Laboratories, Danaher Corporation (Cepheid), Hologic, Inc., Becton, Dickinson and Company, Bio-Rad Laboratories, Inc., Qiagen N.V., Siemens Healthineers AG, Sonic Healthcare Limited, Spectrum Solutions LLC, DNA Genotek Inc. (OraSure Technologies), Salimetrics, LLC, Lucira Health (Vault Health), Clinical Reference Laboratory, Inc., Eurofins Scientific SE, ARUP Laboratories, Mayo Clinic Laboratories, and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Global COVID-19 Saliva Sampling Test Potential Market?
▾ The Global COVID-19 Saliva Sampling Test Potential Market size is estimated to have a value of USD 5.3 billion in 2026 and is expected to reach USD 9.2 billion by the end of 2035.
What is the growth rate in the Global COVID-19 Saliva Sampling Test Potential Market?
▾ The market is growing at a CAGR of 6.2 percent over the forecasted period of 2026-2035.
What is the size of the US COVID-19 Saliva Sampling Test Potential Market?
▾ The US COVID-19 Saliva Sampling Test Potential Market is projected to be valued at USD 1.7 billion in 2026. It is expected to witness subsequent growth as it holds USD 2.8 billion in 2035 at a CAGR of 5.8%.
Which region accounted for the largest Global COVID-19 Saliva Sampling Test Potential Market?
▾ North America is expected to have the largest market share in the Global COVID-19 Saliva Sampling Test Potential Market with a share of about 37.5% in 2026.
Who are the key players in the Global COVID-19 Saliva Sampling Test Potential Market?
▾ Some of the major key players in the Global COVID-19 Saliva Sampling Test Potential Market are Quest Diagnostics, Thermo Fisher Scientific, Roche Diagnostics, Abbott Laboratories, Danaher Corporation, Spectrum Solutions, and many others.