Market Overview
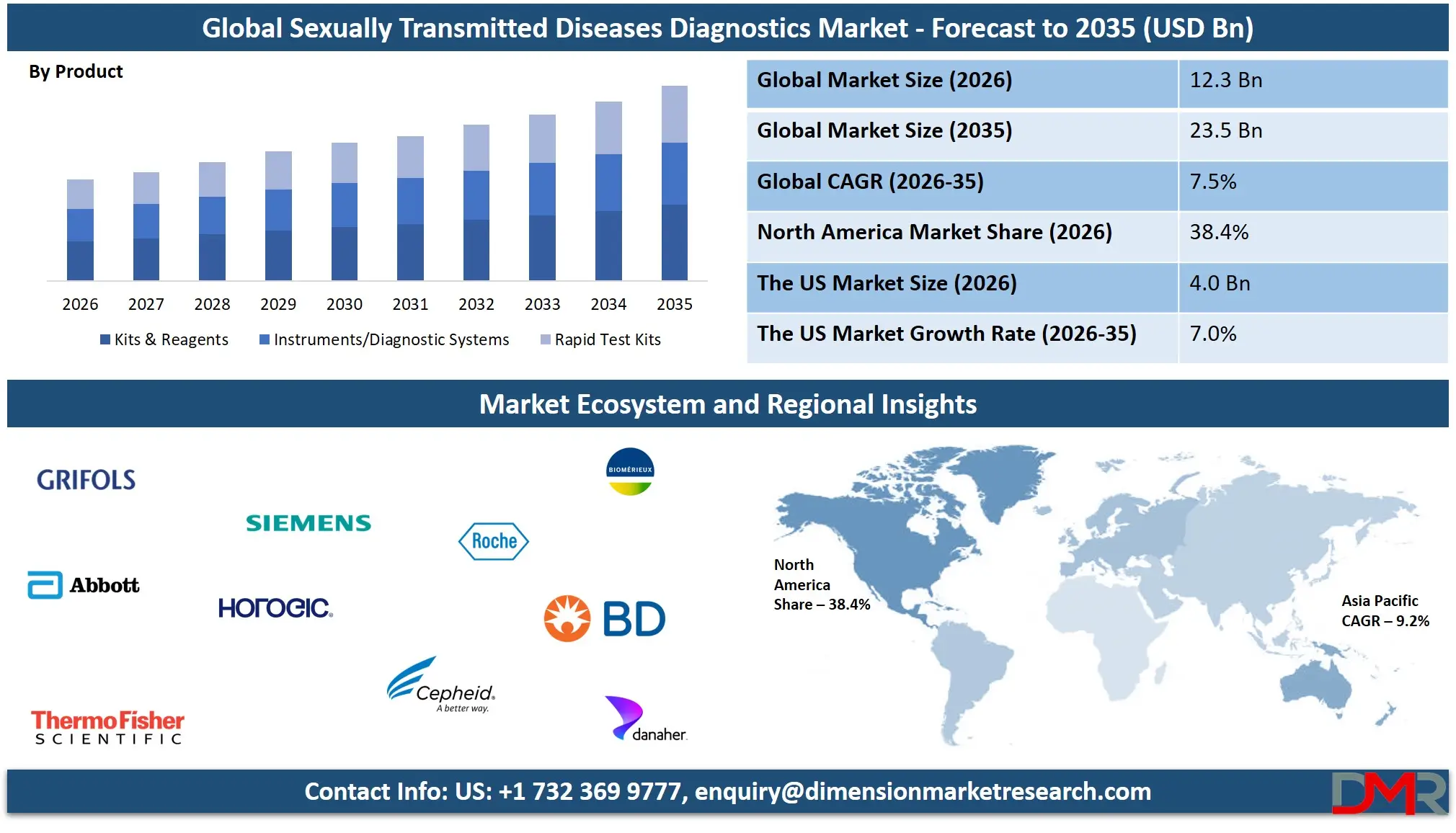
The Global Sexually Transmitted Diseases Diagnostics Market size is projected to reach USD 12.3 billion in 2026 and grow at a compound annual growth rate of 7.5% to reach a value of USD 23.5 billion in 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Sexually Transmitted Diseases Diagnostics refers to a range of medical technologies, laboratory methods, and testing services designed to detect infections transmitted primarily through sexual contact. These diagnostic solutions identify pathogens such as bacteria, viruses, and parasites responsible for conditions including chlamydia, gonorrhea, syphilis, human immunodeficiency virus, and human papillomavirus. The field encompasses laboratory-based molecular diagnostics, immunoassays, rapid point-of-care tests, and culture-based techniques used in hospitals, diagnostic laboratories, community health centers, and home-based testing environments. The primary objective is early detection, accurate diagnosis, and timely treatment to prevent disease progression and transmission.
