Market Overview
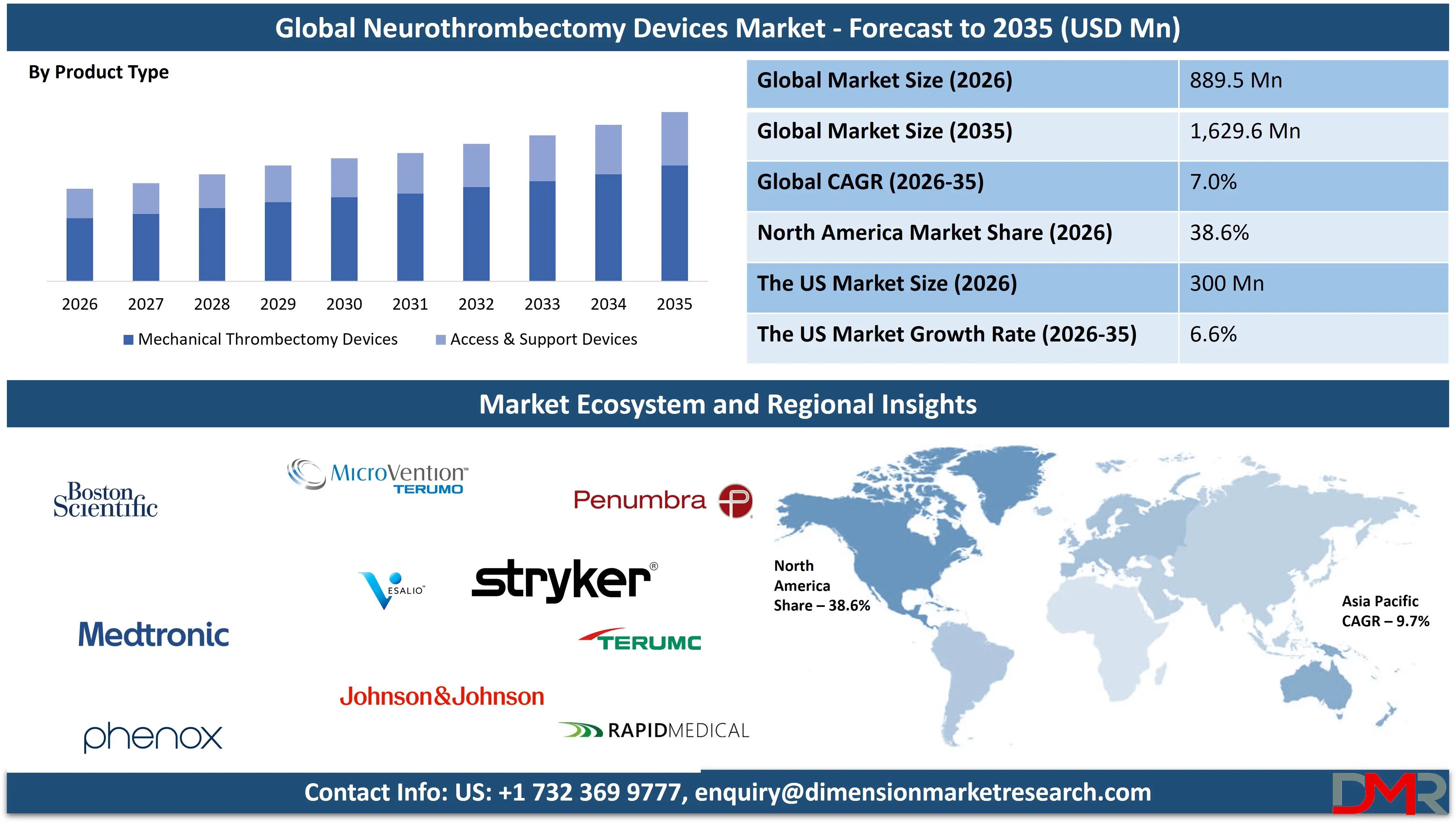
The Global Neurothrombectomy Devices Market size is projected to reach USD 889.5 million in 2026 and grow at a compound annual growth rate of 7.0% to reach a value of USD 1,629.6 million in 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Neurothrombectomy devices are specialized minimally invasive medical instruments used to remove blood clots from cerebral arteries during acute ischemic stroke and other neurovascular occlusions. These devices include stent retrievers, aspiration/suction catheters, combined stent–aspiration systems, microcatheters, balloon guide catheters, guidewires, and introducer sheaths. They are deployed via catheter-based endovascular procedures to rapidly restore blood flow to the brain, minimizing neurological damage and improving functional outcomes. As stroke remains one of the leading causes of death and disability worldwide, these devices play a vital role in modern neurointerventional therapy and emergency stroke management pathways.
Growing stroke incidence, aging populations, and improved awareness of time-sensitive stroke care are accelerating procedural volumes. Advances in imaging technologies, including perfusion imaging and AI-assisted triage tools, are enabling faster patient selection and expanding eligibility criteria beyond traditional time windows. The shift toward comprehensive stroke centers and integrated care networks is also strengthening device adoption globally.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Also, ongoing innovation in catheter flexibility, clot retrieval efficiency, and reduced complication rates is reshaping competitive dynamics. Strategic collaborations between device manufacturers and hospital networks, along with regulatory clearances for expanded indications such as medium vessel occlusions, are supporting broader clinical acceptance and sustained industry expansion.
