Market Overview
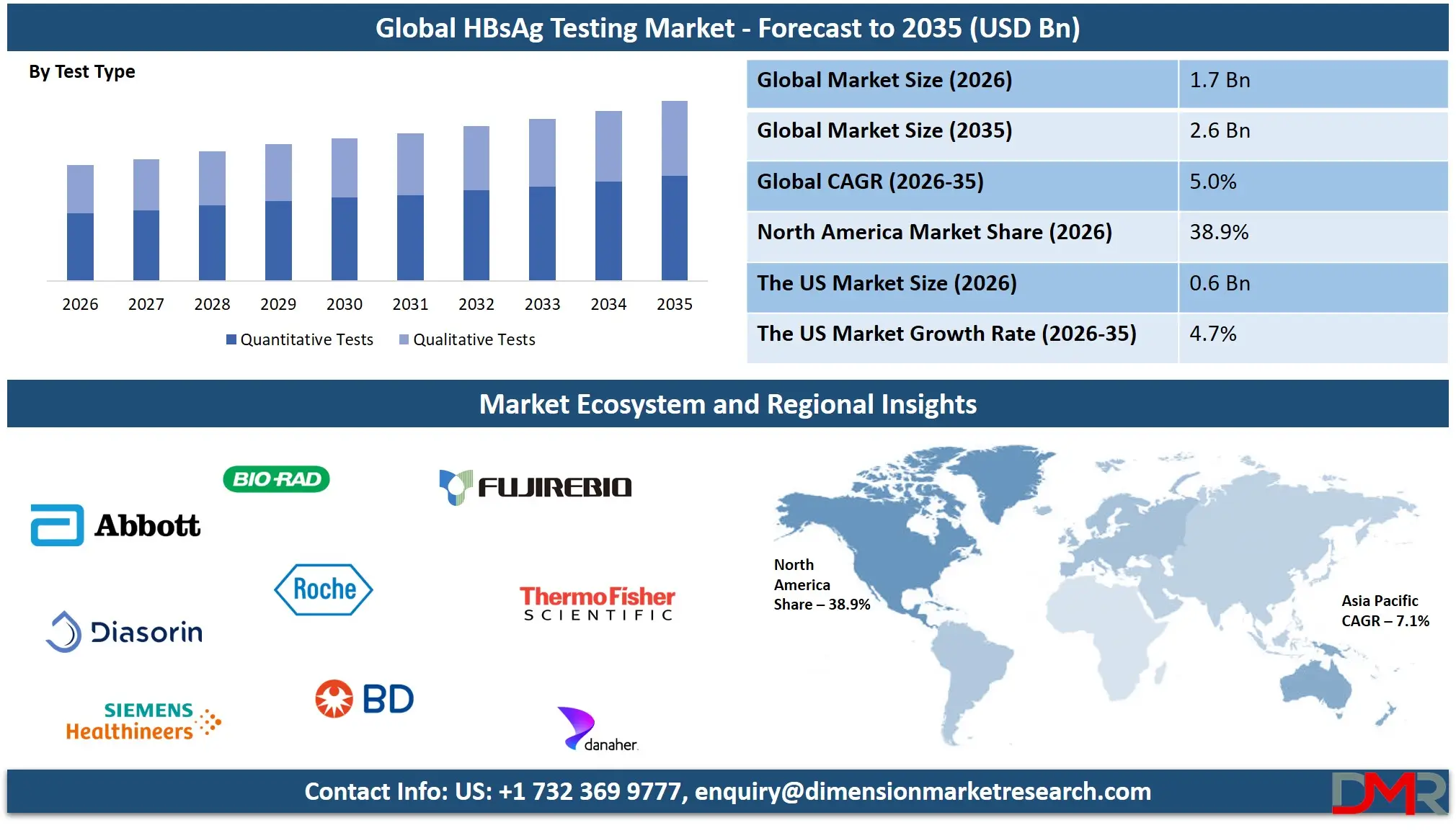
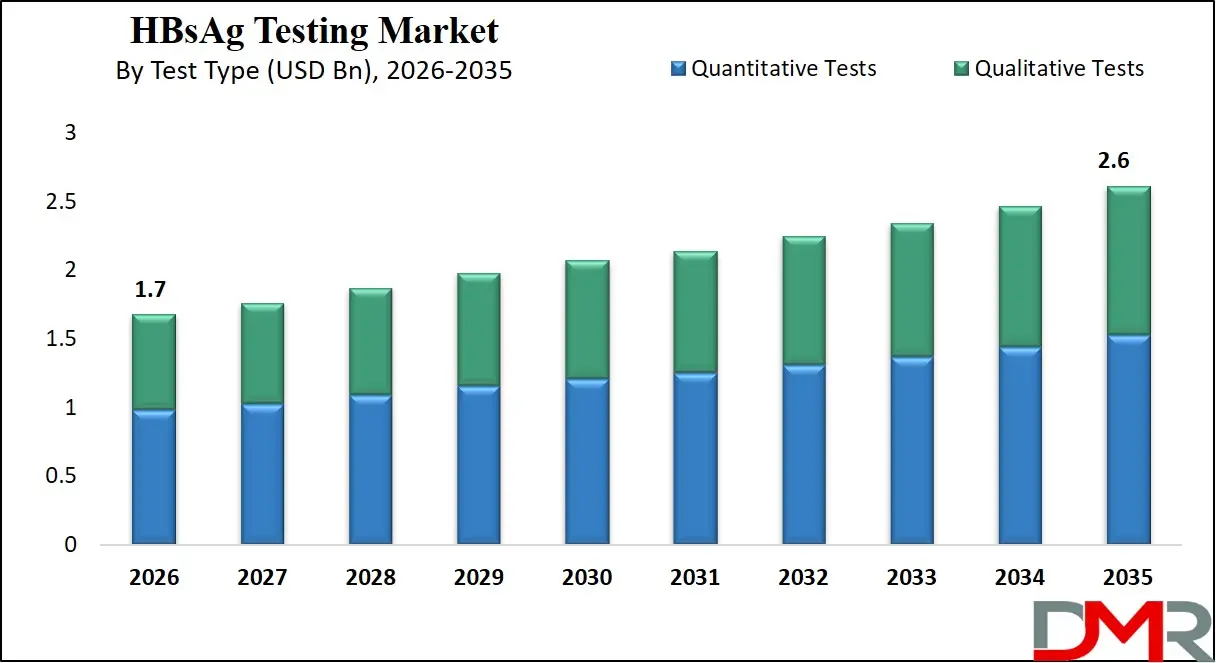
The Global HBsAg Testing Market size is projected to reach USD 1.7 billion in 2026 and grow at a compound annual growth rate of 5.0% to reach a value of USD 2.6 billion in 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
HBsAg refers to the Hepatitis B surface antigen, a protein found on the surface of the hepatitis B virus that is present in the blood during active infection. The detection of this antigen plays a crucial role in identifying individuals infected with hepatitis B and is widely used in clinical diagnostics, blood donor screening, and epidemiological surveillance. Testing methods typically involve immunoassay-based technologies such as ELISA, chemiluminescent assays, rapid diagnostic kits, and molecular techniques that enable accurate identification of viral antigens in serum, plasma, or whole blood samples. The presence of HBsAg is considered a primary indicator of both acute and chronic hepatitis B infection, making it an essential biomarker in viral hepatitis diagnostics.
Development in immunoassay technologies, molecular diagnostics, and rapid testing solutions have significantly improved detection sensitivity and turnaround time. Growing emphasis on early disease detection, improved blood safety protocols, and expanded prenatal screening programs has accelerated adoption of these testing methods across healthcare systems worldwide. Increasing awareness about viral hepatitis and rising screening initiatives supported by global health organizations have further strengthened the importance of HBsAg testing in disease control strategies.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
