Market Overview
The
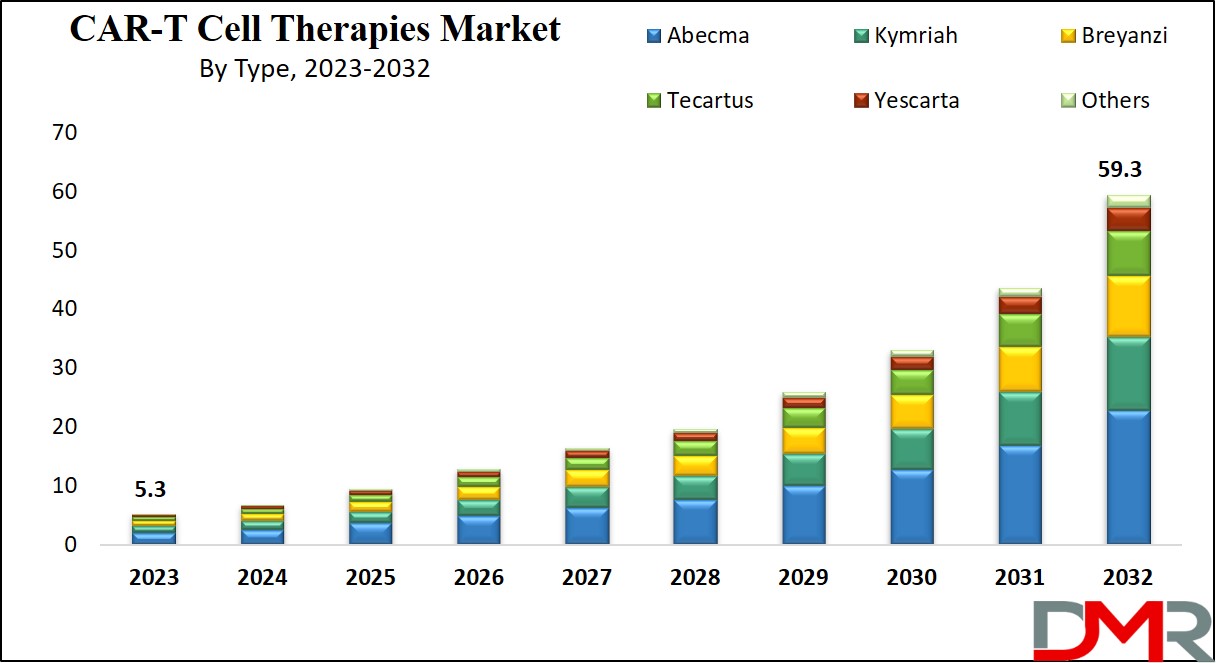
Global CAR-T Cell Therapies Market is expected to have a value of
USD 5.3 billion in 2023, and it is further predicted to reach a market value of
USD 59.3 billion by 2032 at a
CAGR of 30.9%. The market has seen major growth in the recent past and is predicted to grow significantly during the forecasted period as well.
CAR-T cell therapy includes taking T-cells from a patient & modifying them with a unique protein receptor that targets cancer cells, which is commonly used for specific blood cancers & is under investigation for other cancer types. The process includes growing millions of these modified CAR-T cells in a lab & later on delivering them to the patient through an infusion, providing a potential weapon against the cancer.
Market Dynamic
The global rise in cancer cases has driven healthcare providers to grow in favor of CAR-T cell therapy in cancer treatment. As per the Centers for Disease Control & Prevention (CDCP) the United States, in the recent past has seen 1,752,735 new cancer cases & 599,589 cancer-related deaths, with 439 new cases & 146 deaths per 100,000 individuals. The expansion in cancer diagnoses allows healthcare providers to adopt CAR-T therapy due to its various advantages, contributing to market growth. In addition, the growing number of regulatory approvals further drives the expansion of this therapy.
However, CAR T-cell therapies do come with some side effects, including a high reduction in antibody-producing B cells & infections, with Cytokine Release Syndrome (CRS) being a common & severe complication. CRS occurs when T cells release cytokines, which can lead to high immune responses, acting as a constraint on market growth due to safety concerns & the need for effective management of side effects.
