Market Overview
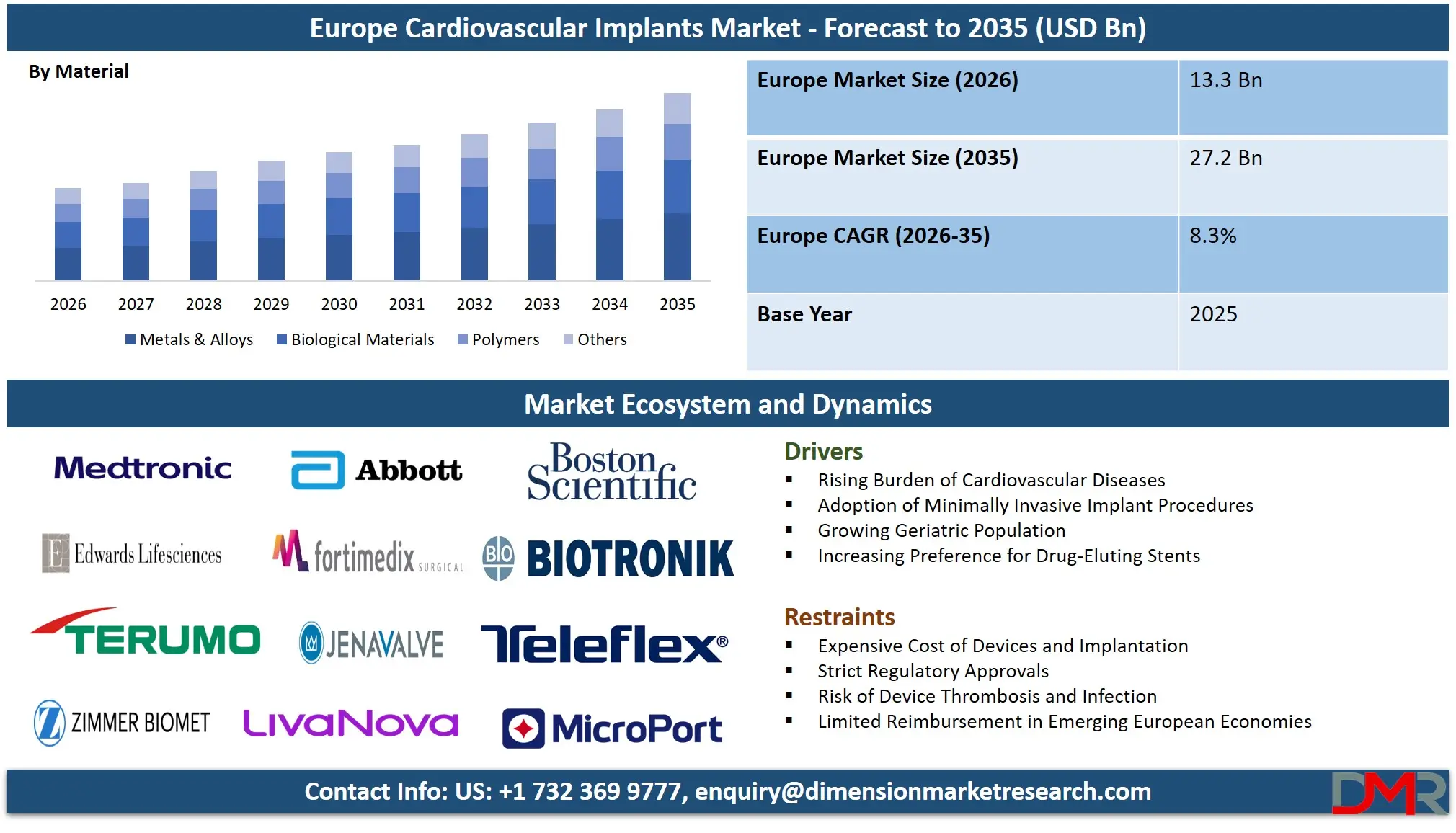
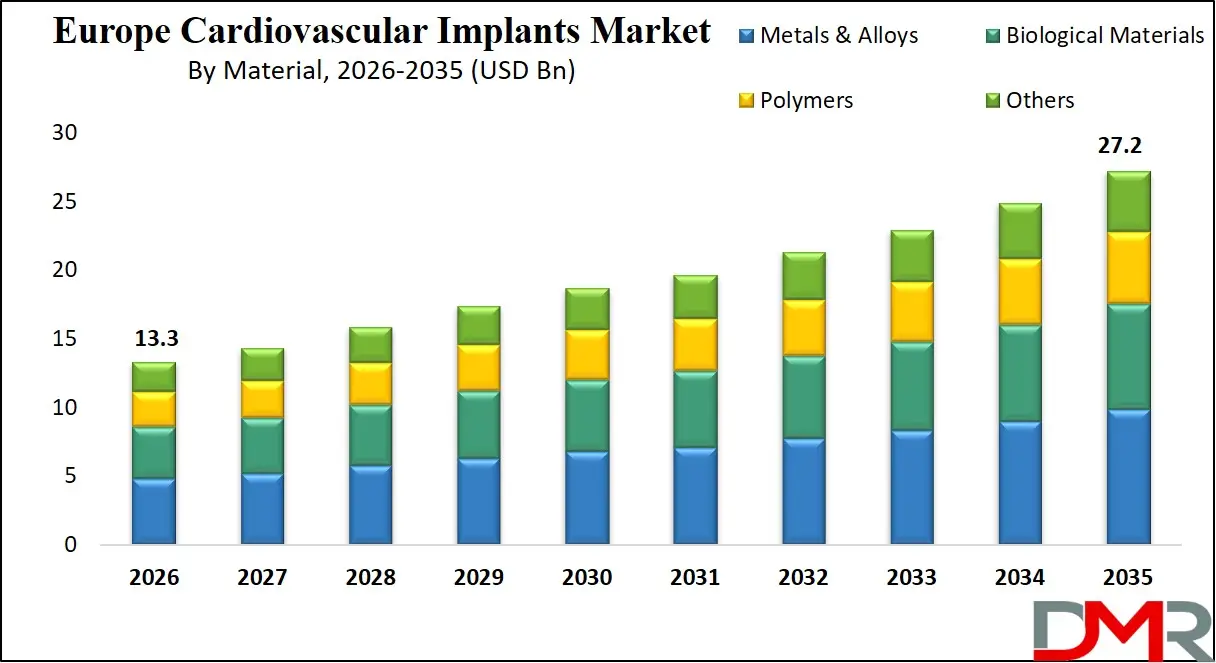
The Europe Cardiovascular Implants Market size is projected to attain USD 13.3 billion in 2026 and grow at a compound annual growth rate of 8.3% to reach a value of USD 27.2 billion in 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
A cardiovascular implant is any device employed in the treatment of different heart and vessel disorders by inserting it within the heart and the vessels. The implantable cardioverter-defibrillators and pacemakers are used in the management of rhythms.
The Europe Cardiovascular Implants Market is expanding due to the rising rate of the use of cardiovascular implants such as coronary stents, heart valves, and cardiac resynchronization therapy in other countries such as Germany, France, and the United Kingdom. This is due to the growing percentage of the aging population, the wide-ranging use of heart failure, and the growing percentage of adopting refined biomaterials within the area.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Europe Cardiovascular Implants Market: Key Takeaways and Other Influencing Factors
- Strong Market Growth Trajectory: The Europe Cardiovascular Implants Market is expected to increase to USD 27.2 billion by 2035 with a strong CAGR of 8.3% due to an increase in the use of minimally invasive cardiac procedures and the next-generation implant materials.
- Dominance of Coronary Stents and Hospitals: Coronary stents dominate with 38.0% share and hospitals with 58.0% with a high preference of the interventional cardiology procedures and well established clinical infrastructure in Europe.