In recent years, significant progress has been observed in diagnostic automation, point-of-care testing capabilities, and laboratory integration. Diagnostic manufacturers are investing heavily in advanced analyzers, high-throughput systems, and multiplex testing platforms. Healthcare providers are increasingly integrating screening programs into routine medical care, particularly for pregnant women and blood donors. Expanding diagnostic infrastructure and growing collaborations between research institutes and healthcare organizations are further shaping the evolution of testing technologies and accessibility.
The US HBsAg Testing Market
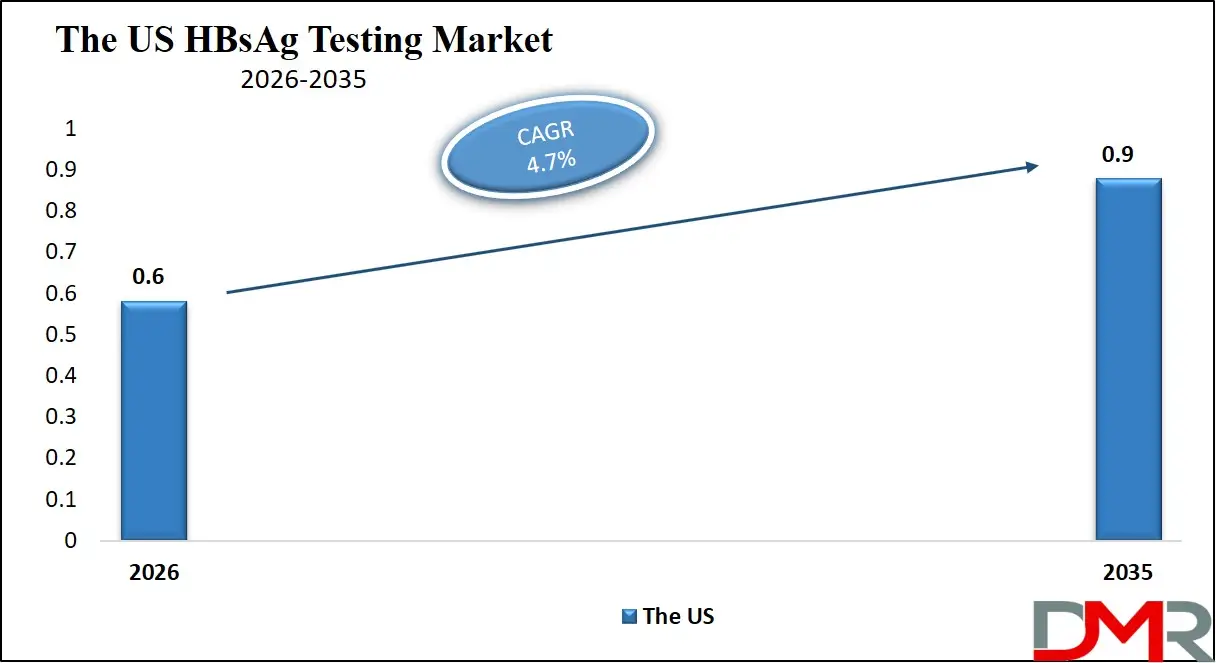
The US HBsAg Testing Market size is projected to reach USD 600 million in 2026 at a compound annual growth rate of 4.7% over its forecast period.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US demonstrates a highly structured environment for HBsAg testing due to strong public health policies, advanced laboratory infrastructure, and extensive screening programs. The country prioritizes hepatitis B detection through blood donor screening, prenatal testing, and routine diagnostics within hospitals and specialized laboratories. High adoption of automated immunoassay systems and molecular diagnostic platforms enables rapid and highly accurate results. The presence of major diagnostic manufacturers and research institutions contributes to continuous innovation in testing technologies. Government agencies and healthcare organizations actively promote hepatitis elimination strategies, encouraging broader screening initiatives among high-risk populations, immigrants from endemic regions, and healthcare workers.