The importance of STD diagnostic technologies continues to grow as global healthcare systems emphasize early screening, disease surveillance, and preventive healthcare. Advances in molecular biology, particularly nucleic acid amplification technologies and next-generation sequencing, are significantly improving test sensitivity and turnaround time. Growing awareness regarding sexual health, expanded screening programs, and the increasing availability of self-testing solutions are reshaping diagnostic approaches. The integration of digital health platforms and telemedicine services is also facilitating wider accessibility to testing and follow-up care.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Healthcare organizations and diagnostic companies are focusing heavily on innovation, strategic partnerships, and regulatory approvals to expand testing capabilities. Significant investments are being directed toward rapid diagnostics and multiplex testing platforms capable of detecting multiple infections simultaneously. Additionally, governments and public health institutions are strengthening screening initiatives to address the rising prevalence of sexually transmitted infections globally. These developments are accelerating the adoption of advanced diagnostic solutions across both developed and emerging healthcare systems.
The US Sexually Transmitted Diseases Diagnostics Market
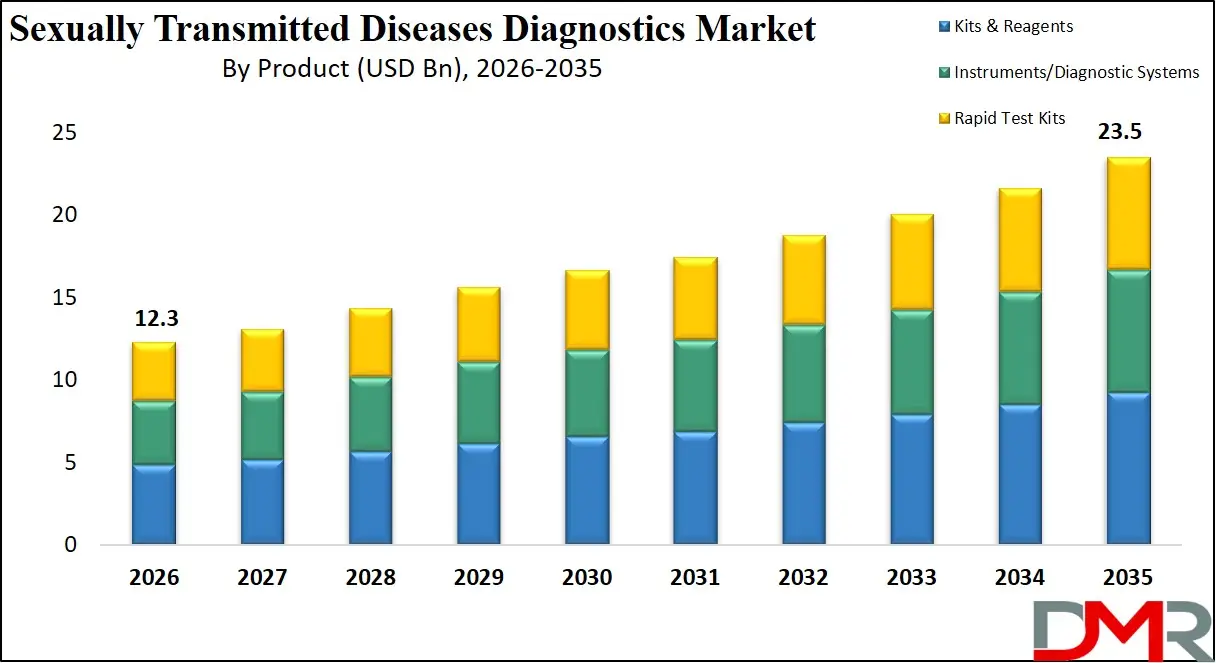
The US Sexually Transmitted Diseases Diagnostics Market size is projected to reach USD 4.0 billion in 2026 at a compound annual growth rate of 7.0% over its forecast period.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US represents one of the most advanced markets for sexually transmitted disease diagnostics due to strong healthcare infrastructure, widespread screening programs, and high awareness of sexual health. Public health initiatives supported by federal agencies encourage routine screening for infections such as chlamydia, gonorrhea, and HIV. Diagnostic laboratories and healthcare providers increasingly rely on molecular diagnostic technologies that offer faster and more accurate results. The country also has a strong presence of biotechnology firms and diagnostic developers investing in research and development for innovative testing solutions. Additionally, government-supported preventive programs, expanding insurance coverage for STI screening, and the growing availability of home-based testing kits are further supporting the growth and modernization of STD diagnostic services.
Europe Sexually Transmitted Diseases Diagnostics Market
Europe Sexually Transmitted Diseases Diagnostics Market size is projected to reach USD 2.8 billion in 2026 at a compound annual growth rate of 6.9% over its forecast period.
Europe demonstrates steady expansion in sexually transmitted disease diagnostic services, driven by strong public health systems and strict regulatory frameworks that ensure high-quality diagnostic standards. Regional initiatives focused on disease prevention and early screening encourage healthcare providers to adopt advanced diagnostic technologies. Several European countries are increasing investments in molecular diagnostics and point-of-care testing to improve detection rates and reduce the spread of infections. Public health campaigns promoting sexual health awareness are also contributing to higher screening volumes. Research institutions and biotechnology companies across the region are actively collaborating to develop innovative diagnostic tools. In addition, digital healthcare integration and community-based screening programs are improving access to testing, particularly among high-risk populations and underserved communities.
Japan Sexually Transmitted Diseases Diagnostics Market
Japan Sexually Transmitted Diseases Diagnostics Market size is projected to reach USD 738.0 million in 2026 at a compound annual growth rate of 7.2% over its forecast period.
