Market Overview
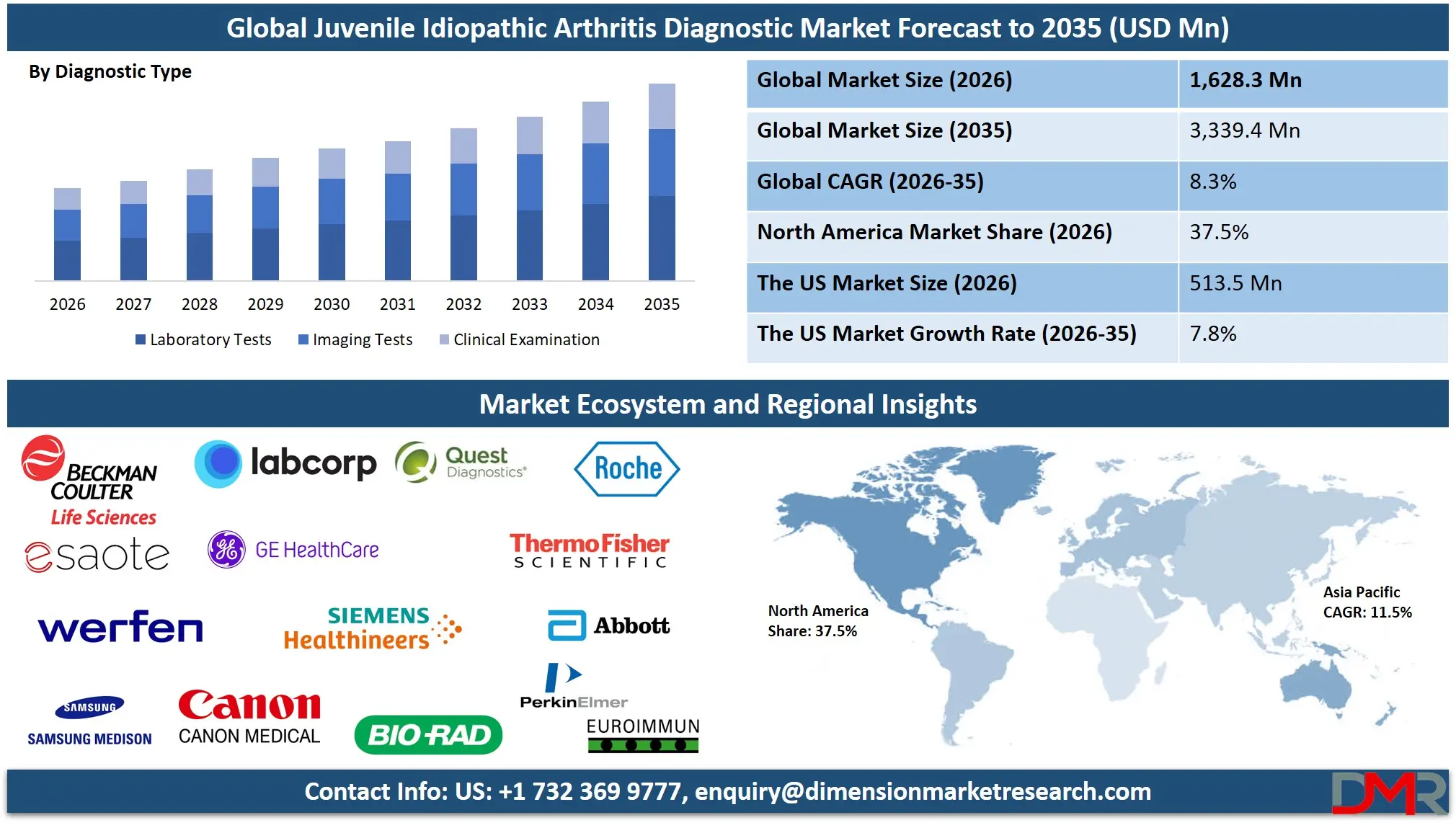
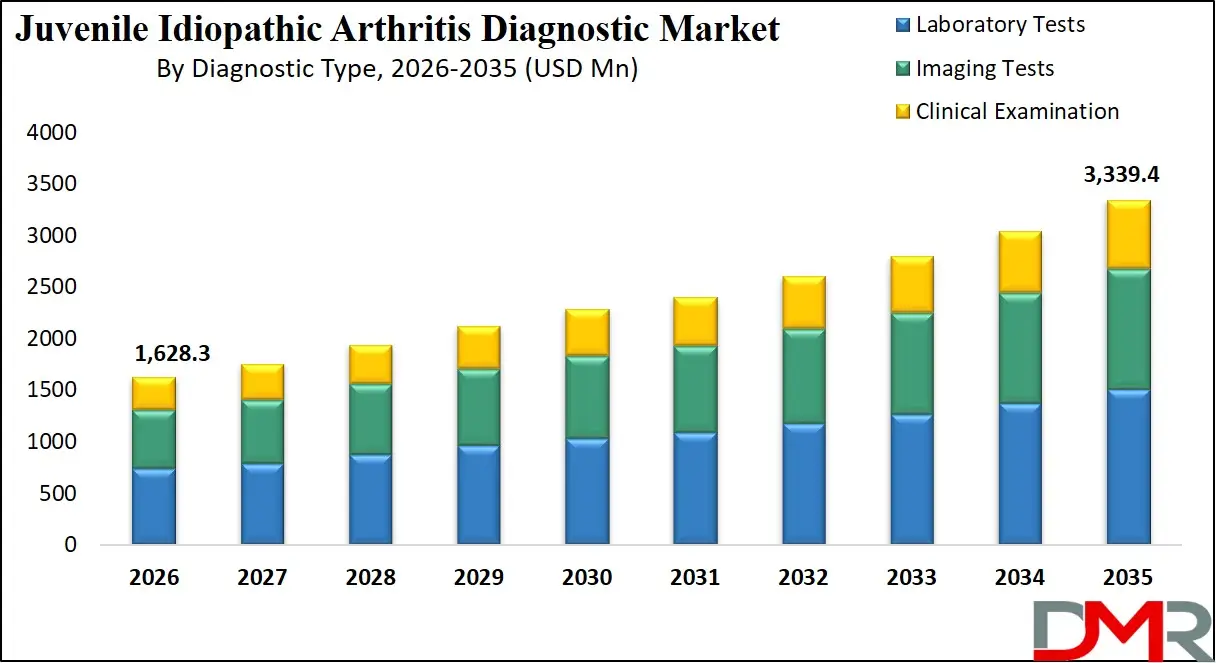
The Global Juvenile Idiopathic Arthritis Diagnostic Market is set for substantial expansion projected to reach a market value of USD 1,628.3 million in 2026 and projected to grow at a strong CAGR of 8.3% from 2026 to 2035, to a market value of USD 3,339.4 million by 2035. This robust growth trajectory is fueled by the increasing global prevalence of JIA, rising awareness among pediatricians, and significant advancements in diagnostic technologies, including biomarker identification and medical imaging.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Rising incidences of autoimmune disorders in children, coupled with a growing emphasis on early and accurate diagnosis to prevent long-term joint damage and disability, are compelling healthcare providers to adopt advanced diagnostic systems. The expansion of specialized pediatric rheumatology centers, improved healthcare infrastructure in emerging economies, and greater access to non-invasive testing methods are significantly increasing demand for reliable JIA diagnostics.
Additionally, growing healthcare expenditure and supportive government initiatives for childhood disease management are further accelerating market adoption. Hospitals and diagnostic laboratories are increasingly integrating automated laboratory systems, high-resolution musculoskeletal ultrasound, and AI-driven imaging analysis to enhance diagnostic accuracy and patient outcomes.
Technological advancements in multiplex biomarker assays and the development of point-of-care testing are enabling faster and more precise diagnosis, reducing the time to treatment. As awareness of JIA increases among parents and primary care physicians, and as treatment guidelines emphasize the importance of early intervention, the Juvenile Idiopathic Arthritis Diagnostic Market is expected to witness sustained growth through 2035, driven by the global prioritization of child health.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Growing collaborations between research institutions and diagnostic companies to develop novel biomarkers and genetic tests further accelerate global adoption. However, barriers such as the high cost of advanced diagnostic systems, a shortage of skilled pediatric rheumatologists, and variability in healthcare access across different regions remain. Despite these limitations, the convergence of immunological research, imaging technology, and AI-powered diagnostics positions JIA diagnostics as a crucial component of pediatric rheumatology through 2035.