Europe HBsAg Testing Market
Europe HBsAg Testing Market size is projected to reach USD 374 million in 2026 at a compound annual growth rate of 4.5% over its forecast period.
Europe represents a mature diagnostic environment where HBsAg testing plays a critical role in hepatitis control and prevention programs. Strong healthcare systems, universal screening policies, and increasing investment in advanced diagnostic technologies support market development across the region. Countries such as Germany, France, and the United Kingdom emphasize blood safety through mandatory screening protocols. The European focus on infectious disease monitoring and early detection has accelerated the adoption of high-throughput automated analyzers and molecular assays. Regional public health initiatives aligned with hepatitis elimination strategies encourage early diagnosis and treatment, while research institutions continue to contribute to improvements in diagnostic sensitivity and testing efficiency.
Japan HBsAg Testing Market
Japan HBsAg Testing Market size is projected to reach USD 68 million in 2026 at a compound annual growth rate of 4.9% over its forecast period.
Japan maintains a well-established diagnostic infrastructure with a strong emphasis on infectious disease screening and preventative healthcare. HBsAg testing is widely implemented in hospitals, clinical laboratories, and public health screening programs. The country has invested significantly in automated laboratory equipment and highly sensitive immunoassay technologies, enabling efficient large-scale testing. Prenatal screening and blood safety programs represent major application areas. Continuous research and development by domestic diagnostic manufacturers have resulted in highly advanced analyzers and reagents. Government healthcare initiatives aimed at reducing the burden of viral hepatitis further promote early detection and monitoring, strengthening the role of HBsAg testing across the national healthcare system.
HBsAg Testing Market: Key Takeaways
- Market Growth: The HBsAg Testing Market size is expected to grow by USD 4.9 billion, at a CAGR of 4.5%, during the forecasted period of 2027 to 2035.
- By Test Type: The quantitative tests segment is anticipated to get the majority share of the HBsAg Testing market in 2026.
- By Sample Type: The serum segment is expected to get the largest revenue share in 2026 in the HBsAg Testing market.
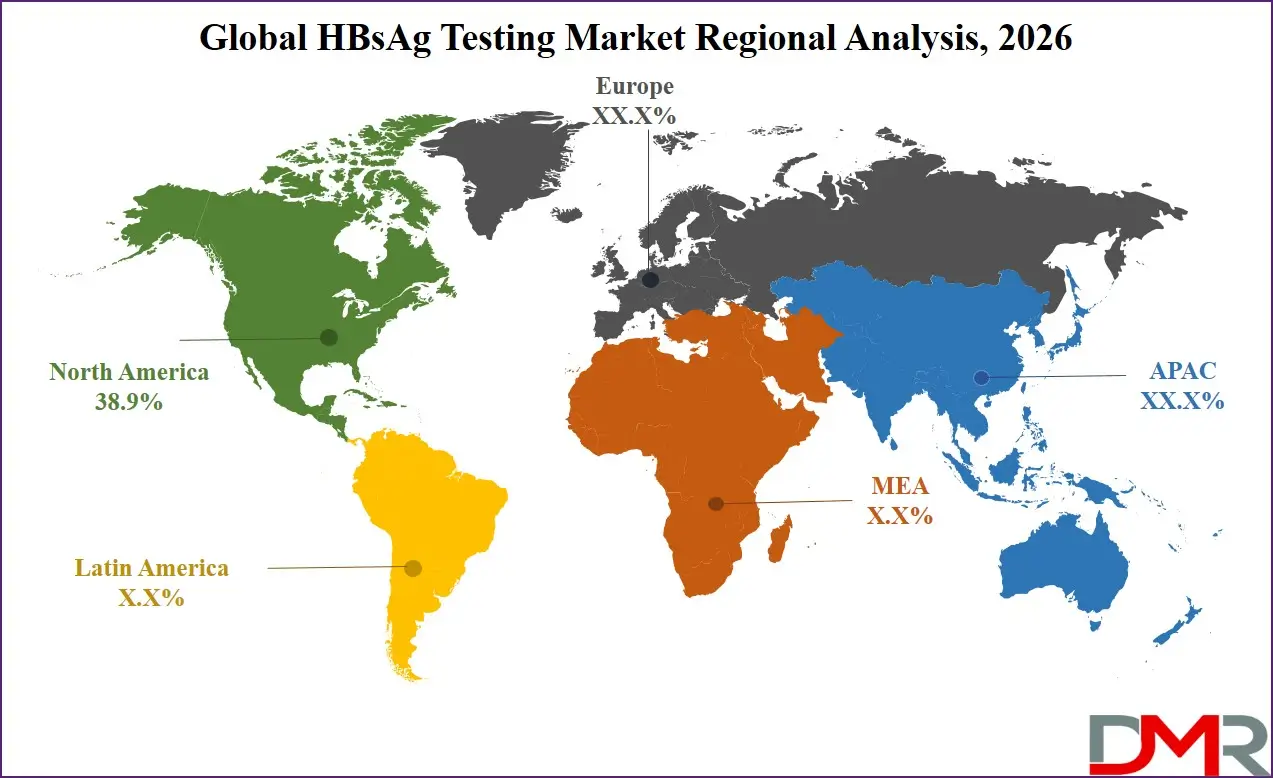
- Regional Insight: North America is expected to hold a 38.9% share of revenue in the global HBsAg Testing market in 2026.