Research Scope and Analysis
By Type
Abecma is a major type of CAR-T cell therapy that drives the growth of the market, as it is developed and designed for the treatment of numerous myeloma, a challenging form of blood cancer. Abecma works by modifying a patient's own T cells to focus on a specific protein found in myeloma cells, which helps the immune system recognize & attack the cancer. This therapy has gained significant attention owing to its success in clinical trials, demonstrating a promising ability to induce deep & lasting responses in some patients. While there can be side effects, Abecma offers hope for those who have exhausted other treatment options for multiple myeloma. Its emergence enhances the potential of CAR-T cell therapies to revolutionize the treatment landscape for various types of cancer. Continued research and developments in this field are eagerly anticipated to expand its application and effectiveness further.
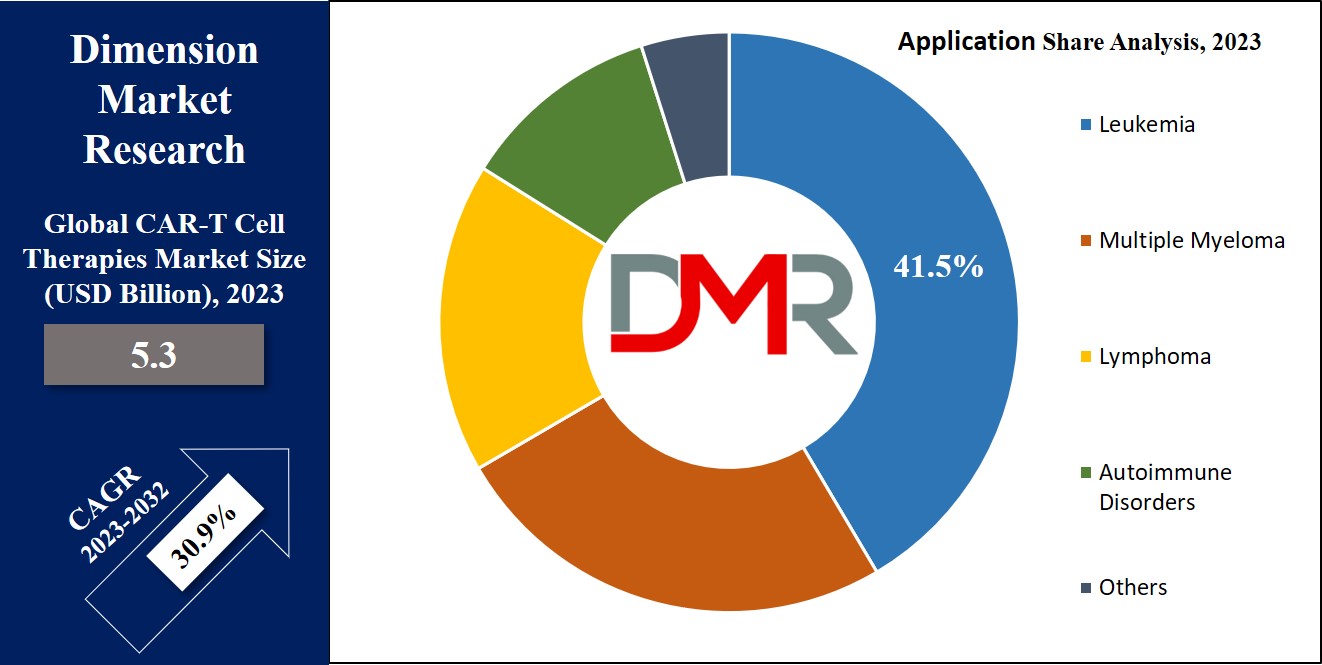
By Application
In terms of application, the market is segmented into leukemia, multiple myeloma, lymphoma, autoimmune disorders, and others, of which leukemia holds the major share as a segment in 2023, and is expected to keep the lead throughout the forecasted period. CAR-T cell therapy has emerged as a new approach to the treatment of leukemia. In addition, it has shown high success in addressing acute lymphoblastic leukemia & few types of chronic lymphocytic leukemia. In addition, a patient's own T cells are genetically modified to target leukemia cells more effectively. , which are then injected back into the patient's body, where they recognize & attack cancer cells with greater accuracy, and also offer hope to patients who may not have responded to traditional therapies and have demonstrated high suspension rates.
