Market Overview
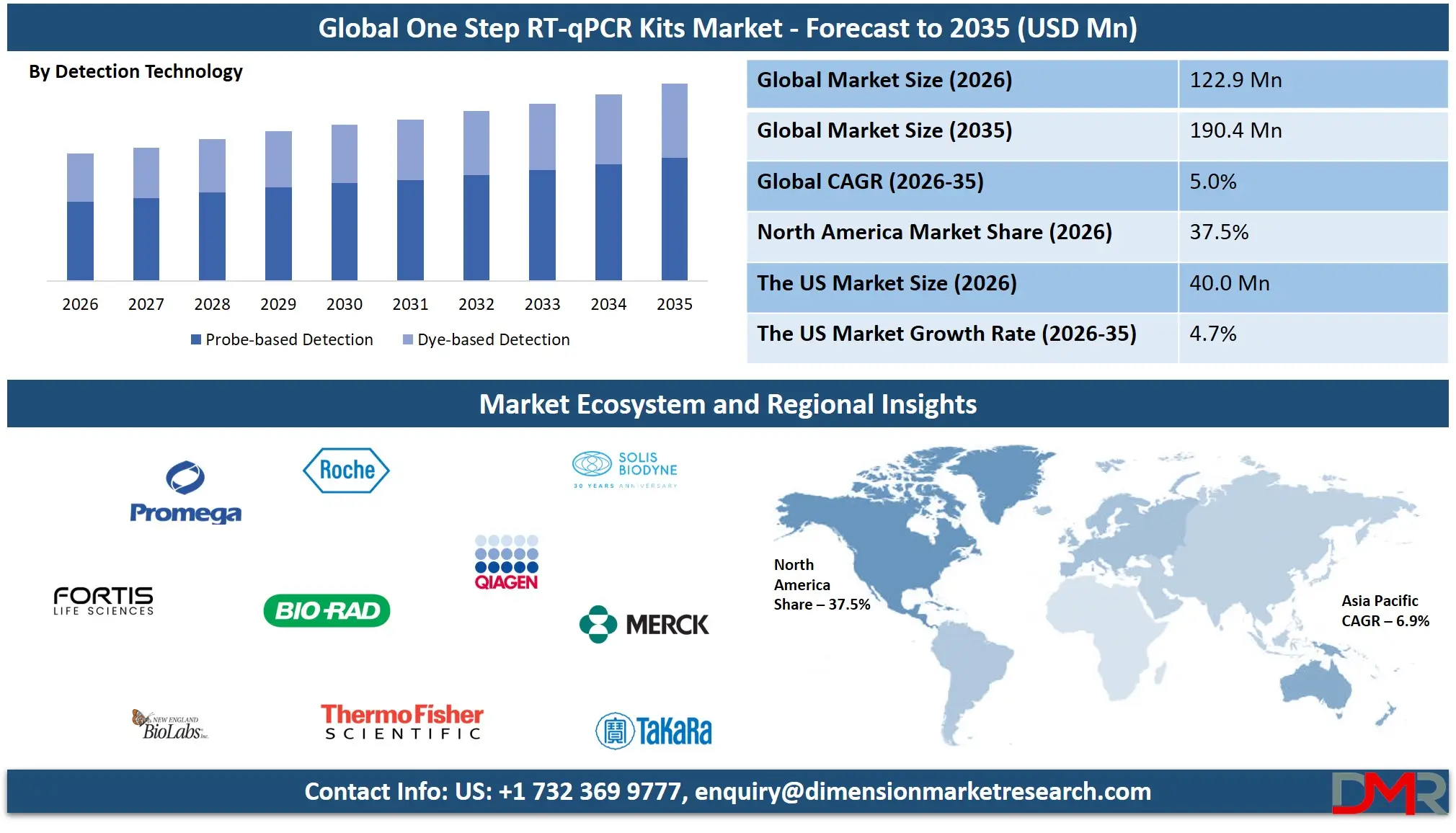
The Global One Step RT-qPCR Kits Market size is projected to reach USD 122.9 million in 2026 and grow at a compound annual growth rate of 5.0% to reach a value of USD 190.4 million in 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
One Step RT-qPCR Kits are molecular diagnostic reagents designed to perform reverse transcription and quantitative polymerase chain reaction within a single reaction tube. These kits integrate enzymes such as reverse transcriptase and DNA polymerase with optimized buffers, primers, and probes to enable simultaneous conversion of RNA into complementary DNA (cDNA) and amplification for quantitative detection. The technology is widely applied in molecular biology, clinical diagnostics, virology research, and genetic studies because it reduces contamination risks, shortens workflow time, and improves sensitivity in RNA detection. These kits are especially valuable for detecting RNA viruses, analyzing gene expression, and performing rapid molecular screening.