- Use Cases: Some of the use cases of HBsAg Testing include prenatal screening, diagnostic testing, and more.
HBsAg Testing Market: Use Cases
- Blood Donor Screening: Blood banks rely on HBsAg testing to identify hepatitis B infection in donors before transfusion. This ensures safe blood supply and prevents transmission of viral infections during transfusion procedures. Rapid and high-throughput testing technologies help screen large donor volumes efficiently.
- Diagnostic Testing: Hospitals and diagnostic laboratories use HBsAg testing as an initial screening tool for suspected hepatitis B infection. It assists physicians in confirming infection status and determining the need for further viral load or liver function testing.
- Prenatal Screening: Pregnant women are routinely screened using HBsAg tests to detect hepatitis B infection early. Early identification allows healthcare providers to implement preventive measures that reduce the risk of mother-to-child transmission.
- Epidemiological Surveillance: Public health organizations conduct HBsAg testing during population surveys to track hepatitis B prevalence. This data helps governments design targeted vaccination and treatment programs.
- Occupational Health Monitoring: Healthcare workers and laboratory staff may undergo periodic HBsAg screening due to higher exposure risk. Early detection ensures timely treatment and prevents workplace transmission.
- Clinical Research: Research institutes utilize HBsAg testing to study hepatitis B virus behavior, vaccine efficacy, and antiviral therapies. The test provides valuable biomarkers for monitoring infection dynamics.
- Point-of-Care Diagnostics: Rapid HBsAg tests are used in remote or resource-limited settings where laboratory infrastructure is limited. These portable diagnostic tools enable quick screening and early disease detection.
Stats & Facts
- World Health Organization reported in 2024 that approximately 296 million people globally are living with chronic hepatitis B infection.
- World Health Organization estimated in 2024 that around 1.2 million new hepatitis B infections occur annually worldwide.
- World Health Organization recorded that hepatitis B caused nearly 820,000 deaths globally in 2024, largely from cirrhosis and liver cancer.
- Centers for Disease Control and Prevention reported in 2025 that about 862,000 people in the United States are living with chronic hepatitis B infection.
- Centers for Disease Control and Prevention estimated in 2024 that approximately 20,700 new hepatitis B infections occur annually in the United States.
- European Centre for Disease Prevention and Control stated in 2024 that more than 4.7 million people in the European region are estimated to have chronic hepatitis B.
- United Nations reported in 2024 that over 90% of infants worldwide receive at least one dose of hepatitis B vaccine.
- World Health Organization confirmed in 2025 that infant hepatitis B vaccination coverage reached about 85% globally.
- Ministry of Health Japan reported in 2024 that hepatitis B prevalence in Japan remains below 1% due to strong vaccination and screening programs.
- Government of India Ministry of Health reported in 2025 that approximately 40 million people in India are estimated to be chronically infected with hepatitis B.
- World Bank reported in 2024 that global health expenditure surpassed USD 9.8 trillion, strengthening infectious disease diagnostic infrastructure worldwide.
- National Institutes of Health reported in 2024 that hepatitis B virus infection remains one of the leading causes of liver cancer globally.
Market Dynamic
Driving Factors in the HBsAg Testing Market
Expansion of Global Hepatitis Screening Programs
Governments and international health organizations have significantly expanded hepatitis screening programs to reduce the burden of liver diseases. Increasing public health campaigns focusing on early detection of viral infections are encouraging routine testing among high-risk populations, including pregnant women, blood donors, and healthcare workers. Healthcare systems worldwide are integrating hepatitis testing into primary care services, which has substantially increased the demand for diagnostic assays. Additionally, elimination targets established by global health organizations are pushing countries to strengthen surveillance and screening infrastructure. These initiatives are driving consistent demand for reliable diagnostic tests and encouraging healthcare providers to adopt advanced HBsAg testing technologies across hospitals and diagnostic laboratories.
Advancements in Diagnostic Technologies
Technological progress in immunoassays, molecular diagnostics, and automated laboratory platforms is playing a major role in improving HBsAg testing efficiency. Modern diagnostic systems offer higher sensitivity, faster turnaround time, and improved detection accuracy compared to traditional testing methods. Automation in laboratory analyzers has enabled large-scale screening, particularly in blood banks and centralized diagnostic facilities. Rapid diagnostic tests have also gained popularity due to their portability and convenience in remote healthcare settings. Continuous research and development by diagnostic manufacturers is leading to innovative testing platforms that integrate multiple biomarkers, enhancing disease detection capabilities and expanding the overall scope of hepatitis screening worldwide.
