Market Overview
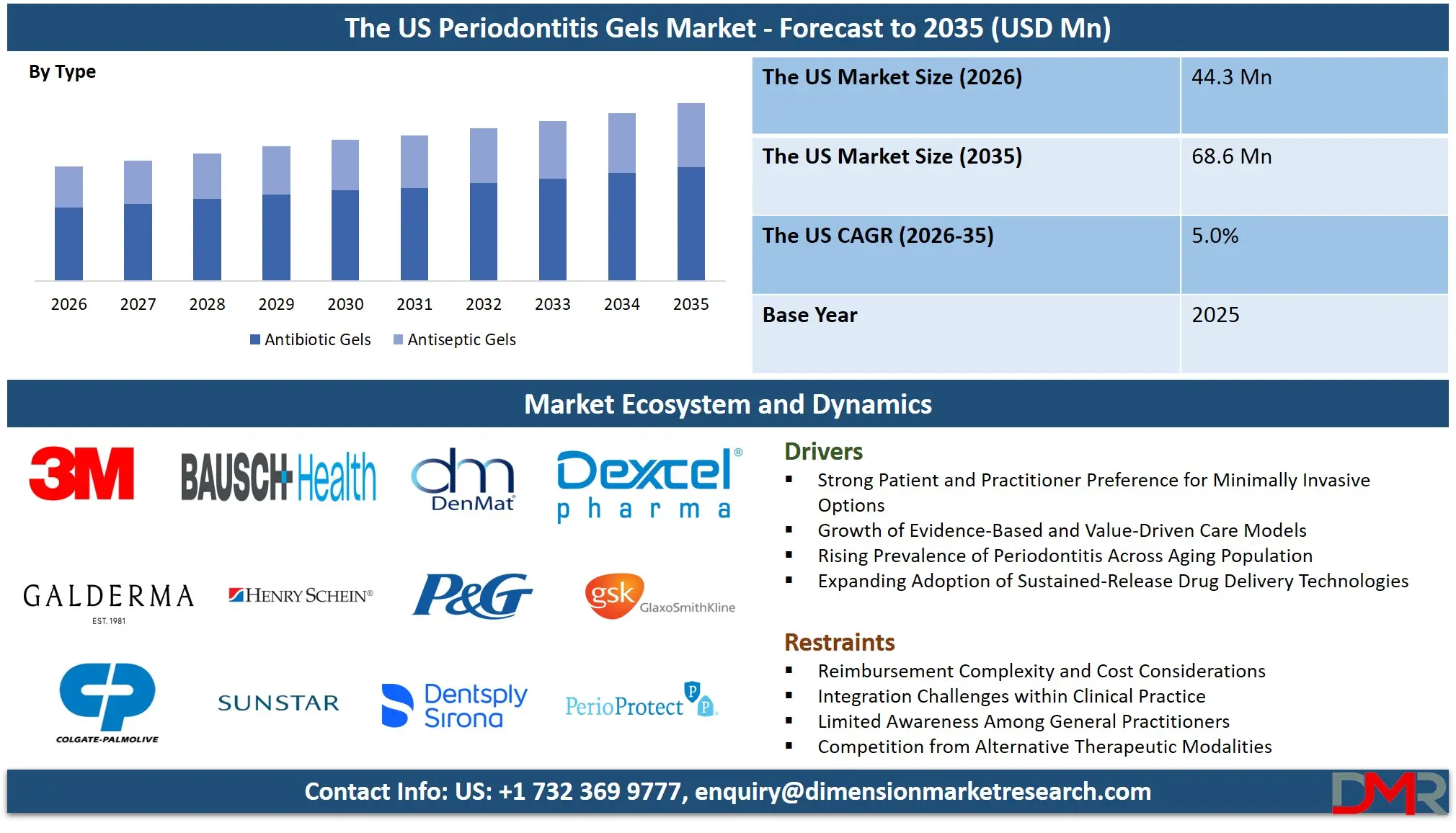
The US Periodontitis Gels Market is projected to reach USD 44.3 million in 2026 and is expected to grow at a CAGR of 5.0% from 2026 to 2035, reaching approximately USD 68.6 million by 2035. The market growth is driven by the escalating incidence of periodontal disease across the adult demographic, growing patient preference for non-invasive treatment modalities, and the clinical advantages offered by site-specific, sustained-release drug delivery systems compared to conventional oral or systemic antibiotics. Furthermore, breakthroughs in biocompatible polymer science, temperature-sensitive gel technologies, and precision-targeted therapeutic carriers are accelerating the availability of more efficacious and patient-friendly treatment options. These scientific advancements are enabling dental practitioners to achieve superior clinical results, minimize treatment frequency, and support the broader transition toward preventive, minimally invasive periodontal care.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The American dental care environment presents distinct characteristics that drive the adoption of periodontitis gels. The nation contends with a substantial and aging population exhibiting high rates of gum disease, a dental insurance landscape increasingly oriented toward cost-efficient chairside therapies, and a clinical community undergoing a significant evolution toward conservative, patient-centered treatment philosophies. This shift is reinforced by a growing repository of clinical evidence validating the effectiveness of locally administered antimicrobial agents, alongside a regulatory framework that encourages innovation in biodegradable drug delivery platforms.