Japan's sexually transmitted diseases diagnostics sector is evolving through technological innovation and improvements in healthcare delivery systems. Advanced laboratory infrastructure and strong clinical research capabilities enable the adoption of sophisticated molecular diagnostic technologies. The country has been increasingly emphasizing early detection and preventive screening programs for sexually transmitted infections, particularly among younger populations and urban communities. Government-backed healthcare initiatives promote regular health check-ups and expanded diagnostic accessibility. Diagnostic laboratories and hospitals are integrating automated platforms and high-throughput testing systems to enhance efficiency and accuracy. Additionally, rising awareness regarding sexual health and the growing demand for confidential testing services are encouraging the development of discreet home-testing options and digital healthcare platforms that support diagnosis and consultation.
Sexually Transmitted Diseases Diagnostics Market: Key Takeaways
- Market Growth: The Sexually Transmitted Diseases Diagnostics Market size is expected to grow by USD 10.4 billion, at a CAGR of 7.5% during the forecasted period of 2027 to 2035.
- By Product: The kits & reagents segment is anticipated to get the majority share of the Sexually Transmitted Diseases Diagnostics market in 2026.
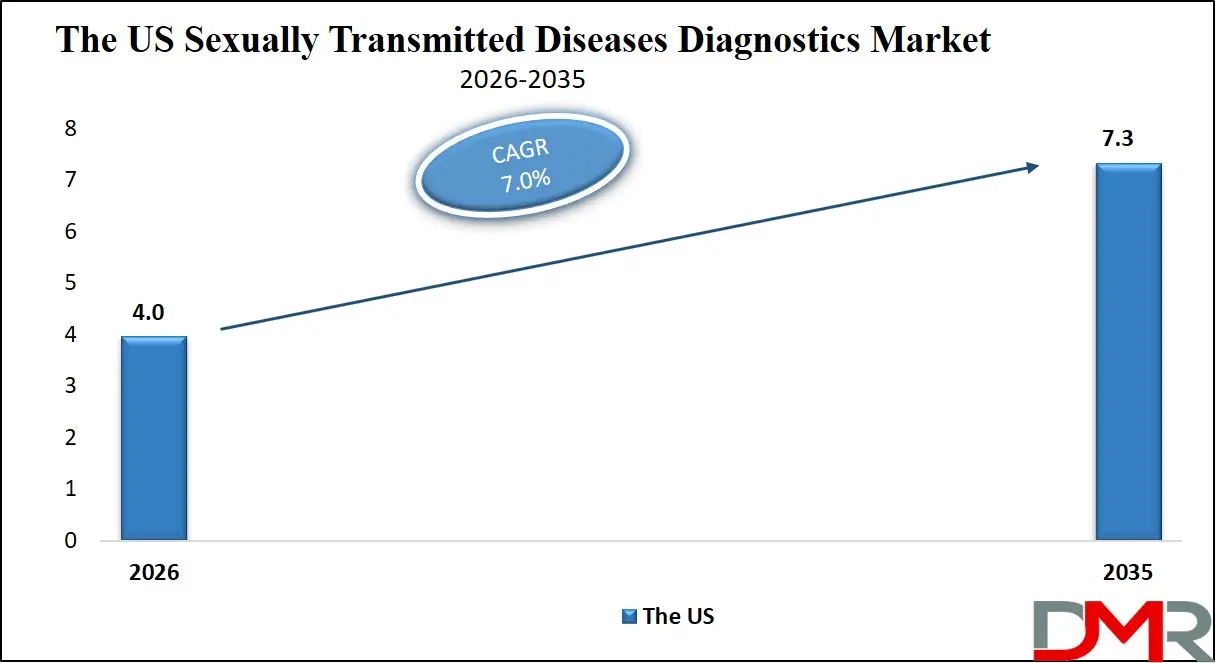
- By Testing Location: The laboratory testing segment is expected to get the largest revenue share in 2026 in the Sexually Transmitted Diseases Diagnostics market.
