What is the Human Microbiome Therapeutics Market Size?
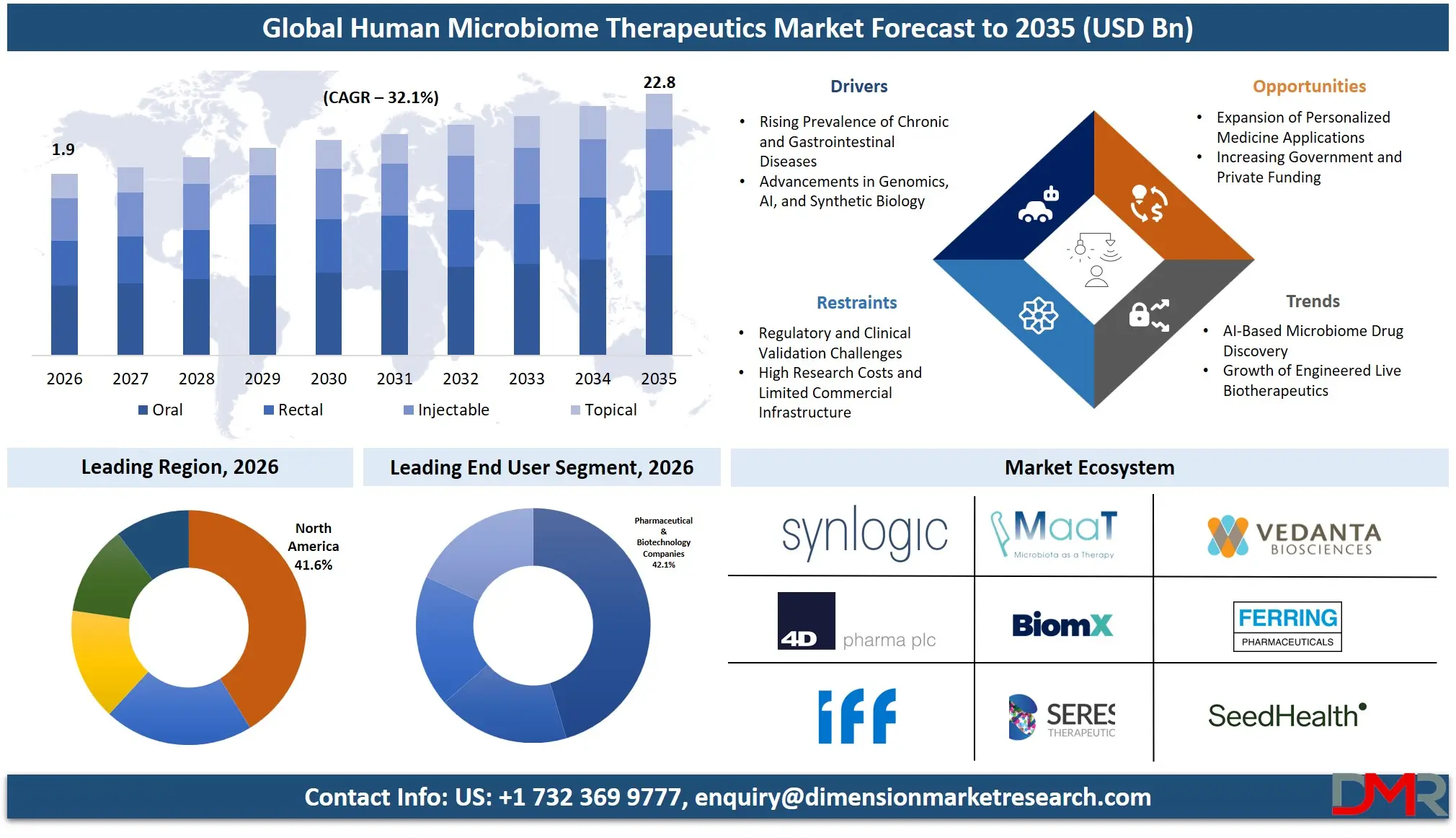
The Human Microbiome Therapeutics Market size is expected to be USD 1.9 billion in 2026 and increase at a compound annual growth rate of 32.1% to USD 22.8 billion in 2035 due to growing number of gastrointestinal, metabolic, autoimmune, and neurological diseases is propelling the market forward.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The Human Microbiome Therapeutics Market can be described by various innovations and business model changes related to restoration and reprogramming of microorganisms within the human body for therapeutic purposes. Within the Human Microbiome Therapeutics Market, there are such products as live biotherapeutic products, fecal microbiota transplantation, postbiotics, synbiotics, bacteriophage therapy, and microbiome-related small molecules for treatment of gastrointestinal diseases, metabolic diseases, neurodegenerative diseases, dermatological conditions, cancers, and immuno-oncology therapies.