The importance of one step RT-qPCR kits has increased significantly due to the rising reliance on molecular diagnostics and precision medicine. Laboratories and diagnostic centers prefer these kits because they streamline complex experimental procedures into a single-step workflow, improving efficiency and reproducibility. Advancements in enzyme engineering, probe chemistry, and real-time PCR instrumentation are improving detection accuracy and reaction speed. Growing demand for rapid RNA detection in infectious disease surveillance, oncology research, and genetic testing is accelerating technological progress in kit formulations and reaction optimization.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Significant industry expansion is through innovations in multiplex PCR capability, higher reaction throughput, and enhanced reagent stability. Biotechnology firms and diagnostic manufacturers are investing in automated PCR workflows, integrated reagent systems, and advanced detection chemistries to support large-scale testing needs. Collaborations between research institutions and biotech companies are driving improvements in sensitivity, reagent shelf life, and compatibility with modern real-time PCR platforms. Increasing investments in molecular diagnostics infrastructure and expanding research funding are shaping the technological evolution of One Step RT-qPCR Kits across clinical and research environments.
The US One Step RT-qPCR Kits Market
The US One Step RT-qPCR Kits Market size is projected to reach USD 40.0 million in 2026 at a compound annual growth rate of 4.7% over its forecast period.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US represents a technologically advanced environment for one step RT-qPCR kits due to its strong biotechnology sector, well-established diagnostic infrastructure, and high research funding. Academic laboratories, pharmaceutical companies, and clinical diagnostic facilities actively utilize these kits for infectious disease detection, cancer research, and genetic studies. Government initiatives supporting genomic research and precision medicine programs further strengthen the adoption of advanced PCR technologies. The presence of major biotechnology manufacturers and reagent developers encourages continuous product innovation and competition. Additionally, regulatory oversight by agencies such as the FDA promotes standardized molecular testing protocols. Increased investment in diagnostic automation and large-scale testing capabilities supports the expansion of RT-qPCR applications across healthcare, research laboratories, and pharmaceutical development sectors in the United States.
Europe One Step RT-qPCR Kits Market
Europe One Step RT-qPCR Kits Market size is projected to reach USD 30.7 million in 2026 at a compound annual growth rate of 4.9% over its forecast period.
Europe demonstrates strong adoption of one step RT-qPCR kits due to robust research ecosystems, supportive healthcare policies, and increasing emphasis on molecular diagnostics. Regional initiatives such as the European Green Deal and Horizon Europe research programs promote biotechnology innovation and laboratory modernization. Countries including Germany, France, and the United Kingdom maintain advanced diagnostic infrastructures and strong academic research networks that rely heavily on RT-qPCR technologies. Regulatory frameworks under the In Vitro Diagnostic Regulation (IVDR) are improving the quality and standardization of molecular testing products across the region. Growing focus on infectious disease monitoring, genetic research, and biotechnology development continues to support demand for efficient PCR reagents. Collaborative research programs between universities and biotech companies are further strengthening technological advancements in One Step RT-qPCR kits.
Japan One Step RT-qPCR Kits Market
Japan One Step RT-qPCR Kits Market size is projected to reach USD 4.9 million in 2026 at a compound annual growth rate of 5.5% over its forecast period.
Japan's one step RT-qPCR kits market is influenced by its advanced biotechnology industry, strong pharmaceutical sector, and government-supported research programs. The country maintains significant investments in life sciences research, genomic medicine, and diagnostic technologies. Academic institutions and pharmaceutical companies frequently utilize RT-qPCR kits for gene expression studies, RNA analysis, and viral detection. Japan's aging population has increased the focus on disease research, including oncology and infectious disease diagnostics, which further supports molecular testing adoption. Government programs promoting biotechnology innovation and laboratory automation are encouraging development of advanced PCR reagents and high-precision diagnostic tools. Despite high technological capability, challenges such as strict regulatory approval processes and cost considerations influence market expansion. Nevertheless, continuous innovation and research funding continue to support steady growth in Japan's RT-qPCR kit market.
One Step RT-qPCR Kits Market: Key Takeaways
- Market Growth: The One Step RT-qPCR Kits Market size is expected to grow by USD 62.0 million, at a CAGR of 5.0%, during the forecasted period of 2027 to 2035.
- By Detection Technology: The probe-based detection segment is anticipated to get the majority share of the One Step RT-qPCR Kits market in 2026.
- By Application: The infectious disease detection segment is expected to get the largest revenue share in 2026 in the One Step RT-qPCR Kits market.
- Regional Insight: North America is expected to hold a 37.5% share of revenue in the global One Step RT-qPCR Kits market in 2026.
- Use Cases: Some of the use cases of One Step RT-qPCR Kits include food RNA quantification, multiplex PCR analysis, and more.
One Step RT-qPCR Kits Market: Use Cases:
- Infectious Disease Detection: One Step RT-qPCR Kits are extensively used for detecting RNA viruses such as influenza, coronaviruses, and other viral pathogens. The technology allows rapid amplification and detection of viral RNA in clinical samples, enabling early diagnosis and effective disease management in hospitals and diagnostic laboratories.