The US Neurothrombectomy Devices Market
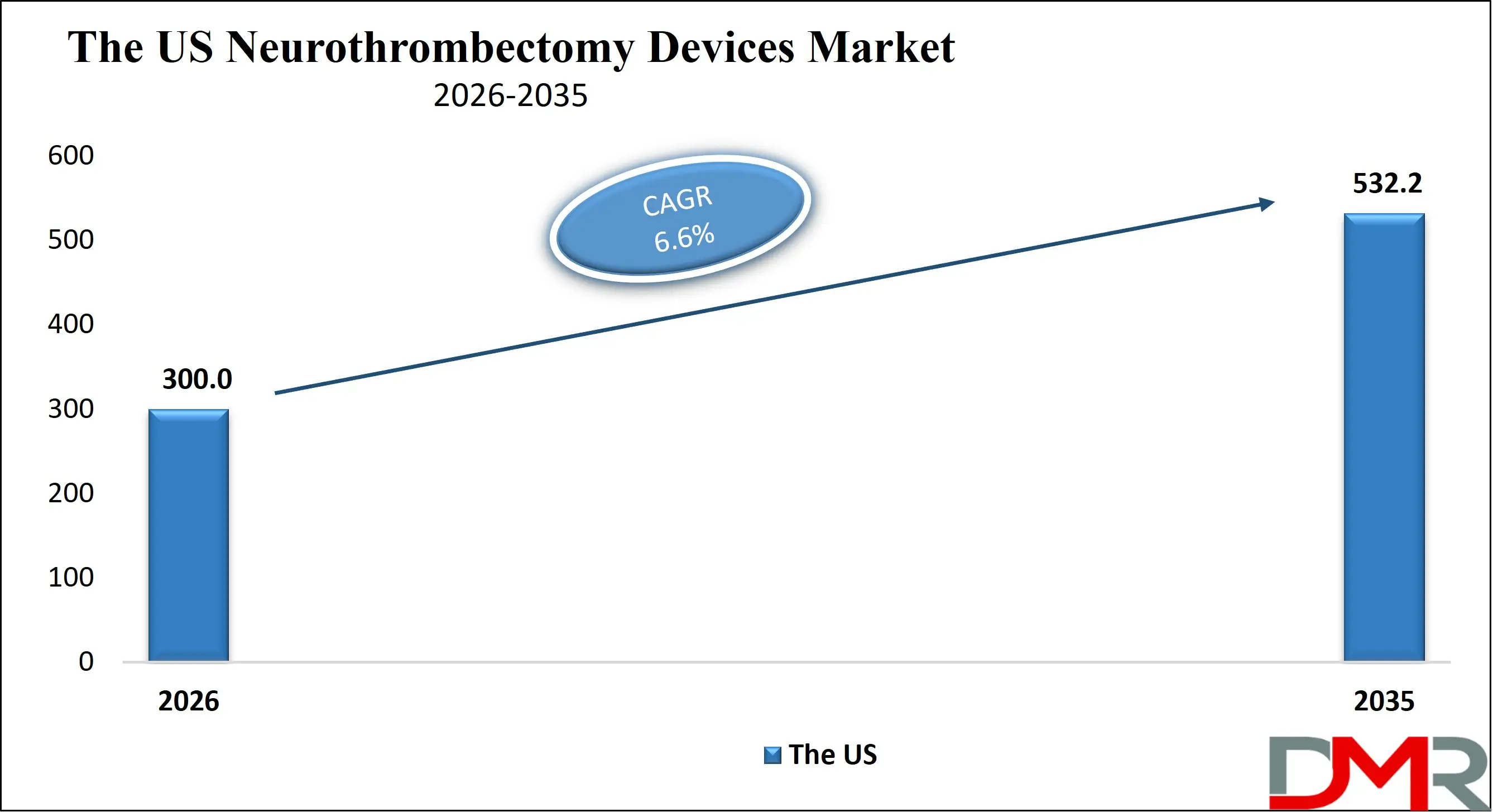
The US Neurothrombectomy Devices Market size is projected to reach USD 300.0 million in 2026 at a compound annual growth rate of 6.6% over its forecast period.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US represents a highly developed landscape driven by advanced healthcare infrastructure, high stroke awareness, and widespread adoption of endovascular therapy. The presence of comprehensive stroke centers, favorable reimbursement frameworks under Medicare and private insurers, and strong clinical guideline support have contributed to robust procedural growth. Continuous FDA approvals for next-generation stent retrievers and aspiration systems further strengthen innovation cycles. In addition, public health initiatives promoting early stroke recognition and rapid emergency response are increasing patient eligibility for mechanical thrombectomy, reinforcing the country’s leadership in neurointerventional adoption and clinical research.
Europe Neurothrombectomy Devices Market
Europe Neurothrombectomy Devices Market size is projected to reach USD 251.9 million in 2026 at a compound annual growth rate of 6.8% over its forecast period.
Europe demonstrates steady expansion supported by structured stroke networks and strong regulatory oversight under the Medical Device Regulation (MDR) framework. Regional initiatives aligned with the European Stroke Action Plan are enhancing access to thrombectomy services across member states. Countries such as Germany, France, and the UK are investing in comprehensive stroke centers and mobile stroke units. Cross-border research collaborations and increasing funding for neurovascular care are accelerating innovation. Although reimbursement structures vary by country, harmonized quality standards and government-backed healthcare systems continue to promote consistent uptake of advanced thrombectomy technologies.