Regulatory developments have reached a pivotal stage. The FDA's efforts to streamline approval pathways for combination products, coupled with its specific guidance on locally administered periodontal therapeutics, are creating more transparent routes to commercialization. Concurrently, the gradual transition toward value-based reimbursement models in dentistry is accelerating the uptake of treatments that demonstrate sustained clinical effectiveness and reduced reliance on surgical interventions. These converging factors are positioning next-generation sustained-release gels to become the cornerstone of non-surgical periodontal therapy.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The competitive arena reflects this dynamic evolution. Established pharmaceutical entities with oral health portfolios, alongside specialized biotechnology firms focused on advanced drug delivery, compete alongside emerging players leveraging novel polymer chemistry and nanoscale formulation techniques. New entrants are introducing gels with enhanced tissue adhesion, extended release durations, and improved patient comfort profiles, while incumbents continue to invest in clinical evidence generation, professional education, and expansive distribution networks. The growing influence of Dental Service Organizations (DSOs) is also reshaping the market, as these entities implement standardized, evidence-based treatment protocols across their affiliated practices, generating consistent demand for reliable, user-friendly gel formulations.
The US Periodontitis Gels Market: Key Takeaways
- Strong Growth Momentum: The US market is forecast to climb from USD 44.3 million in 2026 to USD 68.6 million by 2035, propelled by the convergence of high periodontal disease prevalence, a pronounced shift toward minimally invasive dental procedures, and the proven therapeutic benefits of locally delivered, sustained-release gel formulations.
- Disease Prevalence as Primary Market Driver: The widespread occurrence of periodontitis, affecting approximately half of American adults aged 30 and above, constitutes the most significant factor underpinning market demand. The expectation for effective, non-surgical treatment alternatives that promote patient adherence has become the new standard in periodontal care.
- Localized Drug Delivery as a Growth Engine: Moving beyond systemic treatment paradigms, the precise application of therapeutic gels directly into periodontal pockets represents a highly complex yet rapidly expanding opportunity. Manufacturers are increasingly concentrating on advanced delivery technologies, including thermo-responsive and mucoadhesive systems, not solely for therapeutic efficacy but also to gain competitive differentiation through reduced application frequency and enhanced patient experience.
- AI-Enabled Formulation Science: The industry is transitioning from conventional formulation approaches to intelligent biomaterial design. The US based companies are at the forefront of utilizing artificial intelligence to forecast polymer-drug compatibility, optimize degradation timelines, and engineer gels capable of releasing therapeutic agents in response to specific biochemical signals within the periodontal pocket, enabling a new era of responsive, precision therapy.
- Convergence with Digital Workflows: The integration of digital diagnostics and computer-aided treatment planning is facilitating more precise application of gel-based therapies. The combination of AI-enhanced imaging and digitally guided delivery systems allows clinicians to target affected pockets with unprecedented accuracy, addressing limitations associated with conventional blind application techniques and ensuring consistent drug localization.
Impact of the Iran conflict on the US Periodontitis Gels Market
- Supply Chain Vulnerabilities Exposed: The conflict underscored the US healthcare sector's dependence on overseas sources, particularly from Europe and Asia, for active pharmaceutical ingredients and specialty biodegradable polymers essential to gel manufacturing. This realization is prompting efforts to onshore production capabilities for critical drug delivery components.