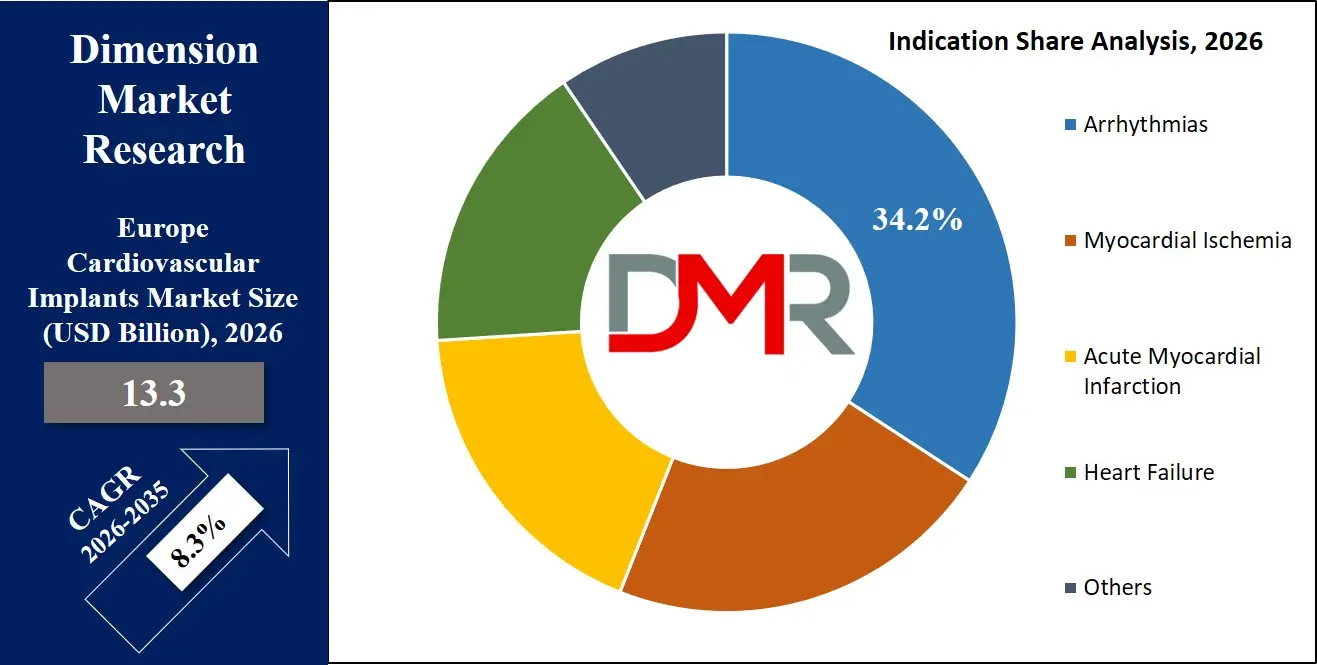
- Arrhythmias Segment Leading Demand: Arrhythmias dominates with 34.2% share due to increasing need for implantable cardioverter defibrillators and cardiac pacemakers for heart rhythm disorders across Europe.
- Shift toward Bioresorbable and Polymer-Based Implants: The gradual adoption of bioresorbable stents, polymer heart valves, and new companies developing advanced metal alloys is creating some rather extraordinary paradigms in cardiovascular implant design across Europe.
- High Burden of Cardiovascular Mortality: According to World Health Organization, cardiovascular diseases cause over 425 million deaths in Europe (more than 42.5% of all deaths) and almost 10000 deaths daily by disease with a significant implication on the demand of cardiovascular implants.
- Large Patient Pool across Europe: According to the OECD, the cardiovascular disease prevalence among the population is estimated to be 62 million individuals in Europe resulting in high demand of the coronary stents, ICDs and pacemakers.
- Significant Clinical and Hospital Load: Cardiovascular diseases constitute 32.4% of the overall EU deaths with hospital discharge rate of over 2,100 per 100,000 people, and this is reported by Eurostat.