- Gene Expression Analysis: Researchers use these kits to quantify gene expression levels in biological samples. By converting RNA into cDNA and amplifying it simultaneously, scientists can study transcription activity, cellular responses, and molecular pathways in biomedical research.
- RNA Quantification: These kits provide highly sensitive measurement of RNA concentration in research samples. Laboratories studying transcriptomics and gene regulation rely on One Step RT-qPCR to accurately quantify RNA molecules across various biological experiments.
- Mutation and Variant Detection: One Step RT-qPCR kits enable detection of genetic mutations and RNA variants, supporting genetic research and clinical diagnostics. This capability is particularly useful in oncology studies and viral mutation monitoring.
- Pharmaceutical Drug Development: Biotechnology and pharmaceutical companies use RT-qPCR kits to analyze gene expression changes during drug discovery and development. These analyses help evaluate drug efficacy, toxicity, and biological impact.
- Multiplex PCR Analysis: Advanced kits allow simultaneous detection of multiple RNA targets within a single reaction. This multiplex capability improves testing efficiency in diagnostic laboratories and large-scale screening programs.
- Academic Research Applications: Universities and research institutes utilize One Step RT-qPCR kits in molecular biology studies involving gene regulation, viral replication, and genetic pathway analysis.
Stats & Facts
- World Health Organization (WHO) reported in 2024 that over 70% of molecular diagnostic tests globally rely on PCR-based technologies for pathogen detection.
- U.S. Centers for Disease Control and Prevention (CDC) reported in 2024 that RT-PCR remains the primary molecular method used for detecting RNA viruses in clinical laboratories across the United States.
- National Institutes of Health (NIH) stated in 2024 that federal biomedical research funding exceeded USD 47 billion, supporting extensive molecular biology and PCR-based research activities.
- European Commission reported in 2024 that the Horizon Europe research program allocated approximately EUR 95.5 billion (USD 110.3 billion) for scientific innovation including biotechnology and genomic research.
- European Centre for Disease Prevention and Control (ECDC) indicated in 2024 that PCR-based diagnostics remain the gold standard for infectious disease monitoring across EU laboratories.
- Japan Ministry of Health, Labour and Welfare reported in 2024 that Japan continues to expand molecular diagnostic testing infrastructure in hospitals and research laboratories.
- National Science Foundation (NSF) reported in 2024 that life sciences research funding in the United States exceeded USD 9 billion supporting advanced molecular techniques.
- Organisation for Economic Co-operation and Development (OECD) reported in 2024 that biotechnology R&D spending among member countries increased by more than 6% annually.
- World Bank reported in 2025 that global healthcare expenditure continues to rise, supporting demand for advanced diagnostic technologies including molecular testing.
- UNESCO Institute for Statistics reported in 2024 that global research and development spending surpassed USD 1.7 trillion, significantly benefiting life sciences research.
- U.S. Department of Health and Human Services reported in 2025 that federal investments in genomic research programs continue to expand across national laboratories.
- European Medicines Agency (EMA) reported in 2024 that molecular diagnostic technologies are increasingly integrated into clinical trial protocols across Europe.
Market Dynamic
Driving Factors in the One Step RT-qPCR Kits Market
Increasing Demand for Rapid Molecular Diagnostics
The growing demand for rapid and accurate molecular diagnostic methods is a major factor driving the adoption of One Step RT-qPCR Kits. Healthcare providers increasingly require fast detection of infectious diseases, genetic disorders, and viral pathogens. Traditional diagnostic techniques often require longer processing times and multiple laboratory steps, whereas One Step RT-qPCR simplifies workflows and reduces testing time. The increasing prevalence of viral infections and the need for real-time pathogen surveillance further emphasize the importance of efficient molecular testing. Hospitals, diagnostic laboratories, and public health institutions are expanding their molecular testing capabilities, which is significantly increasing the utilization of RT-qPCR kits across clinical and research environments.
Advancements in PCR Technology and Enzyme Engineering
Technological innovations in PCR enzymes, reagents, and real-time detection systems are significantly improving the performance of One Step RT-qPCR Kits. Enhanced reverse transcriptases with higher thermal stability and improved DNA polymerases are increasing reaction efficiency and sensitivity. Modern kits also incorporate multiplex detection capabilities, enabling simultaneous amplification of multiple RNA targets. These advancements improve laboratory productivity and reduce operational costs by allowing more data to be generated from a single experiment. Continuous improvements in reagent formulation, probe chemistry, and automated PCR systems are expanding the applications of RT-qPCR technologies in diagnostics, research, and pharmaceutical development.