Japan Neurothrombectomy Devices Market
Japan Neurothrombectomy Devices Market size is projected to reach USD 44.5 million in 2026 at a compound annual growth rate of 7.1% over its forecast period.
Japan’s market is advancing rapidly due to its aging population and high burden of cerebrovascular disease. Government healthcare coverage and strong hospital infrastructure enable broad access to advanced endovascular procedures. Japanese neurointerventionalists are early adopters of compact, highly precise catheter systems tailored to complex vascular anatomies. National initiatives aimed at reducing stroke-related disability and improving rapid transport systems further support procedural growth. However, cost containment pressures and strict regulatory approval timelines remain challenges. Despite these constraints, technological refinement and high procedural standards position Japan as a significant contributor to regional innovation.
Neurothrombectomy Devices Market: Key Takeaways
- Market Growth: The Neurothrombectomy Devices Market size is expected to grow by USD 684.7 million, at a CAGR of 7.0%, during the forecasted period of 2027 to 2035.
- By Product Type: The mechanical thrombectomy devices segment is anticipated to get the majority share of the Neurothrombectomy Devices Market in 2026.
- By Indication: The acute ishemic stroke segment is expected to get the largest revenue share in 2026 in the Neurothrombectomy Devices Market.
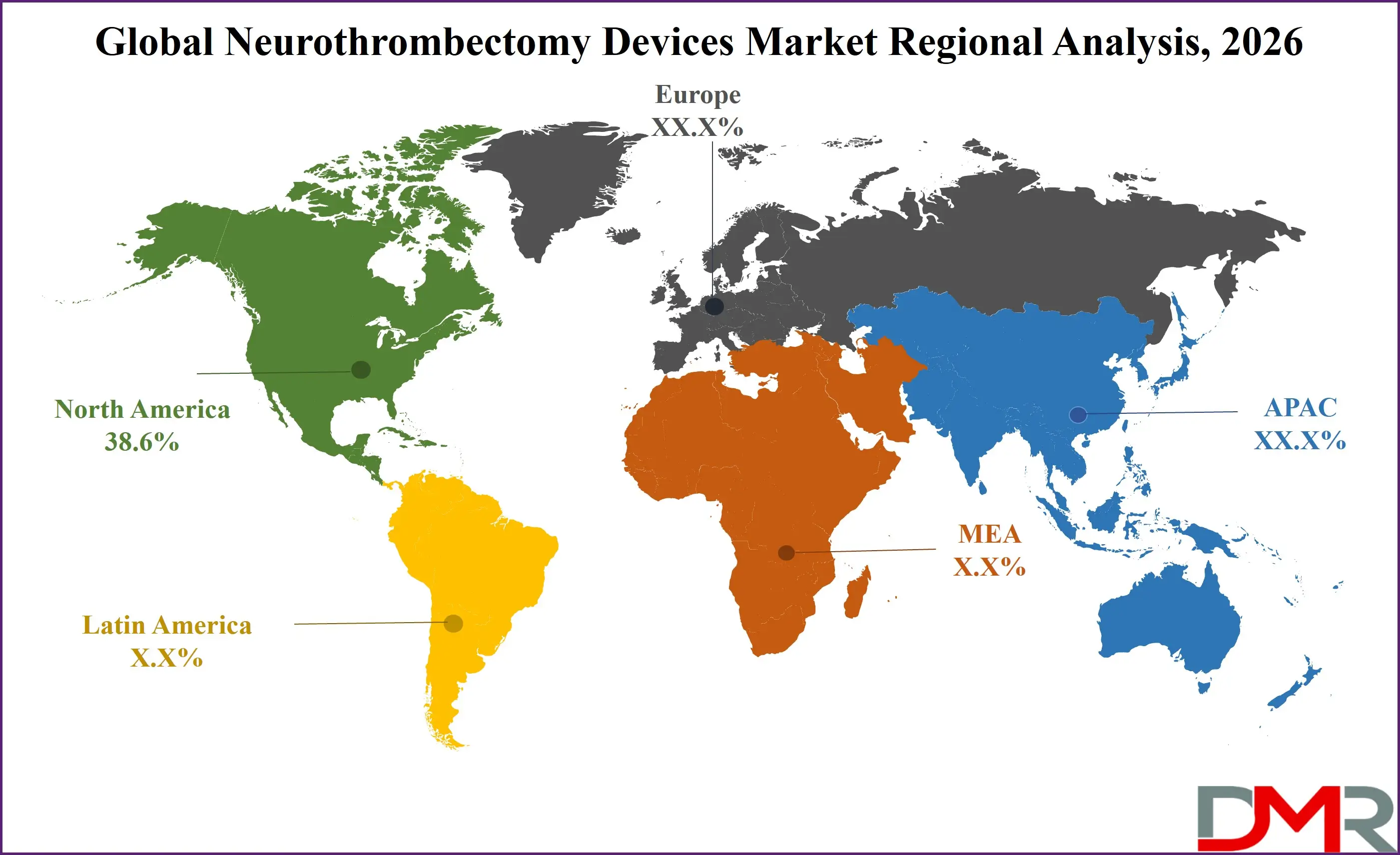
- Regional Insight: North America is expected to hold a 38.6% share of revenue in the Global Neurothrombectomy Devices Market in 2026.
