Market Overview
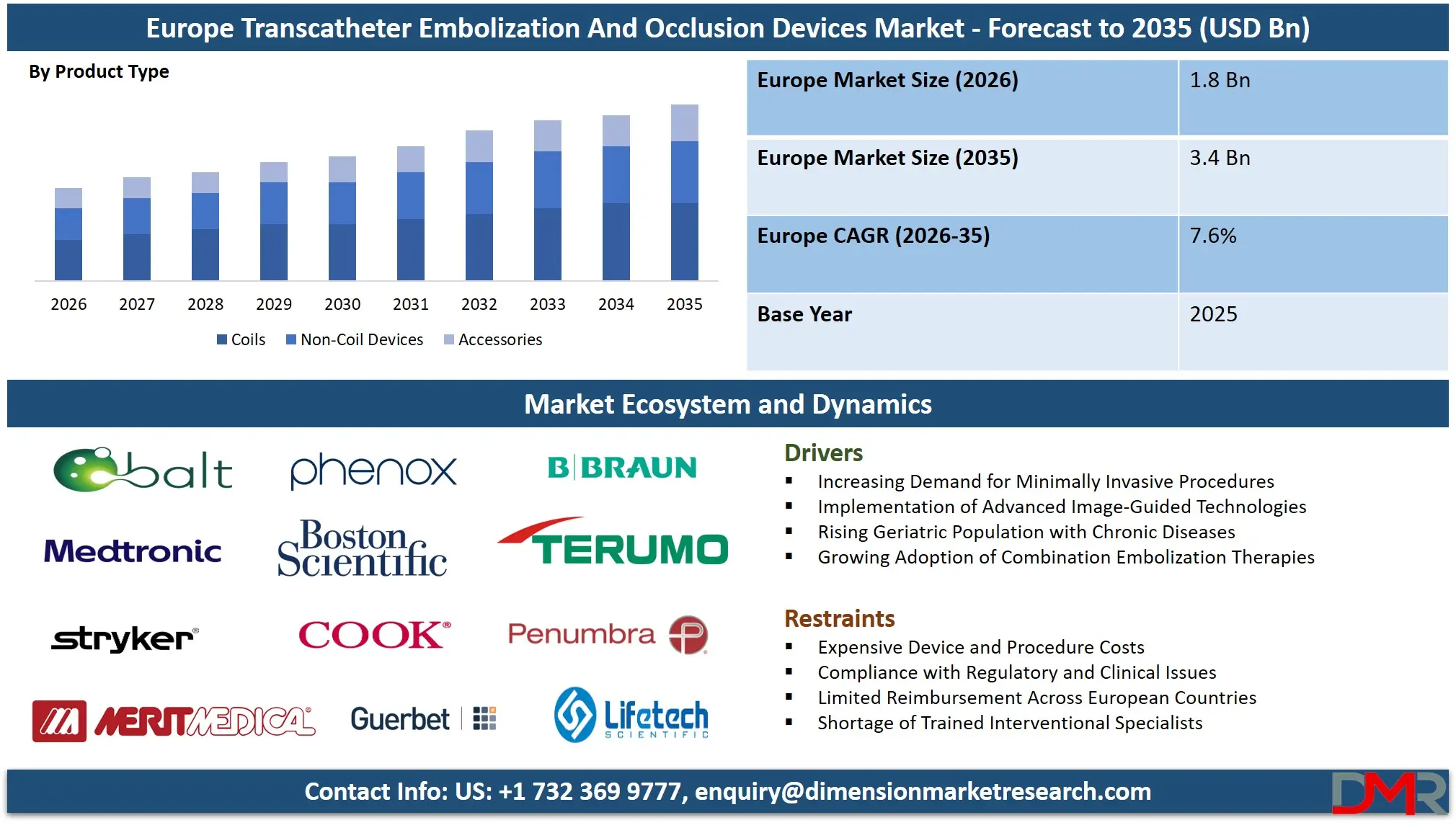
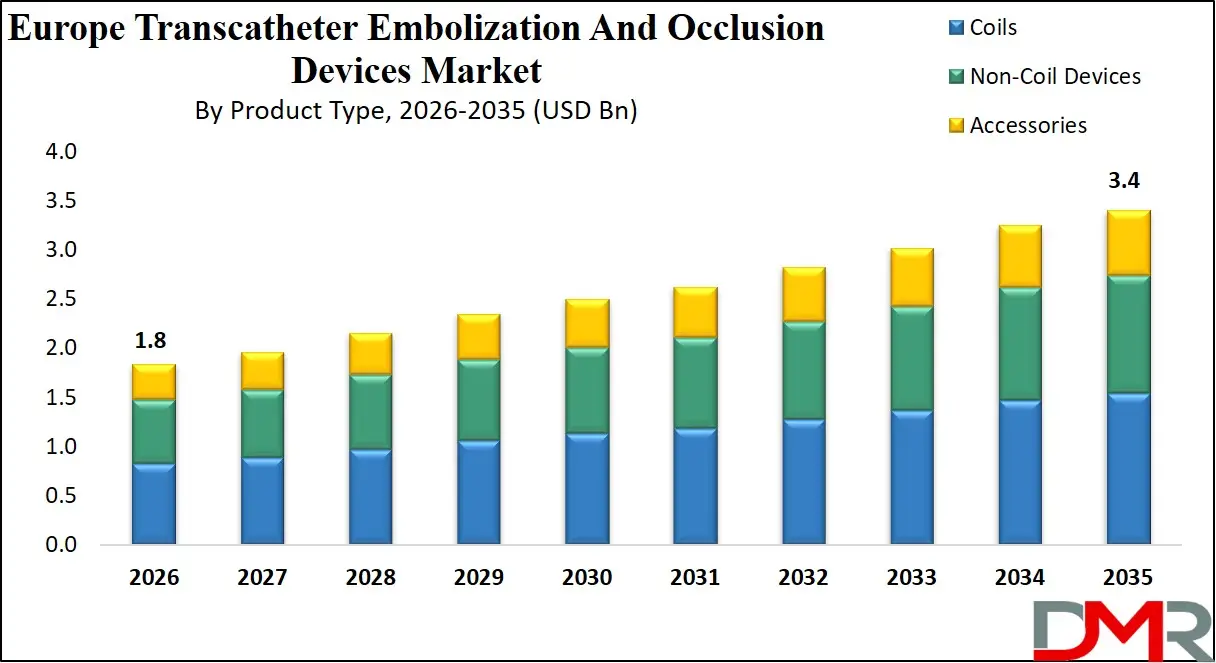
The Europe Transcatheter Embolization And Occlusion Devices Market size is projected to attain a value of USD 1.8 billion in 2026, witnessing growth at a CAGR of 7.6% to reach a value of USD 3.4 billion in 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
A transcatheter embolization and occlusion device (TEOD) is a minimally invasive device designed to block or reduce blood flow through selected vessels, intended for the treatment of aneurysms, tumors, vascular malformations and hemorrhages. The Europe Transcatheter Embolization And Occlusion Devices Market is projected to grow with a significant CAGR in coming years, owing to the rising prevalence of liver cancer, cerebral aneurysms, and uterine fibroids, coupled with increasing adoption of minimally invasive procedures in Germany, France, and the United Kingdom. The increased growth is due to the increasing geriatric population, development in bio-resorbable polymers as well as automation in interventional radiology across Europe.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Europe Transcatheter Embolization And Occlusion Devices Market: Key Takeaways and Other Influencing Factors
- Strong Market Growth Projections: The Europe Transcatheter Embolization And Occlusion Devices Market is expected to grow from USD 1.8 billion in 2026 to USD 3.4 billion by 2035, reflecting steady long-term demand growth driven by minimally invasive embolization and occlusion applications across key medical specialties.
- Strong Growth Momentum: The market is expected to grow at a CAGR of 7.6% during 2026–2035, supported by rising adoption of embolization devices in interventional oncology, neurovascular procedures, and peripheral vascular disease management across Europe.