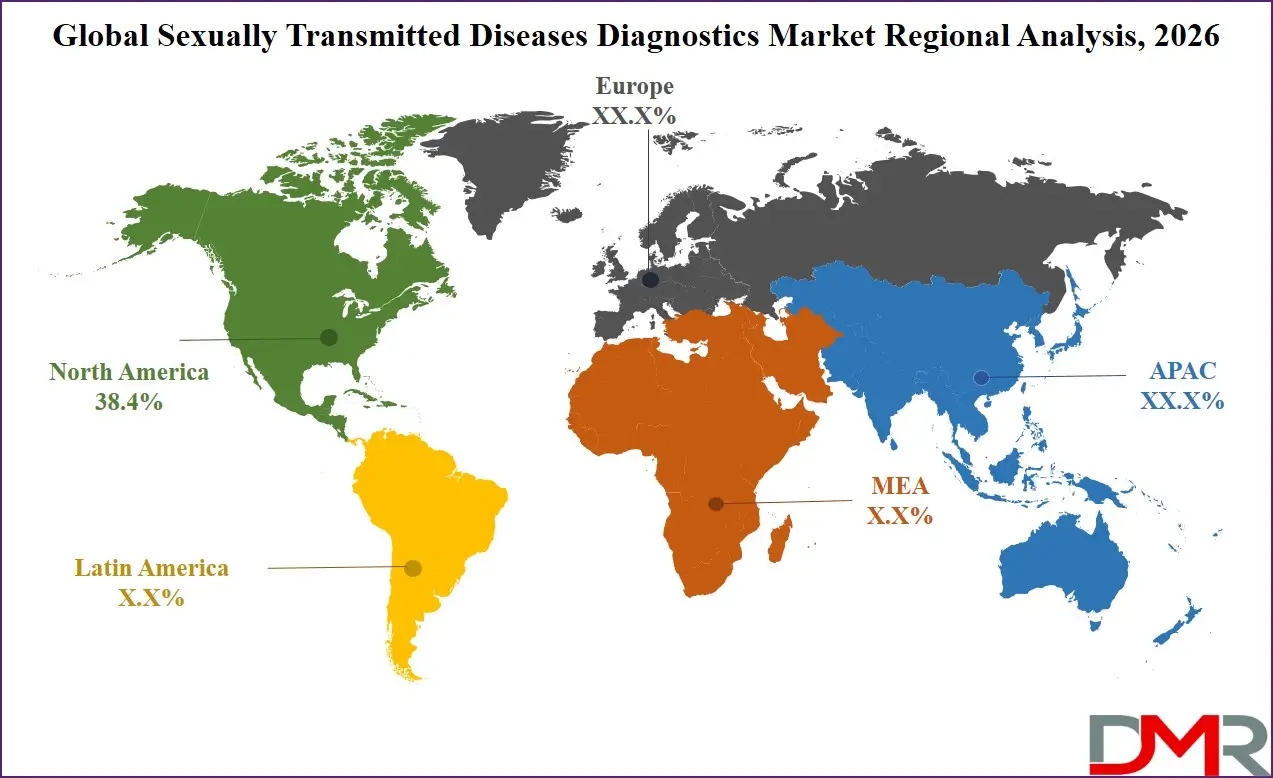
- Regional Insight: North America is expected to hold a 38.4% share of revenue in the global Sexually Transmitted Diseases Diagnostics market in 2026.
- Use Cases: Some of the use cases of Sexually Transmitted Diseases Diagnostics include prenatal infection screening, rapid emergency testing, and more.
Sexually Transmitted Diseases Diagnostics Market: Use Cases:
- Routine Screening Programs: Healthcare providers use STD diagnostics for routine screening among sexually active individuals, particularly in high-risk populations. Early identification helps prevent transmission and enables timely treatment, reducing the long-term health impact of infections.
- Prenatal Infection Screening: Pregnant women undergo diagnostic testing for infections such as syphilis, HIV, and chlamydia. Early diagnosis allows healthcare providers to initiate treatments that significantly reduce the risk of mother-to-child transmission during pregnancy or childbirth.
- Rapid Emergency Testing: Point-of-care diagnostic tools are used in emergency departments and clinics where immediate detection is required. These tests provide quick results, enabling physicians to start treatment during the same clinical visit.
- Public Health Surveillance: Government health agencies utilize diagnostic testing data to track infection trends and outbreaks. These insights support epidemiological monitoring, prevention strategies, and resource allocation for disease control programs.
- Home-Based Self-Testing: Self-testing kits allow individuals to collect samples privately at home and send them to laboratories or obtain rapid results. This improves accessibility and encourages testing among individuals reluctant to visit healthcare facilities.
- Community Health Clinics: Community clinics rely on cost-effective and rapid diagnostics to screen large populations, particularly in underserved areas. These programs help increase early detection rates and improve treatment accessibility.
- Multiplex Infection Detection: Advanced molecular diagnostic platforms can detect multiple pathogens from a single sample. This approach enhances diagnostic efficiency and helps clinicians identify co-infections that require specialized treatment plans.
Stats & Facts
- World Health Organization (WHO) reported in 2024 that more than 1 million sexually transmitted infections are acquired every day worldwide.
- World Health Organization (WHO) estimated in 2024 that 374 million new infections annually occur with one of four STIs: chlamydia, gonorrhea, syphilis, and trichomoniasis.
- Centers for Disease Control and Prevention (CDC) reported in 2024 that the United States recorded over 2.5 million cases of chlamydia, gonorrhea, and syphilis combined.
- Centers for Disease Control and Prevention (CDC) stated in 2024 that chlamydia accounted for more than 1.6 million reported infections in the United States.
- UNAIDS reported in 2024 that 39 million people globally were living with HIV.
- European Centre for Disease Prevention and Control (ECDC) reported in 2024 that chlamydia remained the most frequently reported STI in Europe with over 230,000 cases annually.
- UK Health Security Agency (UKHSA) reported in 2024 that over 400,000 STI diagnoses were recorded in England.
- National Institute of Infectious Diseases Japan reported in 2024 that syphilis infections in Japan exceeded 13,000 reported cases.
- UNAIDS reported in 2025 that 1.3 million new HIV infections occurred globally in 2024.
- World Health Organization (WHO) stated in 2025 that HPV is responsible for nearly 95% of cervical cancer cases worldwide.
- Centers for Disease Control and Prevention (CDC) reported in 2024 that about 48% of new STI infections in the US occur among individuals aged 15–24 years.
- European Centre for Disease Prevention and Control (ECDC) reported in 2025 that gonorrhea cases in Europe increased by more than 30% compared to previous reporting years.