While it comes with some side effects, CAR-T therapy's potential to give long-term remission for leukemia patients marks a significant advancement in cancer treatment. As research continues, this application holds promise for broader leukemia management.
By End User
Hospitals are a major driving factor for the global CAR-T cell Therapy market and are anticipated to grow significantly over the forecasted period, as they play a major role in CAR-T cell therapy. These advanced therapies, designed to treat certain types of cancer, require advanced infrastructure & highly trained medical teams for their administration & patient care. Hospitals serve as the primary centers for CAR-T cell therapy delivery, providing a controlled environment for these intricate treatments. They contain all the necessary facilities for cell collection, genetic modification, & reinfusion, ensuring patients receive the therapy safely. Moreover, hospitals are responsible for monitoring patients during & after treatment, addressing potential side effects, and providing critical support in case of adverse reactions. As CAR-T therapies continue to advance, hospitals remain central to the accessibility and success of these groundbreaking treatments.
The CAR-T Cell Therapies Market Report is segmented on the basis of the following:
By Type
- Abecma
- Kymriah
- Breyanzi
- Tecartus
- Yescarta
- Others
By Application
- Leukemia
- Multiple Myeloma
- Lymphoma
- Autoimmune Disorders
- Others
By End User
- Hospitals
- Cancer Care Treatment Centers
- Others
Regional Analysis
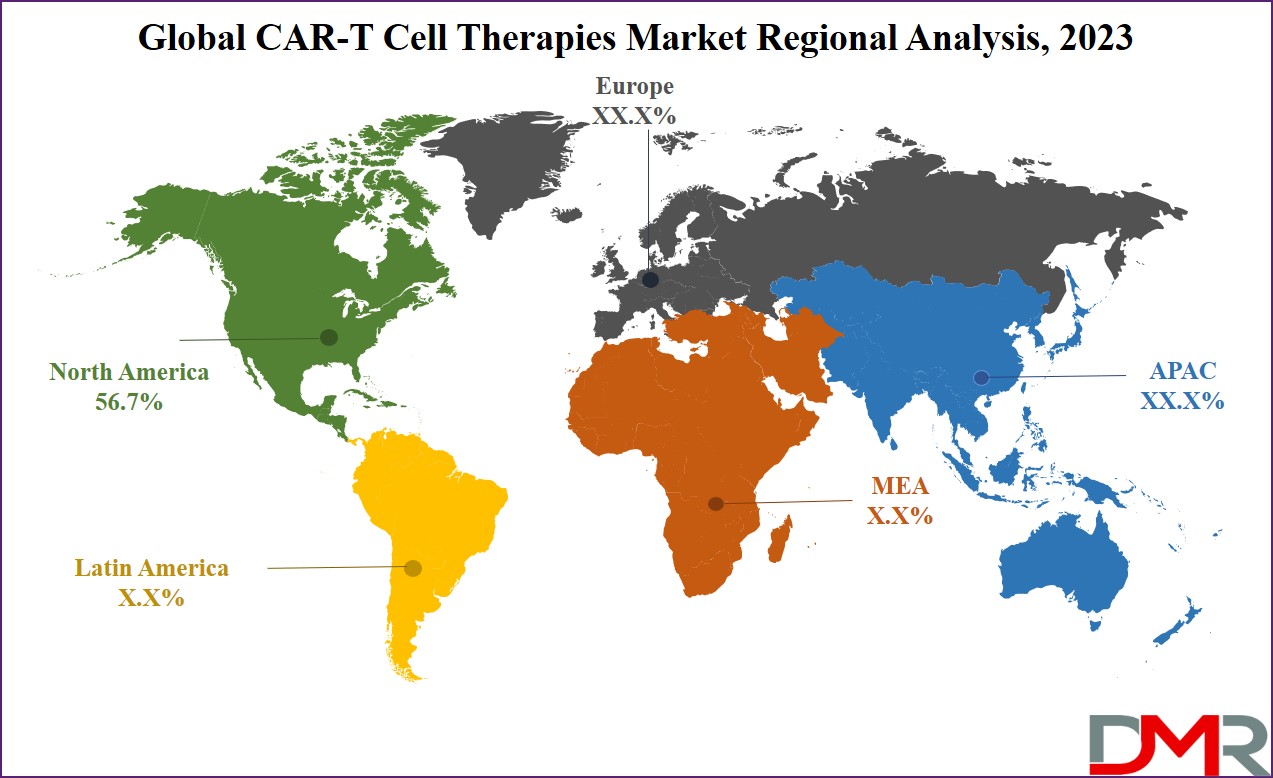
North America leads the CAR-T cell therapy market with a significant
56.7% share of the revenue in 2023, which is primarily due to the strong research infrastructure, commercial activities, & a large number of clinical studies conducted on T-cell therapies across the United States. In addition, the region has experienced a growth in regulatory approvals in both the US & Canada, alongside changes in reimbursement policies, which have driven the growth of adoption of these therapies & promoted substantial market growth.