- Use Cases: Some of the use cases of Neurothrombectomy Devices include acute ischemic stroke, hybrid procedure, and more.
Neurothrombectomy Devices Market: Use Cases
- Acute Ischemic Stroke (LVO): Rapid clot retrieval in large vessel occlusion cases to restore cerebral blood flow and reduce long-term disability.
- Medium Vessel Occlusion (MeVO): Expanding use in smaller vessel blockages through advanced microcatheter systems.
- Cerebral Venous Sinus Thrombosis (CVST): Targeted clot extraction in venous sinus blockages to prevent intracranial pressure buildup.
- Secondary Embolic Occlusions: Removal of emboli resulting from cardiac or arterial sources.
- Rescue Therapy After IV Thrombolysis Failure: Used when clot-dissolving drugs are ineffective.
- Extended Time Window Stroke Intervention: Supported by advanced imaging for patient selection beyond conventional timelines.
- Hybrid Procedures: Combined aspiration and stent retrieval approaches for complex clot morphology.
Stats & Facts
- Centers for Disease Control and Prevention reported in 2024 that approximately 795,000 people experience a stroke annually in the United States.
- World Health Organization stated in 2024 that stroke remains the second leading cause of death globally.
- National Institute of Neurological Disorders and Stroke confirmed in 2025 that ischemic strokes account for nearly 87% of all stroke cases.
- European Stroke Organisation noted in 2024 that over 1.1 million strokes occur annually in Europe.
- The US Centers for Medicare & Medicaid Services reported in 2025 increased reimbursement coverage for mechanical thrombectomy procedures under updated payment models.
- Japan Ministry of Health, Labour and Welfare documented in 2024 that cerebrovascular disease remains a leading cause of long-term disability.
- World Bank indicated in 2024 that populations aged 65 and above account for over 10% of the global population.
- National Health Service (UK) reported in 2025 expanded access to thrombectomy services across major stroke centers.
- American Heart Association stated in 2024 that early thrombectomy significantly improves functional independence rates at 90 days post-stroke.
- Organisation for Economic Co-operation and Development highlighted in 2025 increasing healthcare expenditure across advanced economies supporting advanced neurointerventional procedures.
Market Dynamic
Driving Factors in the Neurothrombectomy Devices Market
Rising Stroke Burden and Aging Population
The global increase in aging populations significantly elevates the risk of ischemic stroke, directly expanding the addressable patient pool for neurothrombectomy devices. As life expectancy rises, comorbidities such as hypertension, diabetes, and atrial fibrillation become more prevalent, contributing to higher stroke incidence. Governments and healthcare systems are prioritizing rapid stroke response frameworks to reduce long-term disability costs. This demographic shift, combined with improved diagnostic capabilities and awareness campaigns, continues to reinforce demand for mechanical thrombectomy procedures worldwide.
Technological Advancements in Endovascular Therapy
Continuous innovation in device design—including improved radial force in stent retrievers, enhanced trackability of aspiration catheters, and hybrid techniques—has increased procedural success rates and reduced complications. Integration with advanced imaging and AI-assisted stroke triage systems accelerates patient identification and workflow efficiency. These advancements improve clinical outcomes and physician confidence, driving higher adoption across both established and emerging stroke centers.
Restraints in the Neurothrombectomy Devices Market
High Procedural and Device Costs
Neurothrombectomy procedures require sophisticated imaging systems, skilled neurointerventionalists, and high-cost single-use devices. Capital investments in angiography suites and comprehensive stroke infrastructure can limit adoption in low-resource settings. Reimbursement disparities across regions may also restrict patient access, particularly in developing economies where healthcare budgets are constrained.
Limited Access to Skilled Specialists
Successful thrombectomy depends heavily on trained neurointerventionalists and 24/7 stroke-ready facilities. Many regions face shortages of specialized physicians, leading to unequal access. Geographic disparities, especially in rural areas, delay treatment times and limit procedural volumes, thereby affecting overall industry penetration rates.
Opportunities in the Neurothrombectomy Devices Market
Expansion into Emerging Markets
Developing economies in Asia-Pacific, Latin America, and the Middle East are investing in healthcare modernization. As awareness and infrastructure improve, these regions present significant untapped procedural volumes. Public–private partnerships and training initiatives can accelerate adoption and create new revenue streams for device manufacturers.
Broader Indication Approvals
Ongoing clinical trials targeting medium vessel occlusions and extended time windows are expanding the treatable population. Regulatory approvals for new indications and device iterations enhance market scope and strengthen physician confidence in adopting next-generation solutions.
Trends in the Neurothrombectomy Devices Market
Shift Toward Combined Techniques
The Solumbra and hybrid approaches combining stent retrieval with aspiration are gaining traction due to improved recanalization rates. Physicians increasingly prefer flexible procedural strategies tailored to clot composition and vascular anatomy, influencing product development priorities.
Digital Integration and AI-Assisted Workflow
Artificial intelligence tools integrated with imaging systems are optimizing stroke triage and transfer decisions. Automated detection of large vessel occlusion supports faster intervention, reducing door-to-puncture times and enhancing overall procedural efficiency.
Impact of Artificial Intelligence in Neurothrombectomy Devices Market
- AI-Based Stroke Detection: Automated imaging analysis rapidly identifies large vessel occlusions, enabling faster treatment decisions.