Restraints in the HBsAg Testing Market
Limited Diagnostic Infrastructure in Developing Regions
Despite significant progress in diagnostic technologies, many low- and middle-income countries continue to face challenges related to limited laboratory infrastructure and healthcare resources. In rural and underserved areas, access to advanced diagnostic equipment and trained laboratory professionals remains restricted. This limitation affects the timely detection of hepatitis B infections and reduces screening coverage. High-precision testing technologies such as automated immunoassay analyzers and molecular diagnostic systems often require substantial investment, making them less accessible for smaller healthcare facilities. These infrastructure gaps can slow the adoption of advanced HBsAg testing solutions and hinder effective disease monitoring in several parts of the world.
Cost Constraints and Testing Accessibility
The cost associated with advanced diagnostic platforms and laboratory automation can be a significant barrier for many healthcare systems. Although rapid diagnostic tests provide cost-effective alternatives, high-sensitivity immunoassay and molecular testing methods may require expensive equipment and reagents. Smaller diagnostic laboratories may struggle to maintain continuous supply chains and equipment maintenance, which can impact testing reliability. Additionally, limited reimbursement policies in certain healthcare systems may discourage frequent screening. These financial challenges can restrict widespread adoption of advanced testing technologies and limit accessibility in price-sensitive healthcare markets.
Opportunities in the HBsAg Testing Market
Expansion of Point-of-Care Diagnostics
The growing demand for decentralized healthcare services has created strong opportunities for point-of-care testing solutions. Portable diagnostic kits enable rapid hepatitis B screening in community clinics, rural healthcare facilities, and emergency settings. These tests require minimal laboratory infrastructure and provide results within minutes, making them highly suitable for large-scale screening campaigns. Governments and global health organizations are increasingly adopting point-of-care diagnostic programs to improve early detection rates in underserved regions. Continuous improvements in test accuracy and reliability are expected to further expand the adoption of rapid HBsAg testing solutions across emerging healthcare markets.
Increasing Focus on Hepatitis Elimination Programs
Many countries have committed to long-term strategies aimed at eliminating viral hepatitis as a major public health threat. These initiatives involve expanding vaccination coverage, improving disease surveillance, and increasing diagnostic screening. Healthcare providers are integrating hepatitis testing into routine medical checkups and maternal healthcare programs. Large-scale testing campaigns supported by international organizations are expected to boost demand for reliable diagnostic tools. As global awareness about hepatitis prevention continues to rise, diagnostic manufacturers are likely to experience significant opportunities for expanding their product portfolios and improving accessibility to testing technologies.
Trends in the HBsAg Testing Market
Integration of Automated Laboratory Systems
Automation is becoming a major trend in modern diagnostic laboratories. High-throughput immunoassay analyzers allow laboratories to process large numbers of samples efficiently with minimal manual intervention. Automated systems also reduce human error and improve result consistency. Healthcare providers are increasingly investing in integrated diagnostic platforms capable of performing multiple infectious disease tests simultaneously. This trend is particularly significant in centralized laboratories and blood banks where large screening volumes require reliable and efficient testing infrastructure.
Growth of Rapid Diagnostic Technologies
Rapid diagnostic tests are gaining widespread attention due to their ability to deliver quick results in decentralized healthcare environments. These tests are especially useful in remote regions where laboratory facilities are limited. Continuous improvements in sensitivity and specificity have enhanced the reliability of rapid HBsAg testing kits. Mobile healthcare units and community health programs frequently rely on these portable testing solutions to conduct large-scale screenings. As healthcare systems move toward faster and more accessible diagnostic services, rapid testing technologies are expected to play an increasingly important role in hepatitis detection strategies.
Impact of Artificial Intelligence in HBsAg Testing Market
- AI-Based Diagnostic Interpretation: AI algorithms assist laboratories in analyzing immunoassay results with greater accuracy, reducing human interpretation errors and improving diagnostic reliability.
- Predictive Disease Modeling: AI tools analyze patient data to predict hepatitis infection risks and guide targeted screening programs for high-risk populations.
- Automated Laboratory Workflow: AI-driven laboratory systems optimize sample processing, result validation, and reporting, improving operational efficiency in diagnostic centers.
- Quality Control Monitoring: AI solutions monitor testing instruments and reagent performance, helping laboratories detect anomalies and maintain testing accuracy.
- Data-Driven Epidemiological Insights: AI analyzes large public health datasets to identify infection patterns and guide government hepatitis control strategies.
- Enhanced Test Development: Diagnostic manufacturers use AI models to design improved assay reagents and enhance antigen detection sensitivity.
- Clinical Decision Support: AI-powered healthcare platforms assist physicians in interpreting test results alongside patient history and laboratory parameters.
- Remote Diagnostic Support: AI-enabled telemedicine platforms support remote healthcare workers in interpreting rapid diagnostic test results.
- Supply Chain Optimization: AI systems forecast diagnostic reagent demand, ensuring uninterrupted supply for large screening programs.