Moreover, the Asia Pacific region is anticipated for rapid expansion in the coming years. China, in particular, stands out as a major market for CAR-T therapies. The nation has taken the lead in hosting the most registered clinical trials for these therapies, making it an important hub for CAR-T developments in recent years, which is a result of concerted efforts through government investments & healthcare reforms, contributing to the region's fastest growth in the CAR-T cell therapy market.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The CAR-T cell therapy market experiences moderate competition, featuring numerous global players who are employing various strategies to strengthen their position in the market, including looking for partnerships or collaborations, launching new products, expanding into different regions, & merging with or acquiring other businesses. This dynamic approach is helping them grow and make their mark in the industry.
For instance, in June 2022, the FDA granted its approval to Bristol Myers Squibb for a CAR-T cell therapy called Breyanzi (lisocabtagene maraleucel), which is particularly developed to treat adult patients with a type of cancer known as large B-cell lymphoma (LBCL). This approval signifies that Breyanzi has met the necessary requirements and is now recognized as a safe & effective treatment for this condition.
Some of the prominent players in the global CAR-T Cell Therapies Market are:
- Johnson & Johnson
- Novartis AG
- Eli Lilly & Company
- Celyad Oncology
- Bristol-Myers Squibb Company
- ACRO Biosystems
- Servier Laboratories
- Miltenyi Biotec
- Gilead Sciences Inc
- Sorrento Therapeutics Inc
- Other Key Players
COVID-19 Pandemic & Recession: Impact on the Global CAR-T Cell Therapies Market:
The COVID-19 pandemic and the subsequent economic recession had mixed effects on the global CAR-T cell therapies market. While the pandemic disturbed the supply chains & clinical trials, creating some delays in research & development, it also highlighted the importance of innovative treatments like CAR-T in fighting diseases. As healthcare systems adapted, the market showed a rebound with constant growth, driven by a growth in the demand for advanced therapies. Moreover, government investments & extensive healthcare spending, aimed at addressing the pandemic & preparing for future health crises, further supported the expansion of CAR-T cell therapies, making it a promising sector amid challenging times.
| Report Characteristics |
| Market Size (2023) |
USD 5.3 Bn |
| Forecast Value (2032) |
USD 59.3 Bn |
| CAGR (2023-2032) |
30.9% |
| Historical Data |
2017 - 2022 |
| Forecast Data |
2023 - 2032 |
| Base Year |
2022 |
| Estimate Year |
2023 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Type (Abecma, Kymriah, Breyanzi, Tecartus,
Yescarta, and Others), By Application (Leukemia,
Multiple Myeloma, Lymphoma, Autoimmune
Disorders, and Others), By End User (Hospitals,
Cancer Care Treatment Centers, and Others) |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia- Pacific– China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA
|
| Prominent Players |
Johnson & Johnson, Novartis AG, Eli Lilly & Company,
Celyad Oncology, Bristol-Myers Squibb Company,
ACRO Biosystems, Servier Laboratories, Miltenyi
Biotec, Gilead Sciences Inc, Sorrento Therapeutics
Inc, and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users), and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
The Global CAR-T Cell Therapies Market is estimated to reach USD 5.3 billion in 2023, which is further
expected to reach USD 59.3 billion by 2032.
North America dominates the Global CAR-T Cell Therapies Market with a share of 56.7% in 2023.
Some of the major key players in the Global CAR-T Cell Therapies Market are Johnson & Johnson,
Novartis AG, ACRO Biosystems, and many others.
The market is growing at a CAGR of 30.9 % over the forecasted period.