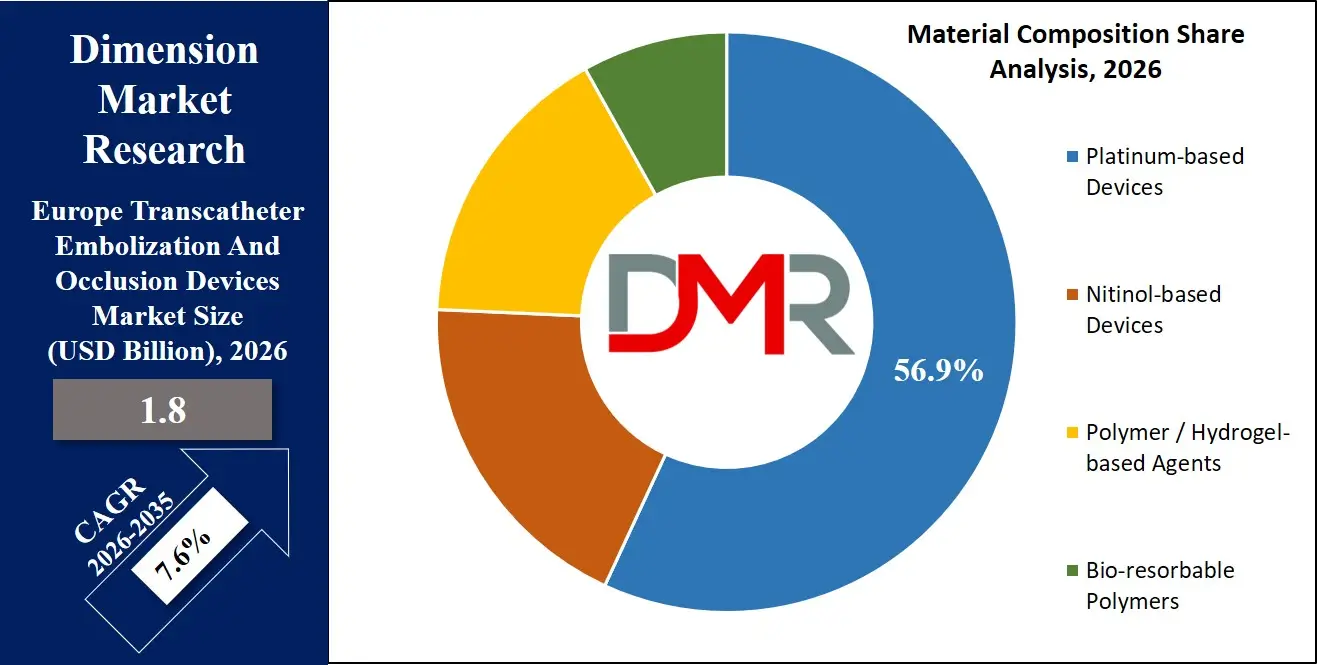
- Product Type and Material Prevalence: The coil product type is projected to capture approximately 45.0% of the coil product type market share in 2026, whereas platinum-based coils will account for nearly 56.9% of the material composition market share, emphasizing a high clinical inclination towards radiopaque, precise delivery systems within complicated vascular architecture.
- Application and End User Leadership: The oncology application is set to capture around 41.9% of the application market share in 2026, while the hospitals & clinics end-user category will command nearly 71.8% of the end-user market share, owing to the high volume of procedures, advanced interventional radiology suites, and broad adoption in cancer and vascular care pathways.
- European Society of Radiology (ESR): More than 75% of interventional radiology departments in Europe conduct transcatheter embolizations on a regular basis (2023-2025), thus depicting an excellent demand scenario of embolization and occlusion devices in the regional vascular intervention setup.
- Health and Food Safety Directorate at the European Commission: More than 80% of the interventional cardiology centers in EU member nations conduct embolization on a minimal intervention basis to ensure safe vascular intervention treatment of patients through occlusion devices.
- European Association of Urology (EAU): EAU has provided guidelines on embolization therapy, stating that embolization could be the preferred option of treatment for uterine fibroids and benign prostatic hyperplasia for all the members.