Market Dynamic
Driving Factors in the Sexually Transmitted Diseases Diagnostics Market
Rising Global Prevalence of Sexually Transmitted Infections
The increasing global incidence of sexually transmitted infections is a major factor driving the demand for advanced diagnostic technologies. Rising infection rates are encouraging healthcare systems to strengthen screening programs and expand access to diagnostic testing. Public health authorities are emphasizing early detection to prevent complications such as infertility, cancer, and chronic infections. Increased sexual health awareness campaigns and improved healthcare access are also encouraging more individuals to undergo routine testing. As a result, healthcare providers are adopting advanced diagnostic platforms capable of detecting multiple pathogens quickly and accurately, contributing significantly to the expansion of STD diagnostic services worldwide.
Technological Advancements in Diagnostic Testing
Continuous innovation in diagnostic technologies is significantly improving the efficiency and reliability of STD detection. Modern molecular diagnostics such as polymerase chain reaction and nucleic acid amplification techniques offer highly sensitive detection even in early infection stages. Automated laboratory systems and multiplex testing platforms allow simultaneous detection of multiple infections from a single sample, reducing diagnostic time and operational costs. Additionally, portable and rapid point-of-care testing solutions are enabling immediate diagnosis in clinics and community healthcare settings. These technological improvements are enhancing diagnostic accuracy, expanding accessibility, and encouraging widespread adoption of advanced STD testing solutions.
Restraints in the Sexually Transmitted Diseases Diagnostics Market
Social Stigma and Limited Awareness
Social stigma associated with sexually transmitted infections remains a significant barrier to widespread diagnostic adoption. Many individuals hesitate to seek testing due to concerns about privacy, discrimination, or social judgment. This reluctance often delays diagnosis and treatment, allowing infections to spread further within communities. In several regions, limited awareness about sexual health and preventive screening programs also contributes to underdiagnosis. Healthcare providers and public health organizations are attempting to address these issues through education campaigns, confidential testing options, and telehealth services. However, cultural and societal barriers continue to slow the full adoption of STD diagnostic services in many parts of the world.
Limited Healthcare Infrastructure in Developing Regions
In many developing countries, insufficient healthcare infrastructure restricts access to advanced STD diagnostic technologies. Laboratories may lack modern testing equipment, trained personnel, or reliable supply chains for diagnostic reagents. This leads to delayed diagnoses or reliance on outdated testing methods that may not provide accurate results. Additionally, financial constraints often limit government investment in comprehensive screening programs. Rural and remote communities are particularly affected by these limitations. While international health organizations and governments are working to strengthen diagnostic capacity through funding and training initiatives, infrastructure gaps remain a major challenge for the expansion of STD diagnostics globally.
Opportunities in the Sexually Transmitted Diseases Diagnostics Market
Expansion of Home-Based and Self-Testing Solutions
The growing demand for privacy and convenience in healthcare services is creating significant opportunities for home-based STD testing solutions. Self-testing kits allow individuals to collect samples in a private environment and receive results either through digital platforms or laboratory analysis. These solutions are particularly attractive for younger populations and individuals reluctant to visit clinics due to social stigma. Advances in diagnostic technologies are enabling the development of reliable self-testing kits with high sensitivity and accuracy. As telemedicine services expand globally, integration with digital consultation platforms is expected to further enhance the adoption of home-based STD diagnostic solutions.
Integration of Digital Health and Telemedicine
Digital health technologies and telemedicine platforms are opening new possibilities for improving access to STD diagnostics and treatment. Online healthcare services allow individuals to consult healthcare professionals, order diagnostic kits, and receive treatment recommendations remotely. This model is especially beneficial for populations in rural areas or regions with limited healthcare infrastructure. Mobile health applications are also being used for appointment scheduling, test reminders, and confidential result delivery. As healthcare systems increasingly adopt digital technologies, these integrated platforms are expected to streamline diagnostic workflows and enhance patient engagement in STD screening programs.
Trends in the Sexually Transmitted Diseases Diagnostics Market
Shift Toward Rapid Point-of-Care Testing
One of the most significant trends in the STD diagnostics sector is the growing adoption of rapid point-of-care testing solutions. These tests allow healthcare providers to detect infections within minutes, enabling immediate treatment during a single clinical visit. This approach improves patient compliance, reduces follow-up visits, and minimizes the risk of disease transmission. Rapid diagnostic tests are particularly valuable in emergency departments, community health clinics, and mobile healthcare programs where immediate results are critical. Continuous improvements in test sensitivity and portability are expected to further accelerate the adoption of point-of-care diagnostic technologies.
Growth of Multiplex Diagnostic Platforms
Multiplex diagnostic technologies are becoming increasingly popular due to their ability to detect multiple sexually transmitted infections simultaneously. Instead of performing separate tests for each infection, multiplex platforms analyze a single sample for multiple pathogens. This approach significantly improves laboratory efficiency and reduces diagnostic turnaround time. Healthcare providers are increasingly adopting these platforms to manage co-infections and complex cases more effectively. Furthermore, multiplex systems help optimize laboratory workflows and reduce operational costs. As diagnostic technology continues to advance, multiplex testing platforms are expected to become a standard tool in STD diagnostic laboratories.