Restraints in the One Step RT-qPCR Kits Market
High Cost of Advanced Molecular Diagnostic Reagents
One of the major challenges affecting the market is the relatively high cost associated with advanced RT-qPCR kits and related laboratory equipment. High-quality enzymes, specialized reagents, and precision detection probes increase manufacturing costs. Laboratories in developing regions may face budget constraints when adopting advanced molecular diagnostic technologies. Additionally, real-time PCR instruments required to perform RT-qPCR testing involve significant capital investment. These cost barriers can limit accessibility for smaller laboratories and research institutes. Although technological improvements are gradually reducing production costs, price sensitivity remains a significant constraint for widespread adoption in cost-conscious healthcare systems.
Technical Complexity and Laboratory Skill Requirements
RT-qPCR testing requires trained laboratory professionals with expertise in molecular biology techniques. Errors in sample preparation, RNA extraction, or reaction setup can influence results and reduce diagnostic accuracy. Maintaining contamination-free laboratory conditions is also critical for reliable PCR testing. Many clinical laboratories in emerging markets face shortages of skilled technicians capable of performing advanced molecular diagnostic procedures. Furthermore, strict quality control procedures and laboratory accreditation requirements may increase operational complexity. These technical challenges can slow the expansion of RT-qPCR kit usage, particularly in regions with limited laboratory infrastructure.
Opportunities in the One Step RT-qPCR Kits Market
Expansion of Precision Medicine and Genomic Research
Precision medicine and genomic research initiatives are creating significant opportunities for One Step RT-qPCR Kits. Healthcare systems are increasingly adopting personalized treatment approaches based on genetic information and gene expression profiles. RT-qPCR plays a critical role in analyzing RNA biomarkers, gene regulation patterns, and molecular pathways associated with diseases. Research institutions and pharmaceutical companies are investing heavily in genomic technologies to develop targeted therapies. As personalized medicine programs expand globally, the demand for highly sensitive molecular testing methods such as RT-qPCR will continue to grow.
Increasing Adoption in Emerging Healthcare Markets
Emerging economies in Asia-Pacific, Latin America, and parts of Africa are expanding their healthcare infrastructure and diagnostic capabilities. Governments are investing in modern laboratories, biotechnology research centers, and infectious disease monitoring systems. These investments are increasing the demand for advanced molecular diagnostic reagents including One Step RT-qPCR Kits. Additionally, rising awareness about early disease detection and improved healthcare access are encouraging diagnostic laboratories to adopt modern testing technologies. As molecular diagnostic infrastructure improves in developing regions, significant growth opportunities are expected for RT-qPCR kit manufacturers.
Trends in the One Step RT-qPCR Kits Market
Growth of Multiplex RT-qPCR Technologies
Multiplex RT-qPCR technologies are becoming increasingly popular in molecular diagnostics and research applications. These advanced systems allow simultaneous detection of multiple RNA targets in a single reaction, significantly improving laboratory efficiency. Diagnostic laboratories benefit from reduced reagent consumption, faster testing workflows, and higher throughput capabilities. Multiplex PCR assays are particularly valuable in infectious disease diagnostics, where multiple pathogens can be screened simultaneously. As researchers continue to develop advanced probe chemistries and detection platforms, multiplex RT-qPCR is becoming a critical trend shaping the evolution of molecular diagnostic testing.
Integration with Automated Laboratory Workflows
Automation is transforming molecular biology laboratories by improving reproducibility and reducing manual errors. Many laboratories are integrating One Step RT-qPCR kits with automated liquid handling systems, robotic sample preparation platforms, and high-throughput PCR instruments. Automated workflows allow laboratories to process large sample volumes efficiently, which is essential for large-scale diagnostic testing and research studies. Automation also helps standardize experimental procedures, improving result accuracy and reducing variability. As laboratory automation technologies continue to evolve, the integration of RT-qPCR kits into automated diagnostic platforms is expected to become a defining trend in the market.
Impact of Artificial Intelligence in One Step RT-qPCR Kits Market
- AI-Based Data Interpretation: Artificial intelligence algorithms help analyze RT-qPCR amplification curves and fluorescence signals with high precision. This improves result accuracy and reduces the chances of false positives or negatives in diagnostic testing.
- Automated Assay Optimization: AI tools assist laboratories in optimizing primer design, probe selection, and reaction conditions. This reduces experimental trial time and improves reaction efficiency in RT-qPCR workflows.
- Predictive Diagnostic Analytics: Machine learning models can evaluate RT-qPCR results alongside clinical data to predict disease progression or infection severity, supporting better clinical decision-making.
- Laboratory Workflow Automation: AI-driven robotic systems automate sample handling, reagent preparation, and PCR setup, significantly increasing laboratory throughput and reducing human error.
- Quality Control and Error Detection: AI systems monitor RT-qPCR reactions in real time to detect anomalies, contamination signals, or irregular amplification patterns, helping laboratories maintain quality assurance standards.
- Genomic Data Integration: AI technologies integrate RT-qPCR outputs with genomic and transcriptomic datasets, enabling deeper insights into gene expression and molecular pathways.