Impact of the Iran conflict on the Europe Transcatheter Embolization And Occlusion Devices Market
The Iran war is causing disruption of supply chain in European market, which makes the cost of medical logistics more expensive and delays some of the raw materials for platinum and nitinol-based embolization devices. The Iran conflict, integrated with the increase in energy prices and medical-grade material costs, is increasing the costs of production of transcatheter devices by doing this raising their price. Mean effect on the market due to diversified sourcing; but long term market variability may lead to price pressure and market stagnation.
Europe Transcatheter Embolization And Occlusion Devices Market: Use Cases
- Tumor Embolization in Oncology: Transcatheter embolization devices are used for targeted occlusion of blood flow to liver, kidney, and lung tumors with paper addressing their use in chemoembolization and radioembolization to maximize tumor kill while limiting systemic exposure.
- Cerebral Aneurysm and AVM Treatment: Detachable coils and flow diverting devices can be used to occlude cerebral aneurysms and arteriovenous malformations by precise endovascular deployment, improving patient outcomes and reducing recovery times.
- Post-Partum Hemorrhage and Uterine Fibroid Management: These types of devices are applied to perform uterine artery embolization, to control bleeding, preserve fertility, and assist improved gynecological outcomes.
- Peripheral vascular and trauma embolization: Transcatheter embolization is increasingly being used in support of peripheral artery disease, GI bleeding, and splenic injury to enhance patient survival and recognize high-risk early on.
Europe Transcatheter Embolization And Occlusion Devices Market: Market Dynamics
Driving Factors in the Europe Transcatheter Embolization And Occlusion Devices Market
Increasing Demand for Minimally Invasive Procedures
The growing preference for endovascular interventions and outpatient treatments are leading to the demand of precise embolization devices, catheters, and occlusion systems in Europe. This can be further reinforced by the increasing consciousness of patient recovery and preventive interventional programs.
Implementation of Advanced Image-Guided Technologies
3D angiography and digital subtraction angiography are growing, increasing the demand to use embolization devices and real-time deployment monitoring systems to track outcomes, enhancing operational results and procedural safety. Adoption is also increasing with an increased penetration of robotic-assisted interventional platforms.
Restraints in the Europe Transcatheter Embolization And Occlusion Devices Market
Expensive Device and Procedure Costs
Complex embolization systems are expensive in terms of devices, imaging integration, and training, meaning they cannot be implemented in cost-sensitive healthcare environments. In the case of large scale deployment, budget constraints in public health systems further limit it.
Compliance with Regulatory and Clinical Issues
The stringent medical device regulations and clinical validation requirements in Europe pose difficulties in the management of embolization device approvals that influence the deployment of new bio-resorbable and liquid embolic systems. Providers are burdened with compliance requirements like MDR and CE marking.
Opportunities in the Europe Transcatheter Embolization And Occlusion Devices Market
Development of Bio-Resorbable and Drug-Eluting Embolics
Increased attention to temporary embolization and targeted therapy is opening up the potential of long-term vascular healing, remote biodegradation, and patient-centric occlusion technologies. This tendency is favorable to reduced foreign body reaction and improved clinical outcomes.
AI and Predictive Analytics Integration
Implementation of AI-based procedure planning and predictive outcome solutions is improving the process of identifying optimal deployment strategies early, making more intelligent clinical choices and personalized embolization. Digital vascular biomarkers are continuously undergoing innovation, which is broadening application.
Trends in the Europe Transcatheter Embolization And Occlusion Devices Market
Gradual Shift to Bio-resorbable Polymer Agents
Bioresorbable polymers-based embolic materials have been attracting attention not only because of their ability to provide better safety characteristics, but also because they allow the gradual degradation of the material, thereby reducing possible negative effects. Moreover, new research is being conducted for improving the efficiency and applicability of bioresorbable materials within the framework of embolization.
Rising Adoption of Liquid Embolics and Microspheres
The increased application of liquid embolic materials and microspheres in embolizations contributes to the precision and efficiency of embolization therapy, particularly in cancer treatment, due to the enhanced accuracy in treating intricate vascular issues. This development is further bolstered by the current clinical inclination towards less-invasive procedures, which promote quicker recovery and lower risks.