The US Juvenile Idiopathic Arthritis Diagnostic Market
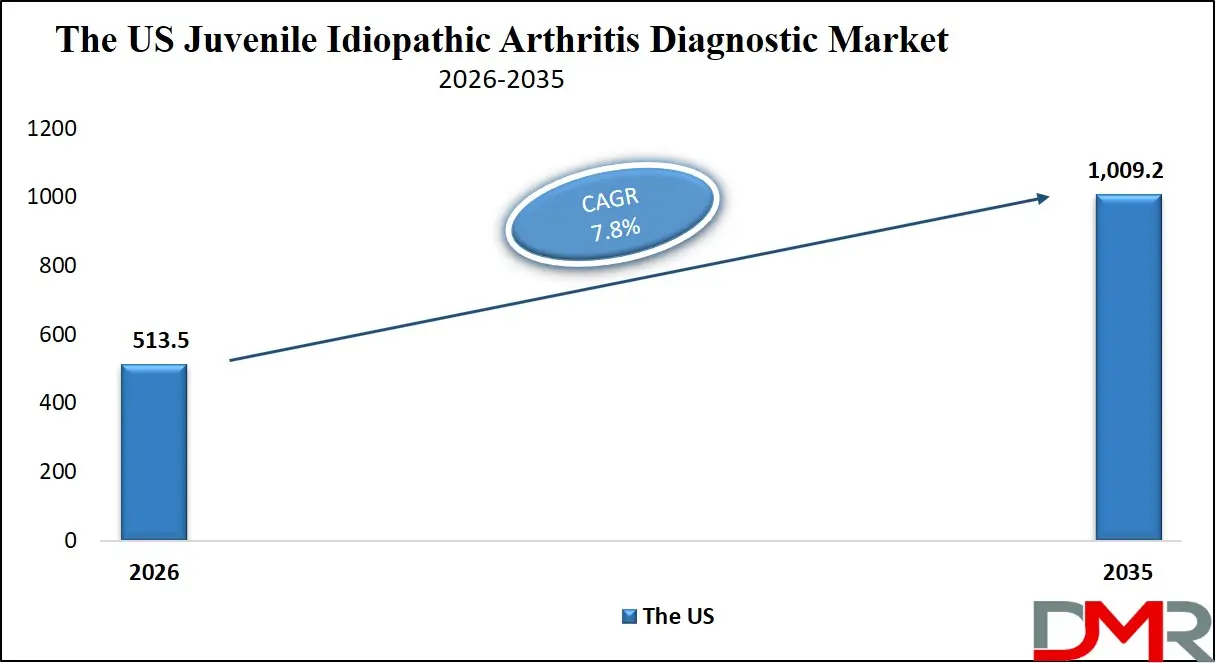
The U.S. Juvenile Idiopathic Arthritis Diagnostic Market is projected to reach USD 513.5 million in 2026 and grow at a CAGR of 7.8%, reaching USD 1,009.2 million by 2035. The U.S. leads global adoption due to its advanced healthcare system, high awareness of pediatric rheumatic diseases, and strong support from organizations like the American College of Rheumatology and the Arthritis Foundation.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The presence of world-renowned pediatric research hospitals and a robust reimbursement framework for diagnostic procedures fuels demand for advanced testing. Major healthcare providers and diagnostic chains such as Quest Diagnostics and LabCorp are integrating comprehensive JIA diagnostic panels to aid in early disease classification and management.
U.S. regulatory support through the FDA's guidance on pediatric medical devices and diagnostics encourages innovation in the field. The market is witnessing a shift toward multiplex autoantibody testing and advanced imaging techniques like power Doppler ultrasound, enabling more precise assessment of disease activity. The focus on personalized medicine in pediatric rheumatology has further intensified the need for sophisticated diagnostic tools, positioning the U.S. as a critical innovator in this space.
The Europe Juvenile Idiopathic Arthritis Diagnostic Market
The Europe Juvenile Idiopathic Arthritis Diagnostic Market is projected to reach USD 465.8 million in 2026 and is expected to grow at a CAGR of 7.6% from 2026 to 2035, reaching approximately USD 900.4 million by 2035. Europe's strong position is anchored by its universal healthcare coverage, extensive research networks, and collaborative pediatric rheumatology initiatives like the Paediatric Rheumatology European Society (PReS).
Countries such as Germany, the U.K., France, and Italy are widely adopting advanced diagnostic services, driven by a high emphasis on early disease management and the establishment of specialized pediatric rheumatology centers. The U.K.'s National Health Service (NHS) long-term plan and EU-funded research projects further necessitate standardized diagnostic pathways for JIA across member states.