- Workflow Optimization: AI-driven triage systems reduce door-to-needle and door-to-puncture times in hospitals.
- Patient Selection Enhancement: Advanced algorithms assess perfusion imaging to determine treatment eligibility beyond traditional windows.
- Procedure Planning: AI-assisted simulation tools help physicians choose optimal catheter pathways.
- Predictive Outcome Modeling: Machine learning predicts recovery probabilities, aiding clinical decisions.
- Remote Consultation Support: Cloud-based AI platforms enable neurologists to evaluate scans across networks.
- Quality Monitoring: Data analytics track procedural success rates and complications.
- Device Development Insights: AI-driven R&D accelerates material optimization and design refinement.
Research Scope and Analysis
By Product Type Analysis
Mechanical thrombectomy devices represent the dominant product category, accounting for an estimated 68.4% share of the global neurothrombectomy devices market in 2026. Their leadership is primarily driven by their central role in endovascular clot removal procedures for acute ischemic stroke, especially in large vessel occlusion (LVO) cases where rapid recanalization is critical. Stent retrievers and aspiration catheters are widely adopted due to strong clinical validation, inclusion in international stroke guidelines, and proven ability to improve 90-day functional outcomes.
Continuous advancements in device engineering, such as enhanced clot integration structures, optimized radial force, improved flexibility, and atraumatic distal tips—are improving first-pass effect rates and minimizing complications. Increasing global procedural volumes, coupled with regulatory approvals for expanded indications like medium vessel occlusions (MeVO), are further strengthening this segment’s dominance. Ongoing investments in R&D and physician training programs are expected to sustain long-term leadership.
Access and support devices are the fastest-growing segment within the product category, reflecting their indispensable role in enabling successful thrombectomy procedures. These devices—including microcatheters, balloon guide catheters, guidewires, and introducer sheaths—facilitate precise navigation through complex and often tortuous cerebral vasculature. As physicians focus on maximizing first-pass recanalization success and minimizing procedural time, demand for highly trackable, kink-resistant, and torque-responsive systems is increasing.
Technological innovations such as hydrophilic coatings, enhanced pushability, and improved compatibility with large-bore aspiration systems are accelerating adoption. Growth is also supported by rising stroke intervention volumes globally and the routine use of single-use sterile components. Emerging markets investing in comprehensive stroke kits and expanding neurointerventional infrastructure are contributing significantly to the rapid expansion of this segment.
By Technology Analysis
Stent-based thrombectomy remains the leading technology segment, projected to hold 46.7% of the market share in 2026. Its dominance is supported by extensive clinical evidence demonstrating high recanalization rates, favorable safety profiles, and improved long-term neurological outcomes. In this approach, self-expanding stent retrievers are temporarily deployed across the clot, allowing thrombus integration before controlled extraction under aspiration support. Physicians widely prefer this technique for large vessel occlusions due to its reliability and predictable performance across diverse clot compositions.
Continuous innovation in stent design—including optimized cell geometry, improved radial strength, and enhanced clot capture mechanisms—is reinforcing its clinical effectiveness. Additionally, strong endorsement from stroke management guidelines and favorable reimbursement structures across developed healthcare systems are sustaining widespread adoption and maintaining its leadership position.
Direct aspiration using the ADAPT (A Direct Aspiration First Pass Technique) approach is the fastest-growing technology segment in the market. This method emphasizes the use of large-bore aspiration catheters to directly engage and remove the clot without initial stent deployment. Its appeal lies in procedural simplicity, reduced device exchanges, shorter operating times, and potentially lower overall costs. Advancements in catheter lumen diameter, suction power optimization, and improved flexibility have significantly enhanced clot retrieval efficiency.
Many neurointerventionalists are adopting aspiration-first strategies, particularly for softer clot types and anatomically favorable cases. The technique’s ability to reduce procedural complexity and minimize device-related trauma contributes to its strong growth momentum, especially in high-volume stroke centers seeking workflow efficiency.