- Rapid Pathogen Identification: AI-assisted diagnostic platforms can quickly analyze RT-qPCR signals to identify pathogens and variants, improving infectious disease surveillance and outbreak response.
- Drug Development Support: Pharmaceutical companies use AI to analyze RT-qPCR gene expression data in drug discovery studies, helping identify therapeutic targets and evaluate treatment effectiveness.
- High-Throughput Screening Efficiency: AI-based analysis tools enable large-scale processing of RT-qPCR data generated in research laboratories, accelerating experimental workflows and reducing analysis time.
Research Scope and Analysis
By Detection Technology Analysis
Probe-based detection dominates the One Step RT-qPCR Kits market due to its high specificity and sensitivity in detecting target RNA sequences. This method utilizes fluorescent probes such as TaqMan probes, molecular beacons, and scorpion probes to bind specifically to the amplified target DNA during PCR cycles. These probes generate fluorescence signals only when hybridized with the correct sequence, which significantly reduces the chances of non-specific amplification. Because of its high accuracy, probe-based detection is widely used in clinical diagnostics, infectious disease testing, and mutation analysis. Hospitals, diagnostic laboratories, and biotechnology companies prefer probe-based RT-qPCR kits for reliable pathogen detection and gene expression analysis. Continuous advancements in probe chemistry and multiplex detection capabilities further strengthen the adoption of this technology. In 2026, probe-based detection is expected to hold approximately 62.4% of the global One Step RT-qPCR Kits market due to its superior diagnostic performance.
Dye-based detection is the fastest-growing segment in the detection technology category because of its cost-effectiveness and simplified assay design. This method relies on fluorescent dyes such as SYBR Green and EvaGreen that bind to double-stranded DNA produced during PCR amplification. Unlike probe-based systems, dye-based assays do not require sequence-specific probes, which reduces reagent costs and simplifies assay development. These kits are widely used in academic research, gene expression studies, and early-stage experimental analysis. Growing research activities in molecular biology laboratories and universities are driving demand for dye-based RT-qPCR kits. Although dye-based detection has slightly lower specificity compared to probe-based methods, improvements in reaction chemistry and melting curve analysis are improving reliability, supporting rapid adoption in research applications.
By Reaction Size Analysis
The 100 reactions segment dominates the reaction size category because it provides a balanced combination of cost efficiency and laboratory convenience. Medium-scale research laboratories, diagnostic centers, and academic institutions frequently prefer this format because it supports multiple experiments without requiring frequent reagent purchases. This reaction size also minimizes reagent wastage while maintaining operational flexibility for routine PCR testing workflows. Many biotechnology companies manufacture RT-qPCR kits in the 100 reaction format as a standard configuration suitable for moderate testing volumes. Additionally, diagnostic laboratories conducting infectious disease screening and gene expression analysis often rely on this format to manage testing throughput efficiently. As molecular testing demand continues to grow globally, laboratories increasingly adopt standardized kit sizes for operational efficiency. In 2026, the 100 reactions segment is projected to hold around 28.7% of the global market share.
The 500 reactions segment is witnessing the fastest growth as high-throughput laboratories and large diagnostic centers expand their testing capacity. Pharmaceutical companies, contract research organizations, and national diagnostic laboratories often require larger kit sizes to support large sample volumes. Bulk reaction kits reduce the cost per test and improve operational efficiency in facilities processing hundreds or thousands of samples. Increasing demand for large-scale infectious disease monitoring and high-throughput genomic studies is accelerating the adoption of larger reaction kits. As molecular diagnostic laboratories continue to scale operations, the demand for 500-reaction and higher capacity kits is expected to increase significantly.