Europe Transcatheter Embolization And Occlusion Devices Market: Research Scope and Analysis
By Product Type Analysis
The Europe Transcatheter Embolization And Occlusion Devices Market is projected to be dominated by Coils, detachable coils to be specific, with approximately close to 45.0% share in 2026 due to its superior precision, controlled deployment, and increasing use in cerebral aneurysm and peripheral embolization. They can offer consistent vascular occlusion and greater navigation compatibility, which is increasing adoption. Pushable coils, however, are still common in peripheral applications where cost efficiency is prioritized. Their demand is being supported by strong clinical familiarity and established use in trauma, but is slowly being deterred by limited control in favor of detachable coils. Non-coil devices including flow diverters and liquid embolics are gaining share due to complex anatomy management.
By Material Composition Analysis
The platinum-based devices segment holds the dominant share of approximately 56.9% in the Europe Transcatheter Embolization And Occlusion Devices Market in 2026 due to high demand for radiopaque, biocompatible solutions in neurovascular and oncological embolization with precise, continuous occlusion.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
These are commonly employed in aneurysm coiling and tumor feeding vessel occlusion. The nitinol-based devices segment is increasing due to the increased use of flow diverting stents and self-expanding occluders. The growing attention to minimally invasive procedures and vascular reconstruction is also contributing to its growth.
By Application Analysis
Oncology is estimated to hold a 41.9% market share in the Europe Transcatheter Embolization And Occlusion Devices Market by 2026 due to the high prevalence of tumor devascularization using embolization and occlusion devices in conditions such as hepatocellular carcinoma, renal tumors, and metastases, with the devices extensively used in chemoembolization and radioembolization procedures; on the other hand, neurology is witnessing rapid growth due to increasing acceptance of cerebral aneurysm coiling and AVM embolization, whereas peripheral vascular disease and urology, which include fibroid and prostate artery embolization, are steadily contributing to the demand for devices that require significant vascular mobility.
By End-User Analysis
The Hospitals & Clinics segment is expected to account for around 71.8% share in the Europe Transcatheter Embolization And Occlusion Devices Market in 2026, driven by high procedure volumes, advanced interventional radiology suites, and strong use of embolization devices for inpatient and outpatient care, where both coil and non-coil systems enable accurate vascular occlusion; in contrast, the Ambulatory Surgical Centers segment is expanding steadily due to rising demand for same-day interventional procedures, followed by Others, which are benefiting from cost-effective outpatient embolization management and increasing adoption of minimally invasive solutions across Europe.
The Europe Transcatheter Embolization And Occlusion Devices Market Report is segmented on the basis of the following:
By Product Type
- Coils
- Pushable Coils
- Detachable Coils
- Non-Coil Devices
- Flow Diverting Devices
- Embolization Particles
- Liquid Embolics
- Other Non-Coil Devices
- Accessories
By Material Composition
- Platinum-based Devices
- Nitinol-based Devices
- Polymer / Hydrogel-based Agents
- Bio-resorbable Polymers
By Application
- Oncology
- Peripheral Vascular Disease
- Neurology
- Urology
- Others
By End User
- Hospitals & Clinics
- Ambulatory Surgical Centers
- Others
Impact of Artificial Intelligence on the Europe Transcatheter Embolization And Occlusion Devices Market
The Europe Transcatheter Embolization And Occlusion Devices Market is changing through the use of artificial intelligence, which allows detecting deployment anomalies and device inefficiencies in a faster and more precise manner due to the development of advanced angiographic data analysis.
The AI-based algorithms enhance the accuracy of the operations by detecting minute changes in flow dynamics that cannot be detected during a manual examination. It can be integrated with smart delivery systems to monitor embolization in real-time and provide early intervention alerts. AI-based predictive analytics is not only increasing the level of risk assessment and individual patient planning in complex embolization, but also decreasing the amount of work done by operators and speeding up clinical decision making.