- Accelerated Technology Transfer from Defense Applications: Military research into advanced wound care hydrogels and controlled-release antimicrobial systems for battlefield injuries is expected to expedite the development of analogous commercial products, particularly for sustained-release formulations designed to manage chronic oral infections in challenging clinical scenarios.
- Intensified Competition for Scientific Talent: Increased defense sector investment in biotechnology, biomaterials science, and controlled-release systems is creating heightened competition for the limited pool of specialized bioengineers and polymer scientists required to advance next-generation periodontal therapeutics.
- Realignment of National Research Priorities: While dental research continues to progress, the redirection of federal focus and funding toward defense-related biomedical initiatives may represent an opportunity cost, potentially delaying public-private collaborations in complementary areas such as microbiome-targeted therapies and personalized periodontal treatment platforms.
The US Periodontitis Gels Market: Use Cases
- Adjunctive Therapy Following Scaling and Root Planing: Following mechanical debridement of periodontal pockets, clinicians frequently apply sustained-release gels to maintain therapeutic antimicrobial concentrations at the treatment site, preventing bacterial recolonization and supporting periodontal attachment recovery without exposing patients to systemic antibiotics.
- Long-Term Maintenance for Medically Compromised Patients: For individuals with diabetes, cardiovascular disease, or other systemic conditions that complicate periodontal management, dental practitioners utilize antiseptic and antibiotic gels as a targeted maintenance strategy to control chronic inflammation while minimizing systemic drug exposure.
- Management of Peri-Implant Infections: In patients with dental implants, specialized degradable gels serve as a vehicle for delivering antimicrobial agents directly to inflamed peri-implant tissues, addressing infection scenarios that often prove resistant to conventional mechanical debridement alone.
- Therapeutic Intervention in Special Populations: For pediatric patients, older adults, or individuals with physical or cognitive limitations that hinder routine oral hygiene, clinicians employ easy-to-apply gel formulations as a non-invasive intervention for managing early periodontal disease, simplifying treatment protocols and improving patient cooperation.
- Controlled Clinical Trial Logistics: Pharmaceutical developers conducting multicenter studies of novel gel formulations are leveraging sophisticated delivery tracking systems integrated with clinical trial management platforms. These systems enable geofencing capabilities that ensure temperature-sensitive investigational products reach designated clinical sites, schedule precise administration windows for study participants, and maintain comprehensive chain-of-custody documentation for regulatory compliance.
The US Periodontitis Gels Market: Stats & Facts
Centers for Disease Control and Prevention (CDC)
- Approximately 47% of adults aged 30 years and older in the US exhibit some form of periodontal disease.
- Disease prevalence increases substantially with age, affecting more than 70% of adults aged 65 and older.
- Periodontal disease ranks as a primary contributor to tooth loss among American adults.
- The condition demonstrates well-established associations with other chronic diseases, including diabetes mellitus and cardiovascular disease.
American Academy of Periodontology (AAP)
- Non-surgical interventions, including locally administered antimicrobial therapies, constitute the initial treatment approach for periodontitis.
- Clinical studies indicate that sustained-release gels used as an adjunct to scaling and root planing yield significant reductions in probing depths and improvements in clinical attachment levels.
- Patient adherence to recommended oral hygiene practices remains a critical determinant of treatment success.
- Minimally invasive treatment modalities are increasingly favored by both practitioners and patients.
National Institute of Dental and Craniofacial Research (NIDCR)
- The Institute provides substantial funding for research into novel drug delivery systems targeting oral diseases.
- Investments in biomaterials science and nanotechnologies are catalyzing innovation in periodontal therapeutics.
- Ongoing research into the oral microbiome is informing the development of more targeted treatment approaches.
- The movement toward personalized medicine is shaping the evolution of patient-specific therapeutic strategies.
FDA & Industry Reports
- The US represents one of the largest global markets for dental therapeutics.
- The segment for locally administered antimicrobial products is projected to experience robust growth.
- FDA's Orphan Drug designation has stimulated development efforts for specific, underserved periodontal conditions.
- Combination products incorporating both drug and device components constitute a growing market segment.