By Application Analysis
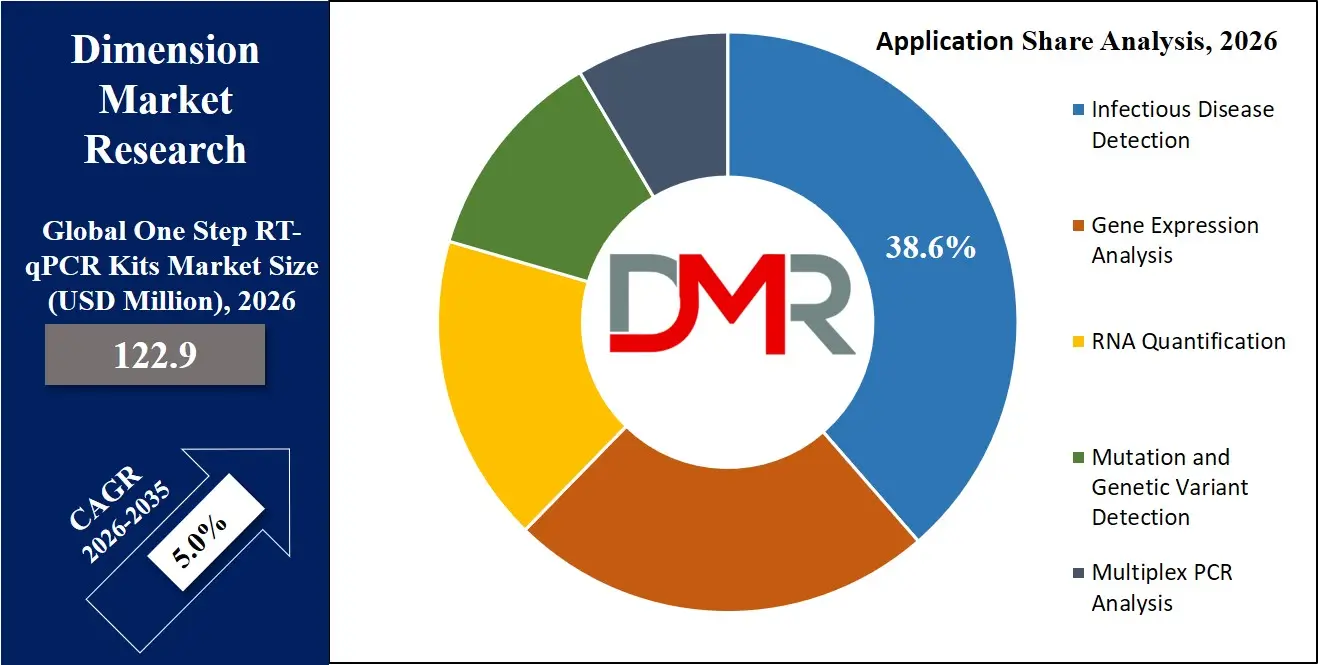
Infectious disease detection represents the dominant application segment in the One Step RT-qPCR Kits market because RT-qPCR remains the gold standard for identifying RNA viruses and other pathogens. Clinical laboratories and public health institutions rely on RT-qPCR testing to detect viral infections, bacterial pathogens, and emerging infectious diseases. The high sensitivity and specificity of RT-qPCR enable early detection of pathogens, which is essential for disease control and patient treatment. Hospitals and diagnostic laboratories frequently utilize these kits for viral detection assays such as influenza, respiratory infections, and other RNA-based pathogens. Growing global emphasis on pandemic preparedness and infectious disease surveillance is further strengthening demand for RT-qPCR testing technologies. In 2026, infectious disease detection is expected to account for approximately 38.6% of the global One Step RT-qPCR Kits market due to its critical role in clinical diagnostics and epidemiological monitoring.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Gene expression analysis is the fastest-growing application segment because researchers increasingly rely on RT-qPCR technologies to study gene regulation, cellular signaling pathways, and molecular responses. Academic research institutes, biotechnology companies, and pharmaceutical organizations utilize RT-qPCR to measure gene transcription levels and analyze biological processes. The expansion of genomic research programs and personalized medicine initiatives is driving demand for advanced gene expression analysis tools. Researchers studying cancer biology, immunology, and metabolic disorders frequently employ RT-qPCR kits to evaluate gene expression patterns. As global research funding continues to increase, the demand for RT-qPCR kits in gene expression studies is expected to grow rapidly.
By End User Analysis
Hospitals and diagnostic laboratories represent the largest end-user segment in the One Step RT-qPCR Kits market due to their critical role in clinical diagnostics and disease detection. These healthcare facilities conduct routine molecular testing for infectious diseases, genetic disorders, and viral infections using RT-qPCR technologies. The demand for rapid and reliable diagnostic testing has increased significantly with the expansion of molecular diagnostic laboratories worldwide. Hospitals prefer one-step RT-qPCR kits because they simplify workflows, reduce contamination risks, and deliver faster results compared to multi-step PCR methods. Continuous expansion of hospital diagnostic infrastructure and increasing investments in molecular testing technologies are strengthening this segment's dominance. In 2026, hospitals and diagnostic laboratories are expected to account for approximately 41.8% of the global market share.
Pharmaceutical and biotechnology companies represent the fastest-growing end-user segment as they increasingly adopt RT-qPCR kits for drug discovery, genetic research, and molecular biology experiments. These organizations use RT-qPCR to analyze gene expression changes, validate drug targets, and study disease mechanisms. Increasing investment in biotechnology research and pharmaceutical innovation is expanding the use of molecular testing technologies across research laboratories. The growing focus on personalized medicine, genomic therapies, and biologics development further increases demand for advanced PCR-based research tools.