By Indication Analysis
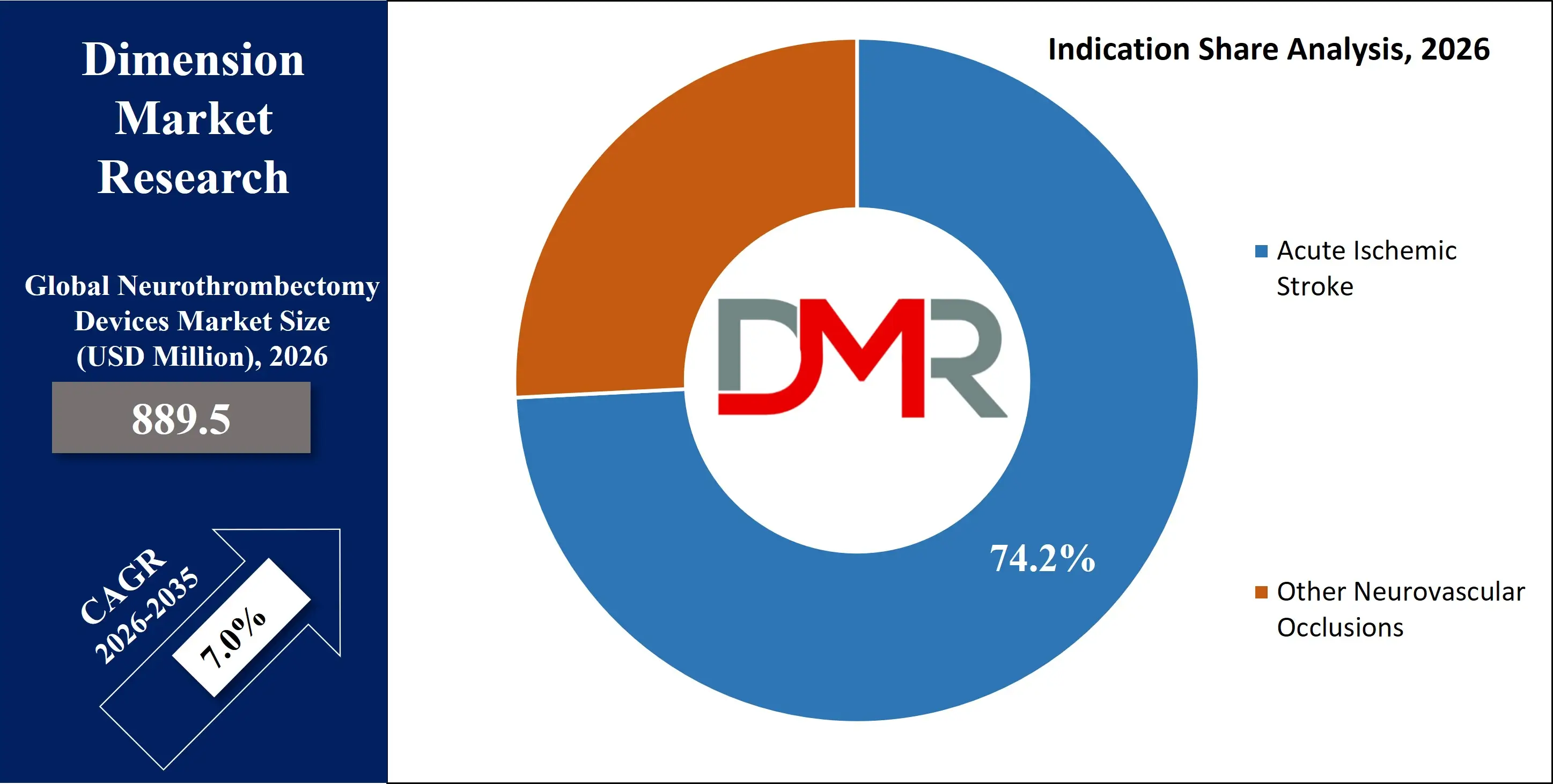
Acute ischemic stroke dominates the indication segment, anticipated to capture 74.2% market share in 2026. The high global prevalence of ischemic stroke, which accounts for the majority of all stroke cases, directly drives demand for mechanical thrombectomy procedures. Strong clinical evidence from landmark trials supports the use of thrombectomy in large vessel occlusions within defined time windows, significantly improving survival and functional independence rates.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Advances in perfusion imaging and AI-assisted patient selection are expanding treatment eligibility beyond traditional six-hour windows, increasing the addressable population. National stroke awareness campaigns and streamlined emergency transport systems are further accelerating procedural volumes. As healthcare systems prioritize reducing stroke-related disability and long-term rehabilitation costs, this segment continues to maintain its dominant position.
Other neurovascular occlusions, including cerebral venous sinus thrombosis (CVST) and secondary embolic occlusions, represent the fastest-growing indication segment. Although these conditions account for a smaller patient pool compared to ischemic stroke, increasing clinical recognition of the benefits of interventional clot removal is supporting growth. Physicians are exploring thrombectomy techniques in complex or refractory cases where medical management alone proves insufficient.
Technological advancements enabling safer navigation in delicate venous structures are encouraging broader procedural adoption. Furthermore, expanding clinical research and case studies highlighting positive outcomes are gradually strengthening confidence in these applications. As evidence continues to accumulate and regulatory pathways evolve, this niche segment is expected to witness steady expansion in specialized neurointerventional centers.
By End User Analysis
Hospitals account for the largest end-user share, projected at 52.9% in 2026, due to their central role in emergency stroke management. Most acute stroke patients are initially admitted to general hospitals equipped with emergency departments and angiography suites capable of performing endovascular procedures. Established reimbursement mechanisms, structured stroke protocols, and multidisciplinary care teams contribute to their leadership. Investments in advanced imaging technologies, hybrid operating rooms, and 24/7 neurointerventional coverage further enhance their procedural capacity.
Hospitals also benefit from high patient inflow and integrated referral systems, ensuring consistent case volumes. As governments continue to upgrade acute care infrastructure and expand access to time-sensitive stroke interventions, hospitals are expected to retain their dominant position in the market.
Comprehensive stroke centers represent the fastest-growing end-user segment, driven by their specialized capabilities in managing complex cerebrovascular cases. These centers offer round-the-clock neurointerventional expertise, advanced imaging modalities, and multidisciplinary stroke teams, enabling rapid and precise treatment decisions. Governments and healthcare authorities are increasingly certifying hospitals as comprehensive stroke centers to improve regional access to thrombectomy services.