Impact of the Iran conflict on the Europe Cardiovascular Implants Market
The Iran conflict is causing supply chain disruption in the European market making transport costly and causing some of the components used in coronary stents and implantable devices to be delayed. Combining with the rise in oil prices and raw material, the Iran conflict is raising the cost of production of cardiovascular implants by this raising their price. Middle level market effects due to diversified sourcing but long-term market volatility may lead to pricing and market stagnation.
Europe Cardiovascular Implants Market: Use Cases
- Lifestyle Disease Management: The European population particularly the Europeans living in the cities are experiencing lifestyle diseases and the hour is high time to offer them ayurvedic remedies, ayurvedic diets, stress relief medicines, and detox therapy.
- Corporate Wellness and Employee Health Programs: European corporations are currently utilizing ayurvedic wellness programs to employee health and wellness programs and the idea of the time is to market ayurvedic stress management, ayurvedic health consultations and immunity boosters as part of employee wellness programs.
- Immunity Enhancement and Seasonal Health: Due to the increased health and wellness awareness among Europeans, the need of the hour is to promote ayurvedic immunity boosters, especially ayurvedic immunity boosters like 'Chywanaprash,' 'Tulsi,' and 'Herbal Adaptogens,' and the ayurvedic solution is being adopted by the Europeans and the populace residing in Europe.
- Skin and Hair Care in Personal Care Segment: The Ayurvedic skin and hair care is rapidly gaining popularity in the Europeans section, and what is required is to market the ayurvedic skin and hair care, particularly ayurvedic products to treat skin and hair diseases such as eczema, acne, premature aging and hair fall.
Europe Cardiovascular Implants Market: Market Dynamic
Driving Factors in the Europe Cardiovascular Implants Market
Rising Burden of Cardiovascular Diseases
The rising cases of heart diseases including arrhythmias, heart failure, and coronary artery disease are pushing the demand of coronary stents, ICDs, and pacemakers in Europe. It is also favored by the increasing knowledge of interventional cardiology and early screening.
Adoption of Minimally Invasive Implant Procedures
The increased use of advanced cardiovascular implants is also speeding up due to growth in transcatheter valve replacement and percutaneous coronary intervention, which is enhancing patient outcomes and lowering recovery time. Adoption is also being enhanced by increased access to hybrid operating rooms and competent interventional cardiologists.
Restraints in the Europe Cardiovascular Implants Market
Expensive Cost of Devices and Implantation
High-tech cardiovascular implant systems are also associated with high costs in terms of equipment, delivery systems, and post-procedural care and, therefore, its use is restricted in the low cost healthcare environment. Increased budget limitations in government health systems also limit large scale implementation.
Strict Regulatory Approvals
The clinically required and stringent medical device regulations in the European region are complicating the process of achieving CE marking on new cardiovascular implants to influence the time to market and innovation cycles. Manufacturers are burdened by compliance requirements like MDR.
Opportunities in the Europe Cardiovascular Implants Market
Growth of Bioresorbable and Polymer-Based Implants
Rising focus on next-generation materials is creating opportunities for bioresorbable stents, polymer heart valves, and biocompatible coatings that reduce long-term complications and improve patient outcomes. This trend supports innovation in implant design and personalized medicine.
Integration with Digital Health and Remote Monitoring
The use of smart implants that have remote monitoring functions is improving the follow-up after the procedure, as they can identify the device malfunction and arrhythmias early. The scope of application is also growing due to continuous innovation in connected cardiac devices.
Trends in the Europe Cardiovascular Implants Market
Shift toward Transcatheter and Leadless Devices
Preference is shifting to transcatheter heart valves and leadless pacemakers compared to traditional surgical implants because of reduced complication rates, reduced hospital stay and better patient recovery. This is accelerating cardiovascular implant product innovation and miniaturization.
Increasing Use of Advanced Biomaterials and Coatings
To improve the thrombosis and enhance the long-term efficacy of the implants, manufacturers are now using drug-eluting stents, polymer-based heart valves, and alloys made of metals that have high biocompatibility. Combining with anticoagulant surfaces is increasing the safety of devices.