The One Step RT-qPCR Kits Market Report is segmented on the basis of the following:
By Detection Technology
- Probe-Based Detection
- TaqMan Probes
- Molecular Beacon Probes
- Scorpion Probes
- Dye-Based Detection
By Reaction Size
- 25 Reactions
- 50 Reactions
- 100 Reactions
- 200 Reactions
- 500 Reactions
- 1000 Reactions
- Above 1000 Reactions
By Application
- Infectious Disease Detection
- Viral Detection
- Bacterial Detection
- Gene Expression Analysis
- RNA Quantification
- Mutation and Genetic Variant Detection
- Multiplex PCR Analysis
By End User
- Hospitals and Diagnostic Laboratories
- Academic and Research Institutes
- Pharmaceutical and Biotechnology Companies
- Contract Research Organizations (CROs)
Regional Analysis
Leading Region in the One Step RT-qPCR Kits Market
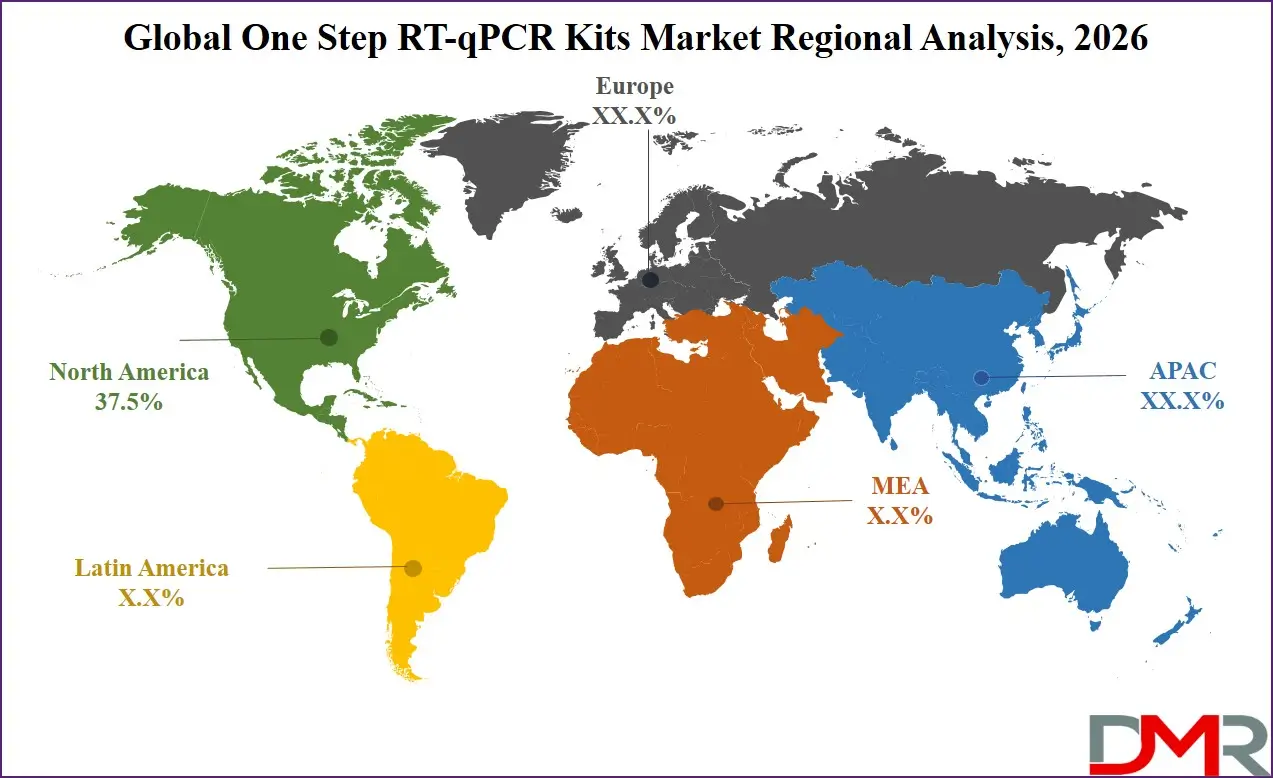
North America holds the dominant position in the global One Step RT-qPCR Kits market due to its advanced biotechnology industry, strong research infrastructure, and high healthcare expenditure. The region hosts numerous biotechnology firms, pharmaceutical companies, and academic research institutions that rely heavily on molecular diagnostic technologies. Significant government funding for genomic research and precision medicine initiatives also contributes to market expansion. Additionally, the presence of well-established diagnostic laboratories and advanced healthcare systems encourages the adoption of high-precision PCR technologies. The United States remains the primary contributor to regional growth because of its strong life sciences research ecosystem and biotechnology investments. Continuous innovation in molecular diagnostic reagents and automated laboratory technologies further strengthens regional market leadership. In 2026, North America is expected to account for approximately 37.5% of the global One Step RT-qPCR Kits market share, maintaining its position as the leading region.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Fastest Growing Region in the One Step RT-qPCR Kits Market
Asia-Pacific is projected to be the fastest-growing region in the One Step RT-qPCR Kits market due to rapid expansion of biotechnology research, healthcare infrastructure development, and increasing government investments in life sciences. Countries such as China, India, South Korea, and Japan are significantly strengthening their molecular diagnostic capabilities. Growing pharmaceutical manufacturing activities and rising research funding in genomics and biotechnology are supporting increased adoption of RT-qPCR technologies. Additionally, the region's expanding diagnostic laboratory networks and growing focus on infectious disease monitoring contribute to market growth. Increasing collaboration between academic institutions and biotechnology companies is further accelerating innovation and adoption of molecular testing technologies across the Asia-Pacific region.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The One Step RT-qPCR Kits market is characterized by intense competition driven by technological innovation, research partnerships, and continuous product development. Market participants focus on improving enzyme efficiency, reaction sensitivity, and multiplex detection capabilities to strengthen their competitive positioning. Companies invest heavily in research and development to create advanced reagent formulations that deliver higher accuracy and faster amplification cycles. Strategic collaborations with academic institutions, pharmaceutical companies, and diagnostic laboratories help manufacturers expand their research networks and product applications. Additionally, firms are investing in automated PCR solutions and integrated diagnostic platforms to meet the growing demand for high-throughput molecular testing. Strong distribution networks, regulatory compliance capabilities, and technical support services also play important roles in maintaining market competitiveness.