American Dental Association (ADA)
- More than 200,000 licensed dentists practice in the US.
- Adoption of digital dentistry technologies, including AI-assisted diagnostic tools, is accelerating across the profession.
- Dental Service Organizations (DSOs) continue to expand, contributing to the standardization of clinical protocols.
- Reimbursement policies for periodontal therapies significantly influence treatment adoption patterns.
US Department of Health and Human Services (HHS)
- The "Healthy People 2030" initiative includes specific objectives aimed at reducing the proportion of adults affected by periodontitis.
- Tele-dentistry platforms are expanding access to oral healthcare, particularly in underserved communities.
- Federal programs emphasize the integration of oral health considerations into broader primary healthcare delivery.
The US Periodontitis Gels Market: Market Dynamic
Driving Factors in the US Periodontitis Gels Market
Strong Patient and Practitioner Preference for Minimally Invasive Options
A primary catalyst for market growth is the widespread preference among both patients and dental professionals for non-surgical treatment alternatives. The desire to avoid invasive procedures, shorten recovery periods, and manage periodontal disease through simple chairside applications has transitioned from a niche consideration to a standard expectation across a substantial portion of the patient population. This shift ensures consistent demand for effective non-surgical therapeutic options. Additionally, the well-documented clinical benefits of locally applied gels, including measurable improvements in pocket depth reduction and clinical attachment, strongly influence treatment protocol adoption. Dental practices are increasingly incorporating these therapies to achieve favorable clinical outcomes and maintain high patient satisfaction ratings.
Growth of Evidence-Based and Value-Driven Care Models
Within the US healthcare environment, treatment efficacy and demonstrated long-term value have become central considerations for both clinical decision-making and reimbursement determinations. Payment models are progressively favoring interventions supported by robust clinical evidence over traditional surgical approaches. Professional awareness, reinforced by decades of clinical research and guidance from organizations such as the American Academy of Periodontology, is highly developed, leading to growing practitioner acceptance of advanced gel-based therapies that offer proven advantages. This clinical pull encourages manufacturers to continue innovating while achieving cost efficiencies through scaled production.
Restraints in the US Periodontitis Gels Market
Reimbursement Complexity and Cost Considerations
Despite strong clinical demand, reimbursement structures for periodontal gels remain inconsistent. Insurance coverage varies significantly across payers, and patient out-of-pocket expenses can pose barriers to adoption, particularly among individuals without comprehensive dental benefits. This situation has the potential to create disparities in treatment access, with patients in lower socioeconomic brackets facing greater challenges in accessing these adjunctive therapies compared to those with more extensive insurance coverage.
Integration Challenges within Clinical Practice
Incorporating novel gel formulations into established dental workflows presents practical challenges. Adjustments to clinical protocols may require additional chair time and training for dental hygienists and periodontists. Workforce constraints and high patient volumes in many general dental practices can lead to variability in application technique, potentially affecting treatment consistency and outcomes. Furthermore, the patient-specific nature of periodontal therapy, requiring individualized treatment planning based on pocket depth, disease severity, and anatomical considerations, can complicate standardization efforts across busy clinical settings.
Opportunities in the US Periodontitis Gels Market
Expanding Geriatric and Medically Complex Patient Population
The aging demographic represents a substantial growth opportunity for periodontitis gels. Older adults frequently contend with both periodontal disease and multiple systemic conditions that complicate treatment. Effective management of oral infections in this population can yield benefits beyond oral health, potentially improving outcomes for conditions such as diabetes and cardiovascular disease. The growing emphasis on integrated healthcare, which recognizes the connections between oral and systemic health, creates opportunities for gel-based therapies as part of comprehensive patient management. The financial implications are significant: uncontrolled periodontal disease has been associated with increased healthcare expenditures related to management of comorbid conditions.
AI-Guided Personalization of Therapeutic Formulations
The next frontier in periodontal therapy involves moving beyond standardized formulations toward personalized treatments designed using artificial intelligence. Emerging platforms are leveraging patient-specific data, including genetic markers, microbiome profiles, and immunological parameters, to predict optimal drug selection, polymer composition, and release kinetics for individual patients. This approach extends to customizing bio-adhesion characteristics and degradation timelines based on the unique biochemical environment of each patient's periodontal pockets. Early-stage companies and established manufacturers alike are investing in these capabilities, aiming to create truly individualized therapeutic systems that adapt to patient-specific biological contexts.