Research Scope and Analysis
By Test Type Analysis
Quantitative tests dominate the HBsAg testing market due to their ability to measure the concentration of hepatitis B surface antigen in the bloodstream. These tests are widely used in hospitals, diagnostic laboratories, and research institutions for monitoring disease progression and evaluating treatment responses. Quantitative analysis provides valuable clinical insights that help physicians determine viral activity levels and guide therapeutic strategies. The demand for these tests is particularly strong in specialized diagnostic laboratories equipped with automated immunoassay analyzers capable of processing large volumes of samples. Improved assay sensitivity and advanced laboratory automation have further strengthened their clinical adoption. Quantitative testing is expected to maintain its leadership in the market and is projected to account for approximately 58.4% of the market share in 2026, supported by increasing clinical demand for precise viral infection monitoring and improved laboratory diagnostic capabilities.
Qualitative tests represent one of the fastest-growing segments in the HBsAg testing market due to their ability to provide quick detection of hepatitis B infection status. These tests are primarily used for initial screening and point-of-care diagnostics where rapid results are essential. Hospitals, blood banks, and community health programs frequently utilize qualitative testing methods to identify infected individuals before conducting confirmatory quantitative analysis. Rapid diagnostic kits and lateral flow assays are commonly used in this segment due to their simplicity and affordability. Growing demand for decentralized healthcare and mass screening campaigns is expected to accelerate adoption of qualitative testing technologies in the coming years.
By Technology Analysis
ELISA remains the most widely used technology in HBsAg testing due to its high sensitivity, reliability, and cost-effectiveness. The technology is extensively used in diagnostic laboratories and blood screening facilities for detecting hepatitis B surface antigen in serum or plasma samples. ELISA testing platforms can process multiple samples simultaneously, making them ideal for large-scale screening programs. Continuous improvements in assay reagents and automation capabilities have significantly enhanced testing efficiency and accuracy. Healthcare systems rely heavily on ELISA-based testing for routine hepatitis screening. This technology is expected to maintain its leadership in the market and is projected to account for approximately 36.7% of the market share in 2026, supported by its widespread clinical adoption and strong laboratory infrastructure across developed healthcare systems.
Molecular testing technologies such as PCR and nucleic acid testing are emerging as one of the fastest-growing segments within the HBsAg testing market. These methods provide highly sensitive detection of viral genetic material, allowing for early identification of infections even before antigen levels become detectable. Molecular diagnostics are increasingly used in advanced healthcare facilities and research institutions where precise viral detection and monitoring are critical. Rising investments in molecular diagnostic infrastructure and improvements in automated PCR platforms are expected to support rapid expansion of this technology segment.
By Product Type Analysis
Test kits and reagents represent the largest segment in the HBsAg testing market due to their recurring usage in diagnostic procedures. Laboratories and healthcare facilities require a continuous supply of reagents, assay kits, and testing consumables to conduct routine screenings and diagnostic tests. ELISA kits, rapid test kits, and CLIA reagents are widely utilized across hospitals, blood banks, and research laboratories. Increasing screening volumes and expanding diagnostic programs are driving steady demand for consumables. This segment is projected to account for around 64.2% of the market share in 2026, supported by consistent testing requirements and the growing need for reliable diagnostic reagents.