Europe's well-established healthcare systems, high awareness among clinicians, and a strong network of patient advocacy groups drive the demand for accurate and timely diagnostic solutions. Funding for pediatric research and cross-border collaborations encourages the standardization of diagnostic criteria and the sharing of best practices across the continent. With a sophisticated medical community and a regulatory landscape that prioritizes patient care, Europe remains a highly advanced and essential region for JIA diagnostic services.
The Japan Juvenile Idiopathic Arthritis Diagnostic Market
The Japan Juvenile Idiopathic Arthritis Diagnostic Market is projected to reach USD 128.7 million in 2026 and is expected to grow at a CAGR of 7.1% from 2026 to 2035, reaching approximately USD 238.6 million by 2035. Japan's advanced universal healthcare system and a government focus on improving pediatric specialty care are driving the adoption of accurate diagnostic tools for rare childhood diseases.
The Ministry of Health, Labour and Welfare (MHLW) actively supports the development of guidelines for childhood rheumatic diseases, promoting the use of standardized diagnostic tests in university hospitals and children's medical centers. Japan's leadership in medical imaging technology and laboratory automation provides a robust foundation for advanced diagnostic methods.
Japan's "Healthy Parents and Children 21" national movement, supported by major medical institutions, integrates early diagnosis of chronic conditions like JIA into the broader child health strategy. Hospitals are deploying advanced diagnostic panels and imaging to differentiate JIA from other causes of pediatric musculoskeletal pain, ensuring appropriate and timely treatment. Japan's cultural emphasis on precision medicine and long-term patient outcomes positions it as a high-growth, quality-focused market for diagnostic solutions.
Global Juvenile Idiopathic Arthritis Diagnostic Market: Key Takeaways
- Steady Global Market Growth Outlook: The Global Juvenile Idiopathic Arthritis Diagnostic Market is expected to be valued at USD 1,628.3 million in 2026 and is projected to reach USD 3,339.4 million by 2035, showcasing steady expansion supported by rising disease prevalence and early diagnosis initiatives.
- Moderate CAGR Driven by Diagnostic Advancements: The market is expected to grow at a CAGR of 8.3% from 2026 to 2035, fueled by increasing awareness, technological improvements in biomarker and imaging tests, and a growing focus on pediatric rheumatology.