Trends in the US Periodontitis Gels Market
Integrated Multi-Agent Formulations
The market is evolving beyond single-active-ingredient products toward sophisticated formulations that combine multiple therapeutic agents with complementary mechanisms of action. This integrated approach brings together antimicrobial compounds, anti-inflammatory agents, and tissue-regenerative biomaterials within unified delivery systems. Such formulations aim to address the complex pathophysiology of periodontitis, which involves both bacterial infection and host inflammatory response, more effectively than single-agent approaches. This trend reflects a broader shift in therapeutic strategy from simple antimicrobial treatment toward comprehensive disease management that supports tissue repair and regeneration.
Advanced Biomaterials with Responsive Properties
Smart biomaterials capable of responding to environmental cues are gaining substantial traction in the periodontal gels market. Temperature-sensitive formulations that remain fluid for easy injection but solidify at body temperature to resist displacement represent one notable advancement. Other innovations include enzyme-responsive systems designed to degrade or release therapeutic agents in the presence of bacterial proteases characteristic of active periodontal disease. These sophisticated delivery platforms are transitioning from academic research to commercial development, supported by evolving FDA guidance on combination products. Ongoing clinical investigations are evaluating formulations that not only manage infection but also deliver growth factors and other regenerative agents to support tissue healing.
The US Periodontitis Gels Market: Research Scope and Analysis
By Type Analysis
Within the US periodontitis gels market, antibiotic gels currently account for the largest share of therapeutic utilization. Among these, formulations containing doxycycline and minocycline represent the most widely used products, supported by extensive clinical evidence, established regulatory approvals, and longstanding acceptance as effective adjuncts to mechanical debridement. For clinicians managing moderate to severe periodontitis, antibiotic gels remain the preferred pharmacologic intervention.
Antiseptic gels represent the fastest-expanding segment within the market. Formulations incorporating chlorhexidine and other antimicrobial agents are increasingly utilized in maintenance protocols, early-stage disease management, and clinical scenarios where antibiotic stewardship considerations are paramount. Evolving treatment paradigms are integrating antiseptic gels as a cornerstone of long-term periodontal maintenance. Advanced antiseptic formulations incorporating bio-adhesive polymers enable sustained local activity, supporting less frequent application intervals and enhanced patient convenience.
By Category Analysis
Degradable gels constitute the foundational product category within the US periodontitis gels market. These formulations, designed to biodegrade naturally within the periodontal pocket following drug release, eliminate the need for separate removal appointments, a significant advantage for patient convenience and clinical workflow efficiency. Their resorbable nature has contributed to widespread adoption across general dentistry and specialty practices.
Non-degradable gels, while representing a smaller market segment, maintain relevance in specific clinical contexts where extended retention or removable delivery systems are preferred. These formulations may be applied as strips or films that clinicians remove following the treatment period. Recent innovations in this category focus on novel polymers that enhance comfort and optimize drug release profiles while maintaining the removability characteristic.