Europe Cardiovascular Implants Market: Research Scope and Analysis
By Product Analysis
Coronary stents are expected to dominate the Europe Cardiovascular Implants Market with around 38.0% share in 2026, driven by high prevalence of coronary artery disease, growing adoption of angioplasty, and increasing use of drug-eluting stents. Their ability to restore blood flow and reduce restenosis is accelerating adoption. Implantable cardioverter defibrillators and cardiac pacemakers remain widely used for rhythm management in clinical settings ICDs and pacemakers continue to be popular rhythm management devices in the clinical environment. Their demand is supported by strong familiarity of physicians and established use in hospitals, but transcatheter devices are moving preference towards heart valves and CRT devices.
By Material Analysis
Metals & alloys are expected to dominate the Europe Cardiovascular Implants Market with around 52.1% share in 2026, due to the strength, durability, and high applicability in the coronary stents, pacemakers, and ICDs. Cobalt-chromium, and titanium alloys are still the choice in terms of structural integrity. There is an increase in the use of polymers and biological materials with the increasing demand of transcatheter heart valves, bioresorbable stents and tissue-based heart valves. Their growth is further being encouraged by the increasing interest in biocompatibility and low thrombogenicity.
By Procedure Type Analysis
Angioplasty is expected to dominate the Europe Cardiovascular Implants Market with around 65.0% share in 2026, driven by strong demand for minimally invasive coronary interventions and high volume of stent placements. These are the common operations applied in the treatment of myocardial ischemia and acute myocardial infarction in the hospitals. Complex valve replacement, coronary artery bypass grafting and CRT device implantation still require an open heart operation. The growing attention to hybrid procedures and transcatheter techniques is slowly moving the preference to the less invasive ones.
By Indication Analysis
Arrhythmias are expected to dominate the Europe Cardiovascular Implants Market with around 34.2% share in 2026, driven by high prevalence of atrial fibrillation and sudden cardiac arrest risk, increasing demand for ICDs and pacemakers for heart rhythm disorders.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Heart failure and myocardial ischemia segments are growing with rising adoption of CRT devices and coronary stents. Increasing focus on preventive cardiology and early intervention is further supporting expansion across all indications.
By End-User Analysis
Hospitals are expected to dominate the Europe Cardiovascular Implants Market with around 57.9% share in 2026, driven by high patient volume, advanced cardiac surgery infrastructure, and strong use of cardiovascular implants for diagnosis and treatment. These environments are based on the use of coronary stents, ICDs, pacemakers and heart valves to provide all-encompassing cardiac care. The presence of qualified surgeons and positive reimbursement also contributes to their supremacy. Laboratories Cardiac catheterization laboratories are increasing in number because of the increasing angioplasty and transcatheter procedures. The use of outpatient-based interventional procedures is also increasing; therefore, their utilization is increasing, particularly in the case of routine stent placement.
The Europe Cardiovascular Implants Market Report is segmented on the basis of the following:
By Product Type
- Coronary Stents
- Implantable Cardioverter Defibrillators (ICDs)
- Heart Valves
- Implanted Cardiac Pacemakers
- Cardiac Resynchronization Therapy (CRT) Devices
- Peripheral Stents
- Others
By Material
- Metals & Alloys
- Polymers
- Biological Materials
- Others
By Procedure Type
- Angioplasty
- Open Heart Surgery
By Indication
- Arrhythmias
- Myocardial Ischemia
- Acute Myocardial Infarction
- Heart Failure
- Others
By End-User
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratories
- Others
Impact of Artificial Intelligence on the Europe Cardiovascular Implants Market
The Europe Cardiovascular Implants Market is being redefined by artificial intelligence that allows selecting patients faster and more precisely, planning the procedure, and monitoring of the post-implant outcomes with the help of the advanced imaging and data processing. AI based algorithms enhance the accuracy of diagnostics by detecting small heart defects and behavior of the device.