- Steady Growth Trajectory in the United States: The U.S. Juvenile Idiopathic Arthritis Diagnostic Market stands at USD 513.5 million in 2026 and is projected to reach USD 1,009.2 million by 2035, expanding at a CAGR of 7.8% due to advanced healthcare infrastructure and high disease awareness.
- Regional Dominance: North America is expected to capture approximately 37.5% of the global market share in 2026, supported by well-established healthcare systems, high diagnostic spending, and strong patient advocacy networks.
- Rapid Advancement in Diagnostic Technologies: Innovations including multiplex biomarker assays, next-generation sequencing for genetic predisposition, AI-powered analysis of musculoskeletal ultrasound, and novel autoantibody tests are significantly enhancing the accuracy and speed of JIA diagnosis.
- Growing Disease Prevalence Boosts Adoption: The increasing global recognition of JIA as a leading cause of childhood disability, coupled with the need for early intervention to improve long-term outcomes, is driving sustained demand for comprehensive and reliable diagnostic solutions.
Global Juvenile Idiopathic Arthritis Diagnostic Market: Use Cases
- Initial Screening in Primary Care: Pediatricians use a combination of physical exams and initial lab tests (e.g., inflammatory markers) to identify children with persistent joint pain and swelling, referring suspected cases to specialists.
- Differential Diagnosis in Rheumatology: Specialists utilize autoantibody panels (e.g., ANA, RF) and advanced imaging (MRI, ultrasound) to distinguish JIA from other conditions like lupus, Lyme disease, or bone cancer.
- Disease Subtype Classification: Specific diagnostic tests help classify a child into a JIA subtype (e.g., oligoarticular, polyarticular, systemic), which is crucial for determining the most effective treatment pathway.
- Monitoring Disease Activity: Imaging techniques like power Doppler ultrasound are used to monitor synovitis and disease progression over time, helping rheumatologists adjust medication regimens.
- Predicting Treatment Response: Genetic and biomarker tests are increasingly used to predict how a patient might respond to specific therapies, such as biologics, enabling a more personalized approach to treatment.
Global Juvenile Idiopathic Arthritis Diagnostic Market: Stats & Facts
National Center for Biotechnology Information (NCBI) – U.S. National Institutes of Health
- The global incidence of Juvenile Idiopathic Arthritis ranges from 1.6 to 23 cases per 100,000 children annually.
- The global prevalence ranges from 3.8 to 400 cases per 100,000 children depending on geographic region and study design.
- In North America, the incidence rate is approximately 0.041–0.061 cases per 1,000 children annually.
- A population-based study in Utah estimated the prevalence of JIA at about 1.2 cases per 1,000 children.
- Oligoarthritis accounts for around 50–60% of all JIA cases, making it the most common subtype.
- RF-negative polyarthritis represents approximately 11–28% of cases.
- RF-positive polyarthritis accounts for about 2–7% of cases.
- Systemic juvenile idiopathic arthritis accounts for roughly 10–20% of all cases.
- Psoriatic arthritis subtype accounts for approximately 2–15% of cases.
- Enthesitis-related arthritis represents about 1–7% of cases.
- The risk of JIA in siblings of affected patients is 15–30 times higher than the general population.
Pediatric Rheumatology Journal
- Approximately 3 million children and adolescents worldwide are estimated to live with JIA.
- The global prevalence ranges between 3.8 and 400 per 100,000 children.
- The global incidence ranges from 1.6 to 23 per 100,000 children annually.
- Girls are more frequently affected than boys across most JIA subtypes.
Kaiser Permanente Division of Research (United States)
- The age- and sex-standardized incidence rate of JIA is about 11.9 cases per 100,000 children per year.
- Incidence among girls is approximately 16.4 per 100,000 children annually.
- Incidence among boys is around 7.7 per 100,000 children annually.
- The point prevalence of JIA is estimated at 44.7 cases per 100,000 children.
- Approximately 45% of identified JIA cases were newly diagnosed during the study period.
Arthritis Research & Therapy (Sweden Population Study)
- The annual incidence of JIA in southern Sweden is about 12.8 cases per 100,000 children under 16 years of age.
- Approximately 10.8% of children with JIA develop uveitis, a major complication requiring ophthalmologic monitoring.
- Around 40% of follow-up years among patients are free from arthritis or uveitis symptoms.
- About 4.4% of patients require orthopedic corrective surgery during long-term follow-up.
Oxford University Rheumatology Epidemiology Study (England)
- The overall incidence of JIA in England is estimated at 5.4 cases per 100,000 children per year.
- Incidence among White children is about 6.2 per 100,000 population.
- Incidence among Asian children is approximately 2.7 per 100,000 population.
- Incidence among Black children is around 2.9 per 100,000 population.
- Incidence among mixed-ethnicity children is about 3.0 per 100,000 population.
BMC Musculoskeletal Disorders (Clinical Research)
- JIA is most frequently diagnosed in children aged 1–3 years.
- Growth delay occurs in approximately 35.75% of children with JIA due to chronic inflammation and treatment effects.
Society of Pediatric Psychology
- The global prevalence of JIA is estimated between 4 and 150 cases per 100,000 children.
- Approximately 300,000 children in the United States are living with juvenile arthritis.