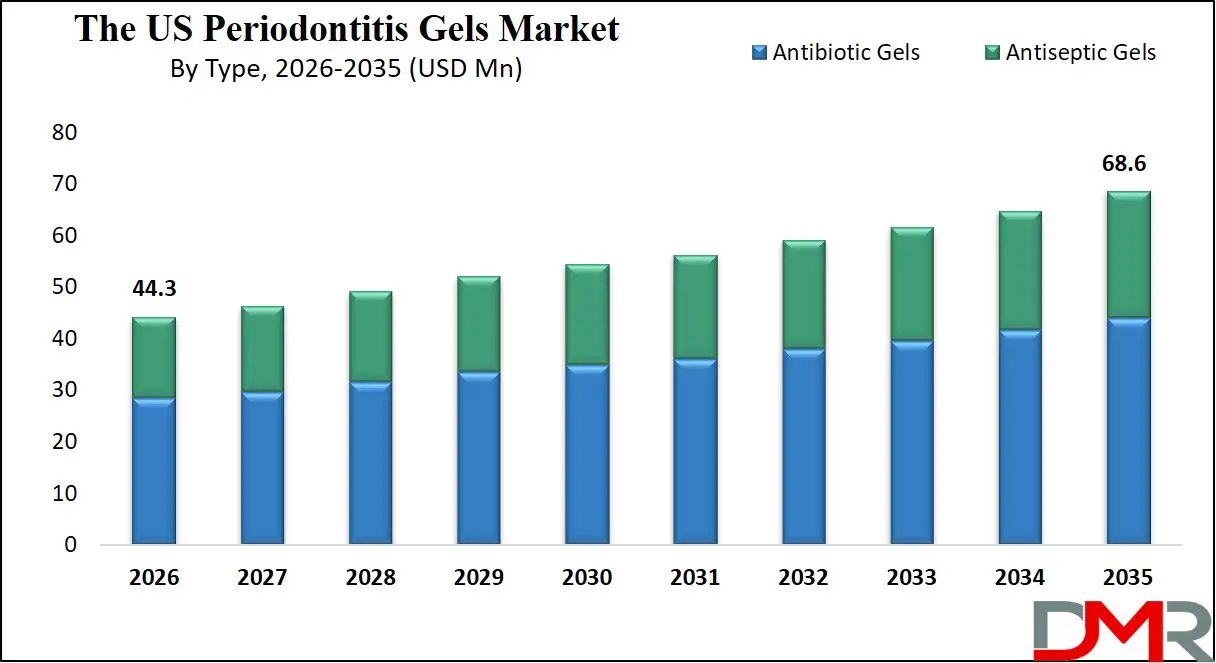
By Application Analysis
Dental clinics represent the largest application segment for periodontitis gels. General dentists and periodontists deliver the majority of non-surgical periodontal therapy in private practice and group practice settings. The integration of gel-based adjunctive therapies into routine periodontal care protocols has been widely adopted across these clinical environments.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Hospitals constitute a growing application segment, particularly for patients with complex medical conditions requiring coordinated care. Hospital dental departments increasingly utilize sustained-release gel formulations for managing periodontitis in patients undergoing cardiac procedures, those with poorly controlled diabetes, or individuals facing other systemic health challenges where oral infection control is critical to overall outcomes.
Other applications include academic dental institutions, Dental Service Organizations (DSOs), and large group practices that serve as early adopters of new technologies and protocol development centers. Manufacturers actively engage with these settings to support clinical education and establish product utilization standards.
The US Periodontitis Gels Market Report is segmented on the basis of the following:
By Type
- Antibiotic Gels
- Antiseptic Gels
By Category
- Degradable Gels
- Non-degradable Gels
By Application
- Hospitals
- Dental Clinics
- Others
Impact of Artificial Intelligence in the US Periodontitis Gels Market
- Accelerated Formulation Discovery: Machine learning algorithms are employed to screen extensive libraries of polymer-drug combinations, predicting optimal compositions for bio-adhesion, controlled release, and stability, thereby significantly reducing research and development timelines.
- Personalized Treatment Prediction: AI platforms analyze patient-specific clinical data, including disease severity, anatomical considerations, and biological markers, to guide clinicians toward optimal gel selection and application protocols for individual cases.
- Enhanced Clinical Trial Design: AI tools synthesize data from patient recruitment, site performance, and real-time outcome monitoring to optimize clinical development pathways and predict trial success probabilities.
- Chairside Diagnostic Support: Advanced analytical tools integrated into diagnostic equipment enable immediate assessment of pocket characteristics and disease activity, providing clinicians with actionable insights to guide gel selection and application parameters.
- Continuous Learning Systems: Manufacturers utilize aggregated data from clinical applications across practice networks to refine formulation models and application guidance, translating real-world outcomes into ongoing product improvement.
- Personalized Clinical Support: AI-driven platforms analyze practitioner preferences and utilization patterns to deliver customized educational content, clinical guidance, and practice management resources.