The connectivity with implantable devices facilitates remote monitoring and automatic alerts on arrhythmia or device failure. AI enabled predictive analytics is not only improving risk assessment and individualized cardiac care to post-procedural follow-up systems but also enhancing clinical workflow efficiency by lessening physician workloads and increasing the speed of decision making.
By Region
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Europe Cardiovascular Implants Market: Competitive Landscape
The Europe Cardiovascular Implants Market is led by Germany, United Kingdom, and France due to advanced healthcare infrastructure and high adoption of interventional cardiology procedures. Western Europe dominates with strong use of coronary stents, ICDs, and transcatheter heart valves. Eastern Europe is gradually growing with improving healthcare access, rising cardiovascular disease awareness, and increasing investments in cardiac surgery centers.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Some of the prominent players in the Europe Cardiovascular Implants Market are:
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- Edwards Lifesciences Corporation
- Johnson & Johnson
- Biotronik SE & Co. KG
- B. Braun Melsungen AG
- LivaNova PLC
- Terumo Corporation
- W. L. Gore & Associates, Inc.
- Getinge AB
- Teleflex Incorporated
- MicroPort Scientific Corporation
- JenaValve Technology GmbH
- Berlin Heart GmbH
- CARMAT SA
- Corcym S.r.l.
- Calon Cardio Ltd.
- Fortimedix Surgical B.V.
- Proxy Biomedical Ltd.
- Other Key Players
Recent Developments in the Europe Cardiovascular Implants Market
- August 2025: Edwards Lifesciences Corporation supported updated ESC/EACTS European guidelines incorporating expanded indications for TAVR, driven by evidence from the EARLY TAVR trial and CE Mark approvals in Europe.
- June 2025: Abbott Laboratories introduced AI-based imaging solutions to enhance precision in transcatheter valve placement, reducing procedural complications and improving outcomes in European cardiovascular implant procedures.
- April 2025: Medtronic plc received expanded regulatory approval in Europe for its transcatheter aortic valve replacement (TAVR) systems for intermediate-risk patients, strengthening its structural heart portfolio in the region.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 13.3 Bn |
| Forecast Value (2035) |
USD 27.2 Bn |
| CAGR (2026–2035) |
8.3% |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Product Type (Coronary Stents, Implantable Cardioverter Defibrillators (ICDs), Heart Valves, Implanted Cardiac Pacemakers, Cardiac Resynchronization Therapy (CRT) Devices, Peripheral Stents, and Others), By Material (Metals & Alloys, Polymers, Biological Materials, and Others), By Procedure Type (Angioplasty and Open Heart Surgery), By Indication (Arrhythmias, Myocardial Ischemia, Acute Myocardial Infarction, Heart Failure, and Others), By End-User (Hospitals, Specialty Clinics, Ambulatory Surgical Centers, Cardiac Catheterization Laboratories, and Others) |
| Regional Coverage |
Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe |
| Prominent Players |
Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, Edwards Lifesciences Corporation, Johnson & Johnson, BIOTRONIK SE & Co. KG, B. Braun Melsungen AG, LivaNova PLC, Terumo Corporation, W. L. Gore & Associates, Inc., Getinge AB, Teleflex Incorporated, MicroPort Scientific Corporation, JenaValve Technology GmbH, Berlin Heart GmbH, CARMAT SA, Corcym S.r.l., Calon Cardio Ltd., Fortimedix Surgical B.V., Proxy Biomedical Ltd., and Other Key Players. |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users), and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days, and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Europe Cardiovascular Implants Market?
▾ The Europe Cardiovascular Implants Market size is estimated to have a value of USD 13.3 billion in 2026 and is expected to reach USD 27.2 billion by the end of 2035.
Who are the key players in the Europe Cardiovascular Implants Market?
▾ Some of the major key players in the Europe Cardiovascular Implants Market are Boston Scientific Corporation, Edwards Lifesciences Corporation, Teleflex Incorporated, MicroPort Scientific Corporation, JenaValve Technology GmbH, and many others.
What is the growth rate in the Europe Cardiovascular Implants Market?
▾ The market is growing at a CAGR of 8.3% over the forecasted period.