Global Juvenile Idiopathic Arthritis Diagnostic Market: Market Dynamic
Driving Factors in the Global JIA Diagnostic Market
Increasing Global Prevalence and Awareness
The rising global incidence and diagnosis rates of Juvenile Idiopathic Arthritis are a primary driver. As awareness of JIA grows among healthcare professionals and the general public, more children with symptoms are being referred to specialists. Parent advocacy groups and awareness campaigns are playing a crucial role in reducing the time to diagnosis. This increased focus on identifying JIA early creates sustained demand for reliable diagnostic tools, from initial screening tests to advanced imaging for confirmation.
Technological Advancements in Diagnostics
Significant progress in diagnostic technologies is fueling market growth. The development of highly specific and sensitive autoantibody tests, combined with advanced imaging techniques like MRI without contrast and high-resolution ultrasound, allows for more accurate and earlier diagnosis. Furthermore, research into genomic and proteomic biomarkers is paving the way for next-generation diagnostic panels that can not only diagnose JIA but also predict disease course and treatment response.
Restraints in the Global JIA Diagnostic Market
High Cost and Complexity of Advanced Diagnostics
The sophisticated nature of tests like MRI and multiplex biomarker assays makes them expensive, limiting accessibility, particularly in low- and middle-income countries. Furthermore, the interpretation of these tests requires specialized expertise in pediatric rheumatology and radiology, which is in short supply globally. This complexity can lead to diagnostic delays and inconsistencies, hindering market growth in regions lacking specialized centers.
Lack of Standardized Diagnostic Protocols
Despite international efforts, there is a lack of universally standardized diagnostic protocols and criteria for JIA. Variations in clinical practice between countries and even between centers can lead to different diagnostic approaches. This fragmentation makes it challenging to compare data across studies and can create uncertainty in the diagnostic process, impacting the adoption of new technologies.
Opportunities in the Global JIA Diagnostic Market
Development of Point-of-Care (POC) Tests
There is a significant opportunity to develop rapid, easy-to-use POC tests for inflammatory markers or specific antibodies that could be used in pediatricians' offices. Such tests would allow for faster triage of children with suspected JIA, accelerating the referral process to specialists. A simple finger-prick test that could indicate inflammation or autoimmunity would be a game-changer in primary care settings globally.
AI and Machine Learning in Diagnostic Imaging
The integration of artificial intelligence into the analysis of musculoskeletal ultrasound and MRI images presents a major opportunity. AI algorithms can be trained to detect early signs of synovitis, bone erosion, or tenosynovitis with high accuracy, potentially aiding less experienced radiologists and reducing the time required for image interpretation. This technology can standardize image analysis and improve diagnostic confidence.
Trends in the Global JIA Diagnostic Market
Shift Towards Non-Invasive or Minimally Invasive Testing
There is a strong trend towards reducing the reliance on invasive procedures like joint aspiration for diagnosis. This is driving the adoption of high-resolution imaging techniques and sophisticated blood-based biomarker tests that can provide comprehensive diagnostic information without the need for more invasive sampling.
Integration of Multi-Omics Data
The future of JIA diagnosis lies in integrating data from multiple sources – clinical examination, genomics (genetic risk factors), proteomics (autoantibodies and cytokines), and imaging. This multi-omics approach aims to provide a holistic view of the patient's disease, enabling precise subtype classification and personalized treatment plans from the point of diagnosis.
Global Juvenile Idiopathic Arthritis Diagnostic Market: Research Scope and Analysis
By Diagnostic Type Analysis
The imaging tests segment is projected to dominate the Global Juvenile Idiopathic Arthritis (JIA) Diagnostic Market, accounting for a significant share of total revenue. Imaging plays a vital role in detecting structural and inflammatory changes in joints, which are often difficult to identify through clinical examination alone. Techniques such as Magnetic Resonance Imaging (MRI) and musculoskeletal ultrasound allow physicians to visualize synovitis, joint effusion, cartilage damage, and early bone erosions with high precision. Early identification of these abnormalities is essential because prompt diagnosis and treatment significantly reduce the risk of long-term joint damage and disability in children.
MRI is particularly valuable because it provides highly detailed images of both bone and soft tissues, enabling clinicians to detect early inflammatory changes before permanent damage occurs. Meanwhile, ultrasound has gained widespread adoption due to its real-time imaging capability, portability, and ability to assess multiple joints during a single session. Advanced technologies such as power Doppler ultrasound enable rheumatologists to evaluate active blood flow within inflamed synovial tissue, offering direct insight into disease activity and treatment response.