Impact of Artificial Intelligence in Sexually Transmitted Diseases Diagnostics Market
- AI-Assisted Diagnostic Interpretation: Artificial intelligence algorithms analyze complex laboratory data and molecular test results to support faster and more accurate diagnosis of sexually transmitted infections.
- Predictive Epidemiology Models: AI systems analyze epidemiological data to predict infection trends and identify high-risk populations, enabling targeted screening programs.
- Automated Laboratory Workflow Optimization: AI-driven automation helps laboratories streamline sample processing, reduce errors, and improve overall diagnostic efficiency.
- Digital Symptom Assessment Tools: AI-powered health applications help individuals evaluate symptoms and determine whether STD testing may be required.
- Personalized Treatment Recommendations: AI platforms analyze patient data and infection patterns to support clinicians in selecting appropriate treatment strategies.
- Population Health Surveillance: AI tools assist public health agencies in tracking infection outbreaks and monitoring disease spread through large-scale data analysis.
- AI-Enhanced Imaging Diagnostics: Machine learning algorithms can assist in identifying infection indicators through microscopic imaging and clinical data analysis.
- Remote Patient Monitoring: AI-enabled digital health platforms allow healthcare providers to monitor patient progress and ensure adherence to treatment after diagnosis.
Research Scope and Analysis
By Disease Type Analysis
Chlamydia is projected to remain the leading disease segment within sexually transmitted diseases diagnostics due to its high global prevalence and strong screening recommendations across healthcare systems. In 2026, this segment is expected to account for approximately 27.8% of the overall diagnostics share. Chlamydia infections are often asymptomatic, which makes routine diagnostic testing essential for early detection and prevention of complications such as infertility and pelvic inflammatory disease. Healthcare authorities in many countries recommend regular screening for sexually active individuals, particularly women under the age of 25, contributing significantly to testing demand. Advances in molecular diagnostic methods, particularly nucleic acid amplification tests, have greatly improved detection accuracy and reduced turnaround times. Additionally, public health programs, expanded sexual health awareness campaigns, and increased accessibility of point-of-care testing solutions are driving diagnostic adoption. High screening rates in hospitals, clinics, and community health centers further reinforce the dominance of the chlamydia diagnostic segment across global healthcare systems.
Human Papillomavirus diagnostics are expected to represent the fastest-growing segment due to increasing awareness regarding HPV-related cancers and expanded screening programs globally. HPV testing is being increasingly integrated into cervical cancer screening initiatives and national immunization programs. The growing adoption of molecular testing technologies capable of detecting high-risk HPV strains is further accelerating the expansion of this segment. Healthcare organizations are emphasizing early HPV detection as part of preventive healthcare strategies. Additionally, the development of advanced HPV genotyping assays and improved laboratory screening workflows is supporting greater testing accuracy. Increasing female health awareness programs and rising investments in women's healthcare infrastructure are also contributing to the rapid expansion of HPV diagnostics worldwide.
By Test Type Analysis
Molecular diagnostics are expected to dominate the STD diagnostics market, accounting for approximately 44.6% share in 2026. These diagnostic techniques provide highly sensitive and specific detection of pathogens by identifying genetic material such as DNA or RNA from infectious agents. Technologies including polymerase chain reaction, nucleic acid amplification tests, and next-generation sequencing are widely used in clinical laboratories due to their accuracy and reliability. Molecular testing is particularly effective in detecting infections during early stages when pathogen concentrations may be low. Laboratories increasingly prefer these methods for their rapid turnaround times and ability to support multiplex testing. Furthermore, advancements in automated molecular diagnostic platforms are improving testing throughput and reducing laboratory workloads. The expansion of infectious disease screening programs and the increasing demand for high-precision diagnostics continue to reinforce the dominance of molecular testing in STD detection.
Rapid or point-of-care testing is emerging as the fastest-growing diagnostic segment due to the increasing need for immediate results and improved patient accessibility. These tests are designed to deliver diagnostic outcomes within minutes, allowing healthcare providers to initiate treatment during the same clinical visit. Rapid diagnostic solutions are especially valuable in community health clinics, emergency departments, and remote healthcare settings where laboratory infrastructure may be limited. Advances in lateral flow technology and portable diagnostic devices are improving the reliability and convenience of these tests. Additionally, growing demand for decentralized healthcare services and expanded screening programs among high-risk populations are contributing to the rapid adoption of point-of-care STD diagnostics.
By Product Analysis
Kits and reagents represent the largest product segment within the sexually transmitted diseases diagnostics market and are expected to hold approximately 39.2% share by 2026. These components are essential for conducting molecular assays, immunoassays, and rapid diagnostic tests across laboratories and clinical settings. Diagnostic kits contain the necessary reagents and consumables required for detecting pathogens responsible for sexually transmitted infections. The demand for these products is increasing due to the high volume of STD screening conducted globally. Hospitals, diagnostic laboratories, and public health institutions rely heavily on standardized testing kits to ensure reliable and consistent diagnostic results. Continuous improvements in reagent formulations and assay sensitivity are also contributing to segment growth. Furthermore, rising adoption of automated diagnostic systems and multiplex testing platforms is driving the demand for compatible kits and reagents, reinforcing their dominant position in the product category.