By Region
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Europe Transcatheter Embolization And Occlusion Devices Market: Competitive Landscape
The Europe Transcatheter Embolization And Occlusion Devices Market is very competitive, with the constant innovation of bio-resorbable agents and digital embolization technologies.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Firms are emphasizing on the development of sophisticated detachable coils, liquid embolics, and AI-based planning solutions to consolidate in the market. The competition is being heightened by strategic alliances, product innovations, and expansion in interventional radiology systems, and facilitating market growth.
Some of the prominent players in the Europe Transcatheter Embolization And Occlusion Devices Market are:
- Balt Group (Balt Extrusion SAS)
- Phenox GmbH
- Acandis GmbH & Co. KG
- B. Braun Melsungen AG
- Guerbet SA
- GEM S.r.l.
- Boston Scientific Corporation
- Medtronic plc
- Terumo Corporation (MicroVention, Inc.)
- Stryker Corporation
- Abbott Laboratories
- Johnson & Johnson (Cerenovus)
- Penumbra, Inc.
- Cook Medical LLC
- Merit Medical Systems, Inc.
- Invamed Saglik Urunleri Sanayi ve Ticaret A.S.
- Lifetech Scientific Corporation
- Wallaby Medical Technology Co., Ltd.
- NeuroSafe Medical Co., Ltd.
- Other Key Players
Recent Developments in the Europe Transcatheter Embolization And Occlusion Devices Market
- February 2026: Medtronic plc strengthened its neurovascular capabilities through positive clinical data from the EMBOLISE trial, supporting the effectiveness of its Onyx liquid embolic system in embolization procedures.
- January 2026: Boston Scientific Corporation expanded its embolization portfolio by securing CE Mark approval for its Embold detachable coil system, enhancing procedural precision and control in peripheral vascular embolization across European markets.
- September 2025: Penumbra, Inc. expanded its embolization portfolio by securing CE Mark approval for its SwiftPAC neuroembolization coil, enabling commercial availability in Europe and supporting improved precision and control in neurovascular embolization procedures.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 1.8 Bn |
| Forecast Value (2035) |
USD 3.4 Bn |
| CAGR (2026–2035) |
7.6% |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors, etc. |
| Segments Covered |
By Product Type (Coils, Non-Coil Devices, Accessories), By Material Composition (Platinum-based Devices, Nitinol-based Devices, Polymer / Hydrogel-based Agents, Bio-resorbable Polymers), By Application (Oncology, Peripheral Vascular Disease, Neurology, Urology, Others), By End User (Hospitals & Clinics, Ambulatory Surgical Centers, Others) |
| Regional Coverage |
Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe |
| Prominent Players |
Balt Group (Balt Extrusion SAS), phenox GmbH, Acandis GmbH & Co. KG, B. Braun Melsungen AG, Guerbet SA, GEM S.r.l., Boston Scientific Corporation, Medtronic plc, Terumo Corporation (MicroVention, Inc.), Stryker Corporation, Abbott Laboratories, Johnson & Johnson (Cerenovus), Penumbra, Inc., Cook Medical LLC, Merit Medical Systems, Inc., Invamed Saglik Urunleri Sanayi ve Ticaret A.S., Lifetech Scientific Corporation, Wallaby Medical Technology Co., Ltd., NeuroSafe Medical Co., Ltd., and Other Key Players. |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Europe Transcatheter Embolization And Occlusion Devices Market?
▾ The Europe Transcatheter Embolization And Occlusion Devices Market size is estimated to have a value of USD 1.8 billion in 2026 and is expected to reach USD 3.4 billion by the end of 2035.
Who are the key players in the Europe Transcatheter Embolization And Occlusion Devices Market?
▾ Some of the major key players in the Europe Transcatheter Embolization And Occlusion Devices Market are Medtronic plc, Boston Scientific Corporation, Terumo Corporation, Stryker Corporation, Johnson & Johnson, Cook Medical LLC, and many others.
What is the growth rate in the Europe Transcatheter Embolization And Occlusion Devices Market?
▾ The market is growing at a CAGR of 7.6% over the forecasted period.