Enhanced referral networks and telemedicine-supported triage systems are channeling eligible patients to these facilities more efficiently. Additionally, comprehensive centers often participate in clinical trials and early adoption of next-generation devices, accelerating technological uptake. Their focus on high-acuity cases and advanced procedural techniques positions them as key growth drivers within the neurothrombectomy devices market.
The Neurothrombectomy Devices Market Report is segmented on the basis of the following
By Product Type
- Mechanical Thrombectomy Devices
- Stent Retrievers
- Aspiration/Suction Catheters
- Combined Stent Retriever + Aspiration Systems
- Access & Support Devices
- Microcatheters
- Balloon Guide Catheters
- Guidewires
- Introducer Sheaths
By Technology
- Stent-Based Thrombectomy
- Direct Aspiration (ADAPT Technique)
- Combined/Hybrid Technique (Solumbra Approach)
By Indication
- Acute Ischemic Stroke
- Large Vessel Occlusion (LVO)
- Medium Vessel Occlusion (MeVO)
- Other Neurovascular Occlusions
- Cerebral Venous Sinus Thrombosis (CVST)
- Secondary Embolic Occlusions
By End User
- Hospitals
- Comprehensive Stroke Centers
- Ambulatory Surgical Centers
Regional Analysis
Leading Region in the Neurothrombectomy Devices Market
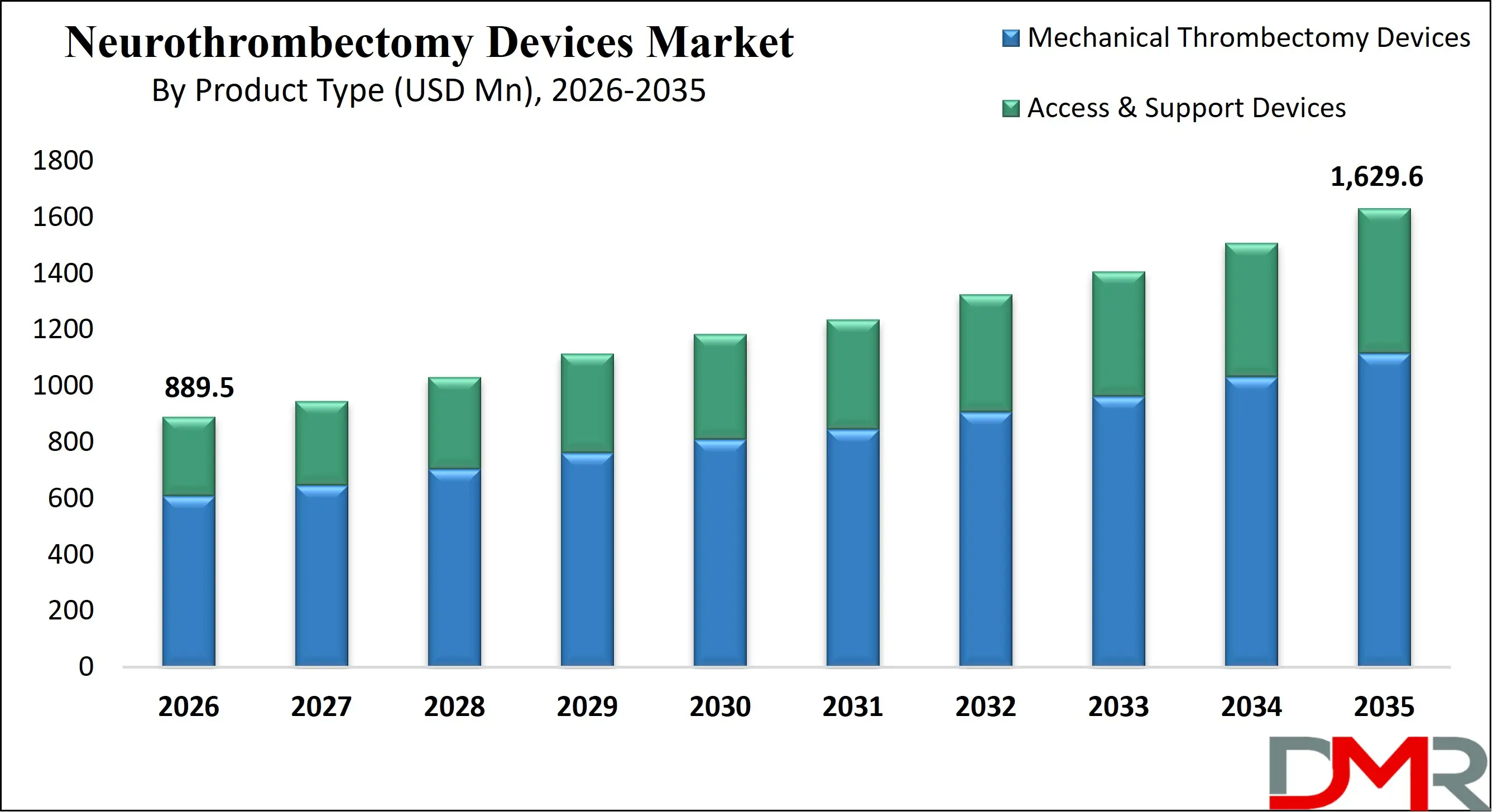
North America is projected to lead the market with an estimated 38.6% share in 2026. The region’s dominance is underpinned by advanced healthcare infrastructure, widespread availability of comprehensive stroke centers, and strong public awareness of stroke symptoms. Favorable reimbursement policies, particularly in the United States, encourage hospitals to adopt advanced thrombectomy systems.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Rapid regulatory approvals and early access to innovative devices further accelerate technology penetration. In addition, extensive clinical research activity and continuous physician training programs support procedural excellence. Well-developed emergency medical services and established stroke transfer networks enable timely interventions, improving outcomes and sustaining high procedural volumes across the region.