The US Periodontitis Gels Market: Competitive Landscape
The US periodontitis gels market features a concentrated competitive structure characterized by established pharmaceutical companies with oral health divisions, specialized biotechnology firms, and emerging drug delivery platform developers. Leading companies such as Bausch Health (OraPharma), Tolmar Inc., and other established players maintain significant market positions through their comprehensive sales organizations, established clinical evidence bases, and longstanding professional relationships. Alongside these incumbents, specialty biotechnology companies are increasingly influential, developing novel polymer systems and next-generation formulations that raise expectations for clinical performance and patient experience.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Academic research institutions continue to serve as important sources of foundational innovation, with many novel technologies originating in university laboratories before transitioning to commercial development through licensing arrangements. The US also hosts a robust ecosystem of contract research and manufacturing organizations that enable smaller innovators to advance products through development and commercialization. Technology companies are increasingly partnering with pharmaceutical developers to provide artificial intelligence and data analytics platforms supporting formulation discovery and personalized medicine applications.
Some of the prominent players in the US Periodontitis Gels Market are:
- 3M Company
- Bausch Health Companies Inc. (OraPharma Inc.)
- Dexcel Pharma Ltd.
- Den-Mat Holdings LLC
- Galderma S.A.
- Colgate-Palmolive Company
- Procter & Gamble Co.
- Johnson & Johnson
- GlaxoSmithKline plc
- Sunstar Group
- Dentsply Sirona Inc.
- Pfizer Inc.
- Henry Schein, Inc.
- Straumann Holding AG
- GC Corporation
- Septodont
- Ivoclar Vivadent AG
- Young Innovations, Inc.
- Tolmar Pharmaceuticals, Inc.
- Perio Protect LLC
- Other Key Players
Recent Developments in the US Periodontitis Gels Market
- February 2026: Bausch Health Companies Inc., announced the 25-year milestone of Arestin (minocycline HCl) microspheres, reinforcing its position as the only FDA-approved locally delivered antibiotic adjunct to scaling and root planing for periodontitis treatment.
- February 2026: Dentsply Sirona Inc. expanded multiple U.S. distribution partnerships (including Patterson Dental, Benco Dental, and Burkhart Dental Supply) to improve access to integrated dental treatment solutions, supporting broader adoption of periodontal therapies in clinical settings.
- September 2025: Henry Schein, Inc. launched its "LinkIt" digital workflow platform, integrating practice management with imaging and treatment planning systems, enhancing clinical efficiency and enabling better delivery of periodontal treatments including adjunctive gel therapies.
- May 2025: Colgate-Palmolive Company expanded its professional oral care programs and collaborations with dental professionals to promote periodontal disease management and improve adoption of adjunctive treatment solutions.
- March 2025: Straumann Holding AG expanded its periodontal and peri-implantitis portfolio through continued innovation in regenerative and non-surgical treatment solutions, strengthening its role in comprehensive periodontal care.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 44.3 Mn |
| Forecast Value (2035) |
USD 68.6 Mn |
| CAGR (2026–2035) |
5.0% |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Type (Antibiotic Gels, Antiseptic Gels), By Category (Degradable Gels, Non-degradable Gels), By Application (Hospitals, Dental Clinics, Others) |
| Country Coverage |
The US |
| Prominent Players |
3M Company, Bausch Health Companies Inc. (OraPharma Inc.), Dexcel Pharma Ltd., Den‑Mat Holdings LLC, Galderma S.A., Colgate‑Palmolive Company, Procter & Gamble Co., Johnson & Johnson, GlaxoSmithKline plc, Sunstar Group, Dentsply Sirona Inc., Pfizer Inc., Henry Schein, Inc., Straumann Holding AG, GC Corporation, Septodont, Ivoclar Vivadent AG, Young Innovations, Inc., Tolmar Pharmaceuticals, Inc., Perio Protect LLC, and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the US Periodontitis Gels Market?
▾ The US Periodontitis Gels Market is valued at USD 44.3 million in 2026 and is projected to reach USD 68.6 million by the end of 2035.
What is the growth rate for the US Periodontitis Gels Market?
▾ The market is growing at a robust compound annual growth rate (CAGR) of 5.0% over the forecast period of 2026 to 2035.
Who are the key players in the US Periodontitis Gels Market?
▾ Some of the major key players in the US Periodontitis Gels Market are 3M Company, Bausch Health Companies Inc. (OraPharma Inc.), Dexcel Pharma Ltd., Den-Mat Holdings LLC, Galderma S.A., and many others.