Some of the prominent players in the global One Step RT-qPCR Kits are:
- Thermo Fisher Scientific Inc.
- QIAGEN
- Takara Bio Inc.
- Bio-Rad Laboratories, Inc.
- Promega Corporation
- New England Biolabs
- Merck KGaA
- Roche Diagnostics
- Agilent Technologies, Inc.
- Avantor, Inc.
- Fortis Life Sciences
- Quantabio
- MP Biomedicals
- Zymo Research Corporation
- Solis BioDyne
- Applied Biological Materials Inc.
- TIANGEN Biotech (Beijing) Co., Ltd.
- EnzyQuest
- Canvax
- NZYtech
- Other Key Players
Recent Developments
- In May 2025, QIAGEN announced the expansion of its molecular diagnostics portfolio through the introduction of a new One Step RT-qPCR kit optimized for gene expression analysis and infectious disease detection. The kit includes advanced probe chemistries designed to improve assay sensitivity and reduce background fluorescence signals. The product is intended for research laboratories, pharmaceutical companies, and diagnostic testing facilities performing high-precision RNA detection. QIAGEN stated that the new solution integrates seamlessly with automated PCR workflows and supports high-throughput testing environments.
- In February 2025, Thermo Fisher Scientific introduced an advanced One Step RT-qPCR kit designed for high-sensitivity RNA detection in clinical and research laboratories. The newly launched kit features improved enzyme stability and optimized reaction buffers that enhance amplification efficiency and reduce reaction time. The product also supports multiplex PCR assays, enabling laboratories to detect multiple RNA targets in a single reaction. According to the company, the new kit is compatible with a wide range of real-time PCR instruments and automated laboratory systems.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 122.9 Mn |
| Forecast Value (2035) |
USD 190.4 Mn |
| CAGR (2026–2035) |
5.0% |
| The US Market Size (2026) |
USD 40.0 Mn |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Detection Technology (Probe-Based Detection, Dye-Based Detection), By Reaction Size (25 Reactions, 50 Reactions, 100 Reactions, 200 Reactions, 500 Reactions, 1000 Reactions, Above 1000 Reactions), By Application (Infectious Disease Detection, Gene Expression Analysis, RNA Quantification, Mutation and Genetic Variant Detection, Multiplex PCR Analysis), By End User (Hospitals and Diagnostic Laboratories, Academic and Research Institutes, Pharmaceutical and Biotechnology Companies, Contract Research Organizations (CROs)) |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
| Prominent Players |
Thermo Fisher Scientific Inc., QIAGEN, Takara Bio Inc., Bio-Rad Laboratories, Inc., Promega Corporation, New England Biolabs, Merck KGaA, Roche Diagnostics, Agilent Technologies, Inc., Avantor, Inc., Fortis Life Sciences, Quantabio, MP Biomedicals, Zymo Research Corporation, Solis BioDyne, Applied Biological Materials Inc., TIANGEN Biotech (Beijing) Co., Ltd., EnzyQuest, Canvax, NZYtech, and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Global One Step RT-qPCR Kits Market?
▾ The Global One Step RT-qPCR Kits Market size is expected to reach USD 122.9 million by 2026 and is projected to reach USD 190.4 million by the end of 2035.
Which region accounted for the largest Global One Step RT-qPCR Kits Market?
▾ North America is expected to have the largest market share in the Global One Step RT-qPCR Kits Market, with a share of about 37.5% in 2026.
How big is the One Step RT-qPCR Kits Market in the US?
▾ The US One Step RT-qPCR Kits market is expected to reach USD 40.0 million by 2026.
Who are the key players in the One Step RT-qPCR Kits Market?
▾ Some of the major key players in the Global One Step RT-qPCR Kits Market include Merck, Qiagen, Bio-Rad, and others.
What is the growth rate in the Global One Step RT-qPCR Kits Market?
▾ The market is growing at a CAGR of 5.0 percent over the forecasted period.