Fastest Growing Region in the Neurothrombectomy Devices Market
Asia-Pacific is the fastest-growing regional market, driven by rising stroke incidence, expanding healthcare investments, and improving access to advanced neurointerventional procedures. Rapid urbanization, aging populations, and increasing prevalence of lifestyle-related risk factors are contributing to higher ischemic stroke cases. Governments in countries such as China, India, and Japan are investing in stroke center expansion, physician training, and public awareness initiatives. Growing health insurance penetration and modernization of tertiary care hospitals are improving affordability and accessibility of thrombectomy procedures. Although disparities persist between urban and rural healthcare infrastructure, accelerating economic development and healthcare reforms are positioning Asia-Pacific as a major engine of future market growth.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The neurothrombectomy devices market is characterized by strong competition centered on innovation, clinical validation, and portfolio expansion. Companies focus heavily on research and development to improve first-pass success rates and device navigability. Strategic collaborations with stroke centers, acquisitions to expand product lines, and geographic expansion into emerging markets are common growth strategies. High regulatory standards and capital-intensive R&D create substantial entry barriers, protecting established players. Competitive differentiation increasingly depends on integrated solutions that combine imaging compatibility, hybrid techniques, and workflow optimization technologies.
Some of the prominent players in the global Neurothrombectomy Devices are
- Stryker
- Medtronic
- Penumbra
- Johnson & Johnson (Cerenovus)
- Terumo Corporation
- Phenox GmbH
- Rapid Medical
- MicroVention (Terumo Group)
- Balt Extrusion
- Vesalio
- Acandis GmbH
- Teleflex Incorporated
- Boston Scientific
- Edwards Lifesciences
- Abbott Laboratories
- Argon Medical Devices
- NeuroVasc Technologies
- Perflow Medical
- Koninklijke Philips
- Biomedical Solutions Inc.
- Other Key Players
Recent Developments
- In July 2025, Inari Medicalintroduced the second-generation InThrill Thrombectomy System, the first purpose-built solution for small vessel and arteriovenous (AV) access thrombectomy designed for rapid, complete clot removal. The 8F over-the-wire system includes a dedicated catheter and sheath. It features higher radial force, an offset open mouth for enhanced clot capture, internal struts for uniform clot distribution, and improved ergonomics for better usability.
- In April 2025, Anaconda Biomed has announced the enrollment and treatment of the first U.S. patient in its ATHENA investigational device exemption (IDE) clinical trial. ATHENA is a global, randomized 327-patient pivotal study evaluating the safety and effectiveness of the company’s ANA5 advanced neurovascular access funnel catheter. The IDE approval from the U.S. Food and Drug Administration followed supportive feasibility studies demonstrating high reperfusion rates, strong first-pass success, and a favorable safety profile.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 889.5 Mn |
| Forecast Value (2035) |
USD 1,629.6 Mn |
| CAGR (2026–2035) |
7.0% |
| The US Market Size (2026) |
USD 300 Mn |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors, etc. |
| Segments Covered |
By Product Type (Mechanical Thrombectomy Devices, Access & Support Devices), By Technology (Stent-Based Thrombectomy, Direct Aspiration (ADAPT Technique), Combined/Hybrid Technique (Solumbra Approach)), By Indication (Acute Ischemic Stroke, Other Neurovascular Occlusions), By End User (Hospitals, Comprehensive Stroke Centers, Ambulatory Surgical Centers) |
| Regional Coverage |
North America – US, Canada; Europe – Germany, UK, France, Russia, Spain, Italy, Benelux, Nordic, Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, Rest of MEA |
| Prominent Players |
Stryker, Medtronic, Penumbra, Johnson & Johnson (Cerenovus), Terumo Corporation, Phenox GmbH, Rapid Medical, MicroVention (Terumo Group), Balt Extrusion, Vesalio, Acandis GmbH, Teleflex Incorporated, Boston Scientific, Edwards Lifesciences, Abbott Laboratories, Argon Medical Devices, NeuroVasc Technologies, Perflow Medical, Koninklijke Philips, Biomedical Solutions Inc., and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users), and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days, and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Global Neurothrombectomy Devices Market?
▾ The Global Neurothrombectomy Devices Market size is expected to reach USD 889.5 million by 2026 and is projected to reach USD 1,629.6 million by the end of 2035.
Which region accounted for the largest Global Neurothrombectomy Devices Market?
▾ North America is expected to have the largest market share in the Global Neurothrombectomy Devices Market, with a share of about 38.6% in 2026.
How big is the Neurothrombectomy Devices Market in the US?
▾ The US Neurothrombectomy Devices market is expected to reach USD 300.0 million by 2026.
Who are the key players in the Neurothrombectomy Devices Market?
▾ Some of the major key players in the Global Neurothrombectomy Devices Market include Stryker, Medtronic, Vesalio, and others
What is the growth rate in the Global Neurothrombectomy Devices Market?
▾ The market is growing at a CAGR of 7.0 percent over the forecasted period.