Rapid test kits are experiencing significant growth due to increasing demand for convenient, quick, and accessible diagnostic solutions. These kits enable healthcare providers and individuals to detect infections without requiring complex laboratory equipment. Rapid test kits are widely used in point-of-care settings, community health programs, and mobile healthcare units. Their portability and ease of use make them highly suitable for large-scale screening initiatives, particularly in resource-limited regions. Technological improvements have enhanced their sensitivity and reliability, making them increasingly comparable to laboratory-based tests. Growing consumer interest in confidential testing and expanding home-testing solutions are also contributing to the rapid expansion of this segment.
By Testing Location Analysis
Laboratory testing continues to dominate the STD diagnostics landscape, accounting for approximately 46.7% of the market share in 2026. Clinical laboratories possess the advanced infrastructure and specialized equipment required for highly accurate diagnostic procedures, including molecular assays and complex immunoassays. These laboratories process large volumes of samples from hospitals, clinics, and public health programs. Laboratory-based testing offers higher diagnostic precision, enabling healthcare providers to confirm infections and identify co-infections effectively. In addition, laboratories often utilize automated platforms capable of performing high-throughput testing, which significantly improves efficiency. Government screening programs and hospital-based diagnostic services further contribute to the strong demand for laboratory testing. The continued expansion of reference laboratories and improvements in diagnostic technologies are expected to maintain the leadership of this segment.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Home or self-testing is rapidly gaining popularity as individuals seek greater privacy, convenience, and control over their healthcare decisions. Self-testing kits allow users to collect biological samples at home and either receive rapid results or send samples to certified laboratories for analysis. The increasing acceptance of telemedicine services has also contributed to the growth of this segment, as patients can consult healthcare professionals online after receiving test results. Rising awareness regarding sexual health and increasing consumer preference for confidential testing solutions are key drivers supporting the expansion of home-based diagnostics. Technological improvements in self-testing kits are enhancing reliability and encouraging wider adoption among younger and digitally connected populations.
By End User Analysis
Diagnostic laboratories represent the largest end-user segment in the STD diagnostics market and are expected to hold approximately 35.9% market share in 2026. These laboratories perform large volumes of specialized diagnostic tests using advanced molecular technologies and automated systems. Public health agencies, hospitals, and clinics often rely on centralized diagnostic laboratories for accurate detection and confirmation of sexually transmitted infections. Laboratories possess trained personnel, advanced equipment, and standardized testing protocols that ensure high-quality diagnostic results. The growing number of screening initiatives and rising infection rates are increasing the demand for laboratory testing services. Additionally, collaborations between laboratories and healthcare providers help improve patient access to diagnostic services and strengthen disease surveillance programs.
Homecare settings are emerging as a rapidly expanding end-user segment as healthcare systems increasingly shift toward patient-centered and decentralized care models. Home-based diagnostic solutions allow individuals to perform tests independently while maintaining privacy and convenience. These testing methods are particularly attractive for populations reluctant to visit healthcare facilities due to stigma or logistical barriers. Advances in self-testing technologies and digital health platforms have made home-based diagnostics more reliable and accessible. The growing popularity of telehealth consultations further supports the expansion of this segment, as individuals can receive professional medical guidance after conducting tests at home.
The Sexually Transmitted Diseases Diagnostics Market Report is segmented on the basis of the following:
By Disease Type
- Chlamydia
- Gonorrhea
- Syphilis
- Human Immunodeficiency Virus (HIV)
- Human Papillomavirus (HPV)
- Herpes Simplex Virus (HSV)
- Trichomoniasis
- Others
By Test Type
- Molecular Diagnostics
- Polymerase Chain Reaction (PCR)
- Nucleic Acid Amplification Tests (NAAT)
- Next-Generation Sequencing (NGS)
- Immunoassays
- Enzyme-Linked Immunosorbent Assay (ELISA)
- Chemiluminescence Immunoassay (CLIA)
- Rapid / Point-of-Care Tests
- Culture & Microscopy
By Product
- Instruments / Diagnostic Systems
- Kits & Reagents
- Rapid Test Kits
By Testing Location
- Laboratory Testing
- Point-of-Care Testing
- Home / Self-Testing
By End User
- Hospitals & Clinics
- Diagnostic Laboratories
- Homecare Settings
- Public Health Laboratories / Community Clinics
Regional Analysis
Leading Region in the Sexually Transmitted Diseases Diagnostics Market
North America is expected to maintain its leadership in the sexually transmitted diseases diagnostics market, accounting for approximately 38.4% share in 2026. The region benefits from a well-established healthcare infrastructure, advanced diagnostic laboratories, and strong public health initiatives focused on disease prevention and early detection. Government agencies actively promote screening programs for infections such as HIV, chlamydia, and gonorrhea.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
High healthcare expenditure and strong investments in diagnostic research and development further support market growth. Additionally, the presence of leading biotechnology and diagnostic companies encourages continuous innovation in testing technologies. Growing awareness regarding sexual health and increasing availability of home-based testing solutions are also contributing to the region's dominant position in the global STD diagnostics landscape.