Diagnostic instruments and automated analyzers represent a rapidly growing segment as laboratories increasingly invest in high-throughput diagnostic platforms. These instruments enable efficient processing of large sample volumes while ensuring consistent accuracy. Modern analyzers are integrated with laboratory information systems, enabling automated result reporting and improved workflow management. Growing adoption of advanced laboratory automation and increasing demand for faster diagnostic turnaround times are expected to drive expansion in this segment.
By Sample Type Analysis
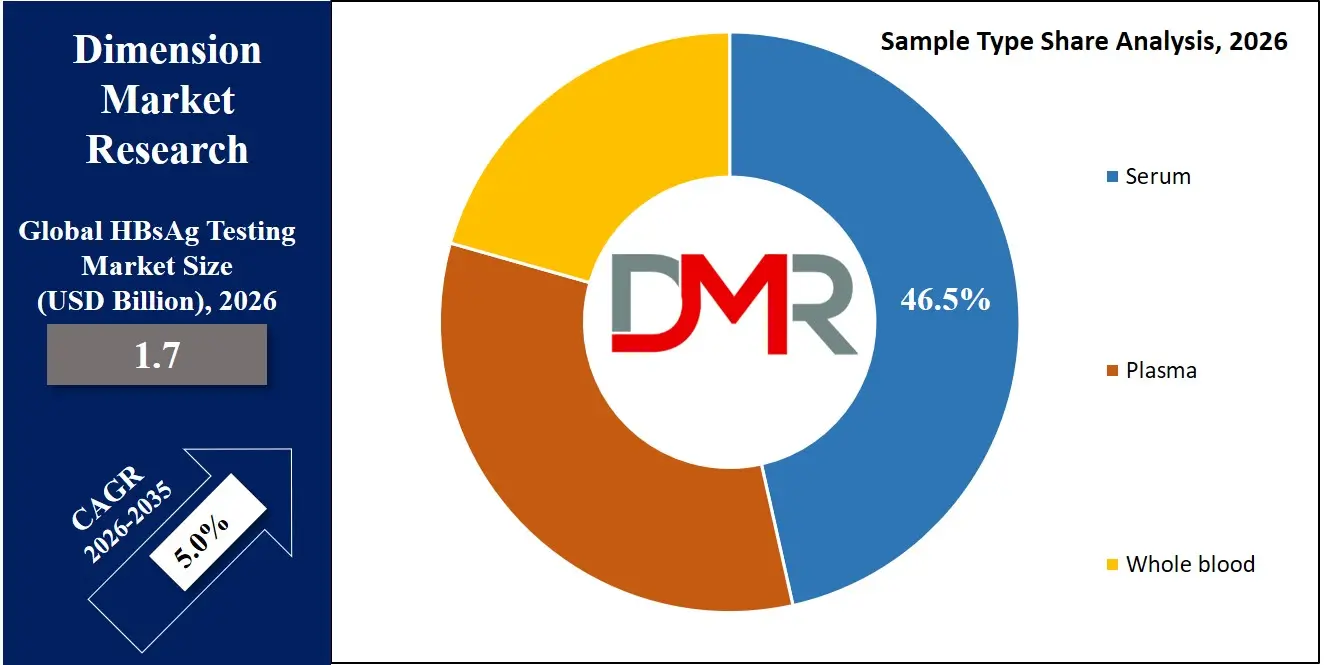
Serum remains the most commonly used sample type for HBsAg testing due to its high reliability and compatibility with various diagnostic technologies. Healthcare providers prefer serum samples for immunoassay-based testing methods such as ELISA and CLIA because they provide clear antigen detection results. Serum samples are widely used in hospitals, blood banks, and diagnostic laboratories where standardized sample collection protocols are followed. The widespread availability of serum testing procedures and their compatibility with automated analyzers contribute to strong demand in this segment. Serum samples are expected to account for approximately 46.5% of the market share in 2026, supported by their widespread clinical usage and consistent diagnostic accuracy.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Whole blood testing is gaining traction as one of the fastest-growing segments due to the increasing use of rapid diagnostic kits. These tests allow healthcare providers to perform screening without complex laboratory processing. Whole blood samples are particularly useful in point-of-care testing environments such as rural clinics, community healthcare centers, and mobile testing units. The growing demand for portable diagnostic solutions and simplified testing procedures is expected to accelerate adoption of whole blood-based HBsAg testing methods.
By Application Analysis
Blood donor screening remains the dominant application in the HBsAg testing market due to strict regulations requiring testing of donated blood for infectious diseases. Blood banks and transfusion centers rely heavily on HBsAg testing to ensure the safety of the blood supply. High-throughput immunoassay systems and automated analyzers enable efficient screening of large donor populations. Governments and healthcare organizations enforce mandatory testing standards, making this application essential for public health protection. Blood donor screening is expected to account for approximately 34.8% of the market share in 2026, supported by expanding blood donation programs and stringent safety regulations.
Prenatal screening represents one of the fastest-growing application segments as healthcare providers prioritize early detection of hepatitis B in pregnant women. Identifying infected mothers allows medical professionals to implement preventive interventions that significantly reduce the risk of mother-to-child transmission. Increasing awareness about maternal health and expanding prenatal care programs worldwide are expected to drive growth in this segment.
By End User Analysis
Diagnostic laboratories dominate the HBsAg testing market due to their specialized capabilities and access to advanced testing technologies. These laboratories process large volumes of patient samples from hospitals, clinics, and blood banks. They often utilize automated immunoassay systems and molecular diagnostic platforms that provide accurate and rapid results. Increasing demand for centralized diagnostic services and improved laboratory infrastructure contribute to strong growth in this segment. Diagnostic laboratories are projected to account for approximately 39.6% of the market share in 2026, supported by rising testing volumes and technological advancements in laboratory automation.
Point-of-care settings are emerging as one of the fastest-growing segments in the HBsAg testing market. Rapid diagnostic kits allow healthcare providers to conduct screening directly at patient locations without requiring laboratory infrastructure. These solutions are widely used in rural healthcare facilities, emergency settings, and community screening programs. The growing emphasis on decentralized healthcare and early disease detection is expected to accelerate the expansion of this segment.