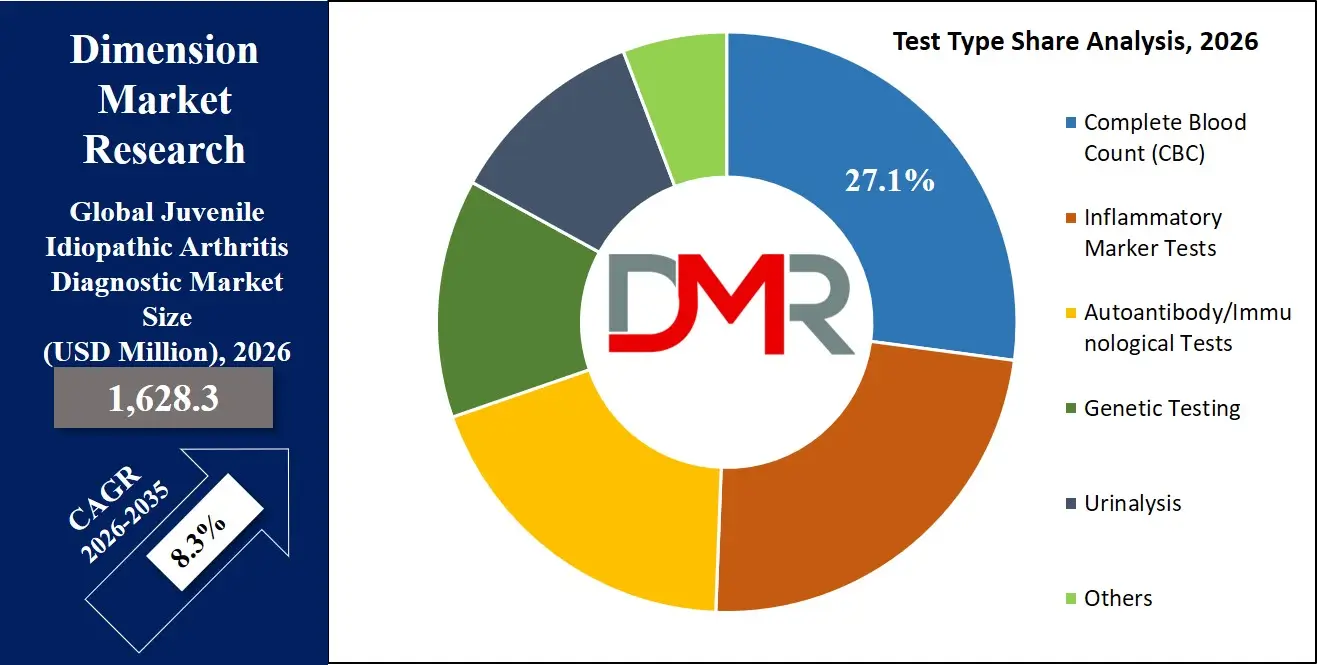
By Test Type Analysis
The autoantibody and immunological tests segment is expected to hold a substantial share within the test type category due to its central role in confirming the autoimmune nature of Juvenile Idiopathic Arthritis. These tests provide crucial biological evidence that supports clinical findings and helps physicians classify the specific subtype of the disease. Common diagnostic tests in this category include Antinuclear Antibody (ANA), Rheumatoid Factor (RF), and anti-cyclic citrullinated peptide (anti-CCP) antibodies. Each of these biomarkers offers valuable insights into immune system activity and disease severity, assisting clinicians in establishing a more precise diagnosis.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
For instance, ANA positivity is frequently associated with certain JIA subtypes and is particularly important for identifying children who may be at increased risk of developing uveitis, a serious eye complication. Rheumatoid Factor, although less common in children compared to adults, is a defining marker for RF-positive polyarticular JIA, which tends to have a more aggressive disease course. Anti-CCP antibodies are also highly specific indicators of autoimmune joint disease and may signal a higher likelihood of joint damage over time.
By Age Group Analysis
The 6–12 years (school-age) segment is projected to dominate the age group analysis of the Global Juvenile Idiopathic Arthritis Diagnostic Market. This dominance is largely attributed to the higher incidence of JIA symptoms during early and middle childhood, when many of the most common disease subtypes begin to manifest. Conditions such as oligoarticular and polyarticular JIA frequently present during this stage, prompting the need for timely and accurate diagnostic evaluations. As children in this age group are actively growing and participating in physical activities, joint pain or swelling may initially be mistaken for minor injuries, sports-related strain, or typical growth-related discomfort.
Because of these overlapping symptoms, healthcare providers often rely on comprehensive diagnostic approaches to differentiate JIA from other musculoskeletal conditions. This increases the demand for specialized diagnostic tests, including imaging studies and immunological assessments, within this age group. Early and accurate diagnosis is especially important because delayed treatment can lead to permanent joint damage, growth abnormalities, and reduced mobility. Identifying the disease at this stage allows physicians to initiate appropriate therapies that can control inflammation, preserve joint function, and improve long-term outcomes.
By End-User Analysis
Hospitals and specialty clinics are forecasted to dominate the end-user segment of the Global Juvenile Idiopathic Arthritis Diagnostic Market. The diagnosis and management of JIA are complex processes that typically require coordinated care from multiple healthcare professionals, including pediatric rheumatologists, radiologists, ophthalmologists, and laboratory specialists. Hospitals and specialized pediatric centers are well equipped to provide this multidisciplinary approach, making them the primary settings where comprehensive diagnostic evaluations take place.
Major hospitals, particularly children's hospitals and academic medical centers, possess advanced diagnostic infrastructure such as MRI scanners, high-resolution ultrasound equipment, and sophisticated laboratory facilities capable of conducting complex immunological assays. These resources enable clinicians to perform detailed assessments of joint inflammation, immune markers, and disease progression in a single healthcare setting. The availability of such integrated diagnostic services improves the accuracy of diagnoses and facilitates faster treatment decisions.