Fastest Growing Region in the Sexually Transmitted Diseases Diagnostics Market
Asia Pacific is projected to be the fastest-growing region in the sexually transmitted diseases diagnostics market due to rapid healthcare infrastructure development and increasing awareness regarding infectious disease prevention. Governments across several countries are strengthening public health programs aimed at improving access to sexual health services and expanding screening coverage. Rising urban populations, improving healthcare access, and growing investments in diagnostic technologies are accelerating regional market growth. Additionally, international health organizations are supporting initiatives that improve disease surveillance and testing capacity in developing economies. Expanding diagnostic laboratory networks and the increasing adoption of molecular diagnostic platforms are expected to further drive the rapid growth of STD diagnostic services throughout the Asia Pacific region.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The sexually transmitted diseases diagnostics market is characterized by intense competition driven by technological innovation, product differentiation, and strategic collaborations. Companies operating in this sector focus heavily on research and development to introduce advanced diagnostic platforms that offer improved sensitivity, faster results, and multiplex testing capabilities. Market participants are increasingly investing in automation technologies and digital diagnostic solutions to enhance laboratory efficiency and expand testing accessibility. Strategic partnerships with healthcare providers, research institutions, and public health agencies are also common strategies used to strengthen market presence. Additionally, regulatory approvals, patent portfolios, and strong distribution networks play a significant role in maintaining competitive advantages within the global STD diagnostics industry.
Some of the prominent players in the global Sexually Transmitted Diseases Diagnostics are:
- Abbott Laboratories
- F. Hoffmann-La Roche
- Becton, Dickinson and Company (BD)
- Hologic Inc.
- Danaher Corporation
- Cepheid
- Siemens Healthineers
- Thermo Fisher Scientific
- QIAGEN N.V.
- bioMérieux SA
- Bio-Rad Laboratories
- DiaSorin S.p.A.
- QuidelOrtho Corporation
- OraSure Technologies
- Trinity Biotech
- Grifols
- Seegene Inc.
- Sekisui Diagnostics
- Chembio Diagnostics
- bioLytical Laboratories
- Other Key Players
Recent Developments
- In December 2025, Roche received CE Mark approval for its cobas® BV/CV assay, designed to accurately detect bacteria and yeast responsible for bacterial vaginosis and Candida vaginitis in vaginal samples collected using the cobas PCR Media tube. Traditional diagnostic methods such as microscopy, pH testing, and clinical observation may produce inaccurate results due to non-specific symptoms. The assay enables precise detection, helping clinicians deliver targeted treatments faster while improving diagnostic efficiency and reducing the risk of delayed or inappropriate therapy.
- In November 2025, Visby Medical has launched the first FDA-authorized at-home PCR test for sexually transmitted infections in women. The palm-sized test detects chlamydia, gonorrhea, and trichomoniasis within 30 minutes and is available nationwide with virtual care support and home delivery. Designed to address barriers such as cost, time, and privacy concerns, the test expands access to convenient screening as STI cases rise. Early detection helps prevent complications including infertility, chronic pelvic pain, and ectopic pregnancy.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 12.3 Bn |
| Forecast Value (2035) |
USD 23.5 Bn |
| CAGR (2026–2035) |
7.5% |
| The US Market Size (2026) |
USD 4.0 Bn |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Disease Type (Chlamydia, Gonorrhea, Syphilis, Human Immunodeficiency Virus (HIV), Human Papillomavirus (HPV), Herpes Simplex Virus (HSV), Trichomoniasis, Others), By Test Type (Molecular Diagnostics, Immunoassays, Rapid / Point-of-Care Tests, Culture & Microscopy), By Product (Instruments / Diagnostic Systems, Kits & Reagents, Rapid Test Kits), By Testing Location (Laboratory Testing, Point-of-Care Testing, Home / Self-Testing), By End User (Hospitals & Clinics, Diagnostic Laboratories, Homecare Settings, Public Health Laboratories / Community Clinics) |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
| Prominent Players |
Abbott Laboratories, F. Hoffmann-La Roche, Becton, Dickinson and Company (BD), Hologic Inc., Danaher Corporation, Cepheid, Siemens Healthineers, Thermo Fisher Scientific, QIAGEN N.V., bioMérieux SA, Bio-Rad Laboratories, DiaSorin S.p.A., QuidelOrtho Corporation, OraSure Technologies, Trinity Biotech, Grifols, Seegene Inc., Sekisui Diagnostics, Chembio Diagnostics, bioLytical Laboratories, and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Global Sexually Transmitted Diseases Diagnostics Market?
▾ The Global Sexually Transmitted Diseases Diagnostics Market size is expected to reach USD 12.3 billion by 2026 and is projected to reach USD 23.5 billion by the end of 2035.
Which region accounted for the largest Global Sexually Transmitted Diseases Diagnostics Market?
▾ North America is expected to have the largest market share in the Global Sexually Transmitted Diseases Diagnostics Market, with a share of about 38.4% in 2026.
How big is the Sexually Transmitted Diseases Diagnostics Market in the US?
▾ The US Sexually Transmitted Diseases Diagnostics market is expected to reach USD 4.0 billion by 2026.
Who are the key players in the Sexually Transmitted Diseases Diagnostics Market?
▾ Some of the major key players in the Global Sexually Transmitted Diseases Diagnostics Market include Roche, Abbott, Danaher, and others.
What is the growth rate in the Global Sexually Transmitted Diseases Diagnostics Market?
▾ The market is growing at a CAGR of 7.5 percent over the forecasted period.