The HBsAg Testing Market Report is segmented on the basis of the following:
By Test Type
- Qualitative Tests
- Quantitative Tests
By Technology
- Enzyme-Linked Immunosorbent Assay (ELISA)
- Chemiluminescent Immunoassay (CLIA)
- Rapid Diagnostic Tests (RDT) / Lateral Flow Assays
- Molecular Tests
- Polymerase Chain Reaction (PCR)
- Nucleic Acid Testing (NAT)
By Product Type
- Test Kits & Reagents
- ELISA Kits
- Rapid Test Kits
- CLIA Reagents
- Instruments / Analyzers
By Sample Type
By Application
- Blood Donor Screening
- Diagnostic Testing
- Prenatal Screening
- Research & Epidemiological Studies
By End User
- Hospitals & Clinics
- Diagnostic Laboratories
- Blood Banks
- Research Institutes
- Point-of-Care Settings
Regional Analysis
Leading Region in the HBsAg Testing Market
North America is expected to account for approximately 38.9% share in the HBsAg testing market in 2026, driven by high diagnostic awareness, strong public health policies, and the presence of sophisticated laboratory infrastructure. Further, the market is driven due to its highly advanced healthcare infrastructure, strong diagnostic capabilities, and extensive infectious disease screening programs. Healthcare systems in the region emphasize early detection and prevention of viral infections through routine diagnostic testing and blood safety regulations. Large diagnostic laboratories, hospitals, and research institutions rely heavily on automated immunoassay analyzers and molecular diagnostic platforms. Government initiatives aimed at reducing hepatitis-related complications and improving disease surveillance further strengthen testing adoption. Continuous investments in healthcare technology and laboratory modernization support market growth.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Fastest Growing Region in the HBsAg Testing Market
Asia Pacific is projected to be the fastest-growing region in the HBsAg testing market due to its large population base and increasing prevalence of hepatitis B infection. Governments across the region are investing heavily in public health screening programs and expanding vaccination coverage. Countries such as China, India, and Southeast Asian nations are strengthening their diagnostic infrastructure and improving access to healthcare services. Rising healthcare awareness, increasing medical tourism, and expanding laboratory networks are contributing to higher testing volumes. The growing adoption of rapid diagnostic kits and point-of-care testing technologies is also accelerating market growth. Continued investments in healthcare modernization and disease prevention initiatives are expected to sustain rapid expansion across the region.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The HBsAg testing market is characterized by strong competition driven by technological innovation, product differentiation, and expanding diagnostic capabilities. Market participants continuously invest in research and development to enhance assay sensitivity, reduce testing time, and improve automation in diagnostic platforms. Strategic collaborations with hospitals, diagnostic laboratories, and public health organizations are commonly adopted to expand testing adoption. Companies also focus on developing cost-effective rapid diagnostic kits to address demand in emerging healthcare markets. Regulatory compliance and quality assurance play a significant role in maintaining market credibility. Continuous product upgrades, global distribution networks, and investments in laboratory automation technologies remain key strategies used to strengthen market positioning and expand customer reach.
Some of the prominent players in the global HBsAg Testing are:
- Abbott Laboratories
- F. Hoffmann-La Roche
- Siemens Healthineers
- Danaher Corporation
- Bio-Rad Laboratories
- Thermo Fisher Scientific
- Becton, Dickinson and Company
- Ortho Clinical Diagnostics
- Fujirebio
- DiaSorin
- bioMérieux
- Sysmex Corporation
- QIAGEN
- Mindray Medical International
- Hologic
- PerkinElmer
- Grifols
- Trinity Biotech
- SD Biosensor
- Wantai BioPharm
- Other Key Players
Recent Developments
- In April 2025, Roche announced an expansion of its molecular diagnostic portfolio with new testing capabilities targeting hepatitis B virus detection and monitoring. The company introduced an upgraded molecular testing solution designed to work with its automated PCR-based diagnostic platforms. The system enables laboratories to perform highly sensitive detection of hepatitis B viral markers while processing large numbers of samples simultaneously. The development supports growing demand for advanced molecular diagnostics in infectious disease testing.
- In January 2025, Abbott announced the launch of an advanced hepatitis B diagnostic assay designed to enhance detection accuracy for hepatitis B surface antigen. The new assay was developed to improve sensitivity and enable earlier detection of infection in clinical settings. It is compatible with the company's automated immunoassay analyzers, allowing high-throughput testing for hospitals and diagnostic laboratories. The launch aims to support large-scale screening programs, particularly in blood banks and infectious disease monitoring initiatives.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 1.7 Bn |
| Forecast Value (2035) |
USD 2.6 Bn |
| CAGR (2026–2035) |
5.0% |
| The US Market Size (2026) |
USD 0.6 Bn |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Test Type (Qualitative Tests, Quantitative Tests), By Technology (Enzyme-Linked Immunosorbent Assay (ELISA), Chemiluminescent Immunoassay (CLIA), Rapid Diagnostic Tests (RDT) / Lateral Flow Assays, Molecular Tests), By Product Type (Test Kits & Reagents, Instruments / Analyzers), By Sample Type (Serum, Plasma, Whole Blood), By Application (Blood Donor Screening, Diagnostic Testing, Prenatal Screening, Research & Epidemiological Studies), By End User (Hospitals & Clinics, Diagnostic Laboratories, Blood Banks, Research Institutes, Point-of-Care Settings) |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
| Prominent Players |
Abbott Laboratories, F. Hoffmann-La Roche, Siemens Healthineers, Danaher Corporation, Bio-Rad Laboratories, Thermo Fisher Scientific, Becton, Dickinson and Company, Ortho Clinical Diagnostics, Fujirebio, DiaSorin, bioMérieux, Sysmex Corporation, QIAGEN, Mindray Medical International, Hologic, PerkinElmer, Grifols, Trinity Biotech, SD Biosensor, Wantai BioPharm, and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Global HBsAg Testing Market?
▾ The Global HBsAg Testing Market size is expected to reach USD 1.7 billion by 2026 and is projected to reach USD 2.6 billion by the end of 2035.
Which region accounted for the largest Global HBsAg Testing Market?
▾ North America is expected to have the largest market share in the Global HBsAg Testing Market, with a share of about 38.9% in 2026.
How big is the HBsAg Testing Market in the US?
▾ The US HBsAg Testing market is expected to reach USD 0.6 billion by 2026.
Who are the key players in the HBsAg Testing Market?
▾ Some of the major key players in the Global HBsAg Testing Market include Abbott, Roche, BD and others.
What is the growth rate in the Global HBsAg Testing Market?
▾ The market is growing at a CAGR of 5.0 percent over the forecasted period.