Specialty clinics dedicated to pediatric rheumatology also play an important role as referral centers for complex or severe cases. Patients who initially present with persistent joint pain or unexplained inflammation are often referred to these clinics for specialized evaluation and confirmation of JIA. These facilities provide expert clinical interpretation of imaging findings, laboratory results, and patient history to ensure a precise diagnosis.
The Global Juvenile Idiopathic Arthritis Diagnostic Market Report is segmented on the basis of the following:
By Diagnostic Type
- Laboratory Tests
- Blood Tests
- Inflammatory Marker Tests
- Genetic Tests
- Imaging Tests
- Magnetic Resonance Imaging (MRI)
- Ultrasound (including Power Doppler)
- X-Ray
- Clinical Examination
- Physical Joint Examination
- Medical History Evaluation
By Test Type
- Complete Blood Count (CBC)
- Inflammatory Marker Tests
- C-Reactive Protein (CRP)
- Erythrocyte Sedimentation Rate (ESR)
- Autoantibody/Immunological Tests
- Antinuclear Antibody (ANA) Test
- Rheumatoid Factor (RF) Test
- Anti-Cyclic Citrullinated Peptide (Anti-CCP) Test
- Genetic Testing
- Urinalysis
- Others
By Age Group
- 0-5 Years (Infants & Preschool)
- 6-12 Years (School-age)
- 13-18 Years (Adolescents)
By End-User
- Hospitals & Specialty Clinics
- Diagnostic Laboratories
- Research & Academic Institutes
- Ambulatory Surgical Centers
Impact of Artificial Intelligence in the Global Juvenile Idiopathic Arthritis Diagnostic Market
- AI-Powered Medical Imaging Analysis: AI algorithms are being developed to analyze musculoskeletal ultrasound and MRI images, automatically detecting and quantifying synovitis, joint effusion, and bone erosions with high precision, aiding radiologists and rheumatologists.
- Predictive Modeling for Disease Onset: AI models can analyze genetic, immunological, and clinical data to identify children at high risk of developing JIA, potentially enabling pre-symptomatic monitoring and earlier intervention strategies.
- Integration of Multi-Omics Data for Subtyping: Machine learning algorithms can integrate complex datasets from genomics, proteomics, and clinical records to identify novel JIA subtypes and stratify patients more accurately than current methods.
- Clinical Decision Support Systems (CDSS): AI-powered CDSS can assist general pediatricians in evaluating children with musculoskeletal complaints, providing guidance on when to order specific tests and refer to a specialist, reducing diagnostic delays.
- Automated Analysis of Laboratory Data: AI can be used to detect subtle patterns in routine lab results (e.g., CBC, inflammatory markers) that may be indicative of an underlying autoimmune condition, flagging at-risk children for further evaluation.
Global Juvenile Idiopathic Arthritis Diagnostic Market: Regional Analysis
Region with the Largest Revenue Share
North America is projected to dominate the regional segment with the highest market share, anticipated to hold approximately 37.5% of the total market revenue by the end of 2026. This is due to a highly advanced healthcare infrastructure, widespread availability of specialized pediatric rheumatology centers, and high adoption of cutting-edge diagnostic technologies. The region benefits from strong reimbursement frameworks, significant healthcare spending, and high awareness among both clinicians and the public regarding childhood rheumatic diseases. The presence of major diagnostic manufacturers and active patient advocacy groups further strengthens market demand. The United States accounts for the largest share within North America due to its large population, high disease awareness, and concentration of leading children's hospitals. Although Asia Pacific is the fastest-growing region, North America continues to hold the largest revenue share due to its mature market and high per-patient diagnostic spending.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Region with the Highest CAGR
Asia-Pacific holds the highest CAGR and is poised to achieve rapid market share growth due to its large pediatric population, improving healthcare infrastructure, and increasing awareness of chronic childhood diseases. Countries like China, India, and Japan are investing heavily in expanding specialized healthcare services, including pediatric rheumatology. Rising disposable incomes and the growth of medical tourism in countries like Thailand and Malaysia are also contributing factors. The vast number of undiagnosed or misdiagnosed JIA cases in the region represents a significant growth opportunity. Partnerships between global diagnostic companies and local distributors, combined with efforts to train more pediatric rheumatologists, are making diagnosis more accessible. This, combined with a huge potential patient pool, positions APAC as the fastest-growing market for JIA diagnostic systems.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Global Juvenile Idiopathic Arthritis Diagnostic Market: Competitive Landscape
The Global Juvenile Idiopathic Arthritis Diagnostic Market is moderately consolidated, featuring a mix of large in-vitro diagnostics (IVD) companies, specialized immunology assay developers, and advanced medical imaging firms. Leading players like Siemens Healthineers, F. Hoffmann-La Roche AG, and Abbott Laboratories leverage their vast installed bases of laboratory equipment and comprehensive immunoassay portfolios to offer key diagnostic tests. Specialized players such as Euroimmun (a PerkinElmer company), Inova Diagnostics (a Werfen company), and Thermo Fisher Scientific are driving market dynamics with advanced autoantibody testing solutions and multiplex platforms.
Medical imaging giants like GE Healthcare, Canon Medical Systems, and Esaote SpA play a crucial role by providing high-resolution ultrasound and MRI systems optimized for musculoskeletal applications. Niche players and research-focused entities are also active, developing novel biomarker assays and AI-powered analysis software.
Some of the prominent players in the Global Juvenile Idiopathic Arthritis Diagnostic Market are:
- F. Hoffmann-La Roche AG
- Abbott Laboratories
- Siemens Healthineers AG
- Thermo Fisher Scientific Inc.
- Bio-Rad Laboratories, Inc.
- Euroimmun AG (PerkinElmer Inc.)
- Inova Diagnostics (Werfen)
- Quest Diagnostics Incorporated
- Laboratory Corporation of America Holdings (LabCorp)
- GE Healthcare
- Canon Medical Systems Corporation
- Esaote SpA
- Samsung Medison Co., Ltd.
- Danaher Corporation (Beckman Coulter)
- Sysmex Corporation
- Becton, Dickinson and Company (BD)
- bioMérieux SA
- DiaSorin S.p.A.
- Trinity Biotech plc
- Other Key Players
Recent Developments in the Global Juvenile Idiopathic Arthritis Diagnostic Market
- November 2025: Thermo Fisher Scientific launched a new multiplex immunoassay panel capable of simultaneously measuring 12 key cytokines and autoantibodies associated with JIA, enabling a more comprehensive immunological profile from a single small blood sample.
- October 2025: Siemens Healthineers announced a collaboration with a leading AI firm to integrate deep learning algorithms into its ultrasound systems for automated detection and scoring of synovitis in pediatric patients.
- September 2025: Quest Diagnostics expanded its pediatric autoimmune test menu, introducing a new proprietary genetic risk score for JIA to help clinicians assess susceptibility in high-risk populations.
- August 2025: Canon Medical received FDA clearance for a new pediatric-specific software upgrade for its MRI systems, significantly reducing scan times and motion artifacts in young children without the need for sedation.
- July 2025: A consortium of European research institutions, partly funded by the EU, launched a major initiative to identify and validate novel urinary biomarkers for non-invasive monitoring of JIA disease activity.
- June 2025: bioMérieux partnered with a major children's hospital network in Asia to conduct a large-scale study validating its new biomarker test for distinguishing JIA subtypes, aiming to improve diagnostic accuracy in the region.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 1,628.5 Mn |
| Forecast Value (2035) |
USD 3,339.4 Mn |
| CAGR (2026–2035) |
8.3% |
| The US Market Size (2026) |
USD 513.5 Mn |
| Historical Data |
2020 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Diagnostic Type (Laboratory Tests, Imaging Tests, Clinical Examination), By Test Type (Complete Blood Count (CBC), Inflammatory Marker Tests, Autoantibody/Immunological Tests, Genetic Testing, Urinalysis, Others), By Age Group (0–5 Years (Infants & Preschool), 6–12 Years (School-age), 13–18 Years (Adolescents)), By End-User (Hospitals & Specialty Clinics, Diagnostic Laboratories, Research & Academic Institutes, Ambulatory Surgical Centers) |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
| Prominent Players |
F. Hoffmann-La Roche AG, Abbott Laboratories, Siemens Healthineers AG, Thermo Fisher Scientific Inc., Bio-Rad Laboratories, Inc., Euroimmun AG (PerkinElmer Inc.), Inova Diagnostics (Werfen), Quest Diagnostics Incorporated, Laboratory Corporation of America Holdings (LabCorp), GE Healthcare, Canon Medical Systems Corporation, Esaote SpA, Samsung Medison Co., Ltd., Danaher Corporation (Beckman Coulter), Sysmex Corporation, Becton, Dickinson and Company (BD), bioMérieux SA, DiaSorin S.p.A., Trinity Biotech plc., and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Global Juvenile Idiopathic Arthritis Diagnostic Market?
▾ The Global Juvenile Idiopathic Arthritis Diagnostic Market size is estimated to have a value of USD 1,628.3 million in 2026 and is expected to reach USD 3,339.4 million by the end of 2035.
What is the growth rate in the Global Juvenile Idiopathic Arthritis Diagnostic Market?
▾ The market is growing at a CAGR of 8.3 percent over the forecasted period of 2026-2035.
What is the size of the US Juvenile Idiopathic Arthritis Diagnostic Market?
▾ The US Juvenile Idiopathic Arthritis Diagnostic Market is projected to be valued at USD 513.5 million in 2026. It is expected to witness subsequent growth as it holds USD 1,009.2 million in 2035 at a CAGR of 7.8%.
Which region accounted for the largest Global Juvenile Idiopathic Arthritis Diagnostic Market?
▾ North America is expected to have the largest market share in the Global Juvenile Idiopathic Arthritis Diagnostic Market with a share of about 37.5% in 2026.
Who are the key players in the Global Juvenile Idiopathic Arthritis Diagnostic Market?
▾ Some of the major key players in the Global Juvenile Idiopathic Arthritis Diagnostic Market are F. Hoffmann-La Roche AG, Siemens Healthineers, Abbott Laboratories, Thermo Fisher Scientific, Euroimmun, Inova Diagnostics, and many others.