There are such driving factors for the development of the Human Microbiome Therapeutics Market as an increase in prevalence of chronic diseases, investments into scientific research on the microbiome, clinical studies, and positive regulatory policy. Innovation factors for the Human Microbiome Therapeutics Market are development of metagenomics, synthetic biology, CRISPR/Cas gene-editing technology, and utilization of AI in identification of products in the microbiome sector. Moreover, the Human Microbiome Therapeutics Market is boosted by increasing demand for personalized medicine and healthcare.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US Human Microbiome Therapeutics Market
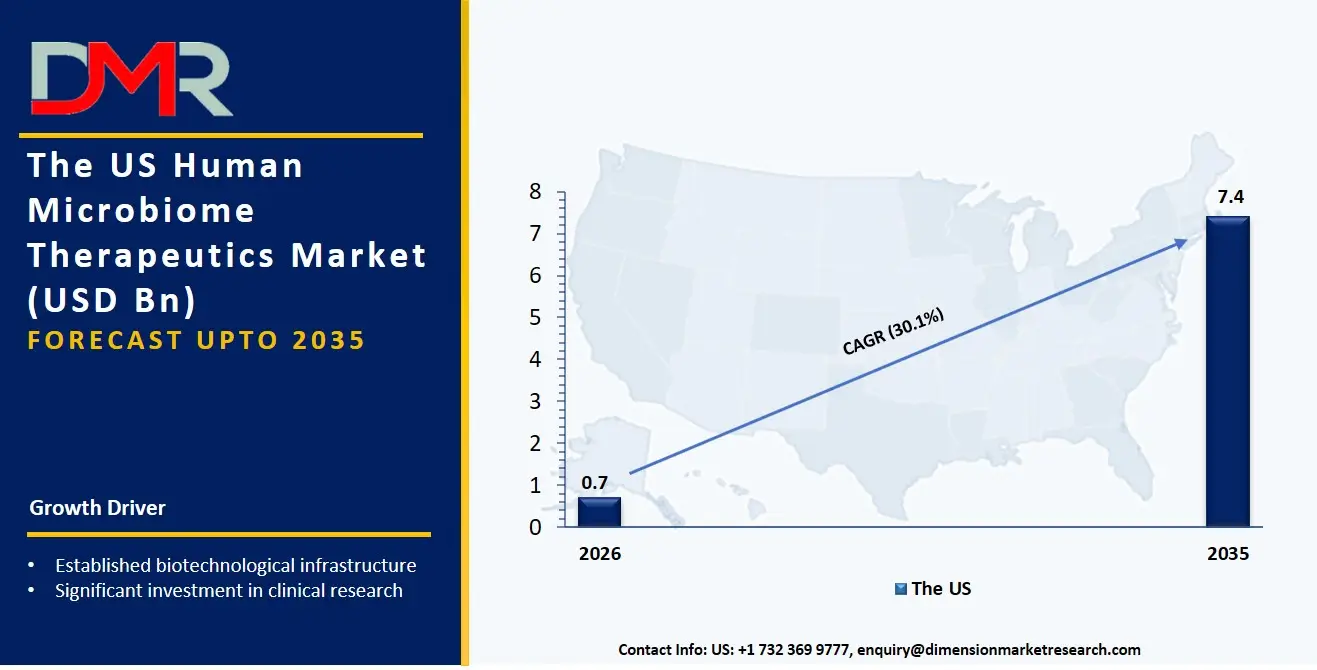
The US Human Microbiome Therapeutics Market size is estimated to be USD 700 million in 2026 and is expected to increase at a CAGR of 30.1% over the forecast period.
The driving factors behind the growth of the US Human Microbiome Therapeutics Market include well-established biotechnological infrastructure, significant investment in clinical research, and conducive regulatory environment for the development of microbe-based drugs. The US dominates in microbiome-related clinical trials, especially for gastroenterology and oncology applications. Growing approval rate of fecal microbiota preparations by the FDA and the growing number of collaborations between pharma and biotech companies will drive market growth. The presence of government-driven microbiome research programs, state-of-the-art genomics, and AI-driven microbiome analysis platforms adds more impetus to the US market.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Europe Human Microbiome Therapeutics Market
The Europe Human Microbiome Therapeutics Market size is estimated to be USD 475 million in 2026 and is expected to increase at a CAGR of 31.3% over the forecast period.
Europe can be described as an expanding market owing to the availability of highly sophisticated healthcare infrastructure, microbiome research collaborations, and regulations that foster the growth of biotechnology innovations. There have been various initiatives geared towards the development of therapeutics based on sustainable and precision healthcare policies, thus promoting the development of microbiome medicines in Germany, France, and the United Kingdom. The European Medicine Agency has been at the forefront of fostering innovations in live biotherapeutics as well as microbiome oncology treatments.
Japan Human Microbiome Therapeutics Market
The market size of Japan Human Microbiome Therapeutics will be USD 95 million in 2026 and at a CAGR of 31.9% in the forecast period.
The Japanese market for human microbiome therapeutic products is showing explosive growth due to an aging population, high incidence of diseases related to the digestive and metabolic systems, and a focus on preventive medicine. There is active governmental support towards R&D activities involving biotechnology and personalized medicines, which could be the driving force behind innovation in microbiome technology. There have been significant developments by the pharmaceutical companies in Japan based on treatments involving gut microbiome, probiotics, and metabolomics.
Key Takeaways
- Market Size & Forecast: The Human Microbiome Therapeutics Market size is projected to reach USD 1.9 billion in 2026 and is anticipated to have a value of USD 22.8 billion in 2035.
- Growth Rate & Outlook: The Human Microbiome Therapeutics Market size is set to grow at a compound annual growth rate of 32.1% during the forecast period of 2026 to 2035.
- Primary Growth Drivers: Some of the major growth drivers in the market include Increasing Growing Occurrence of Chronic and GI Diseases, and more.
- Key Market Trends: Some of the major trends in the market are Evolution of Engineered Live Biotherapeutics, and more.
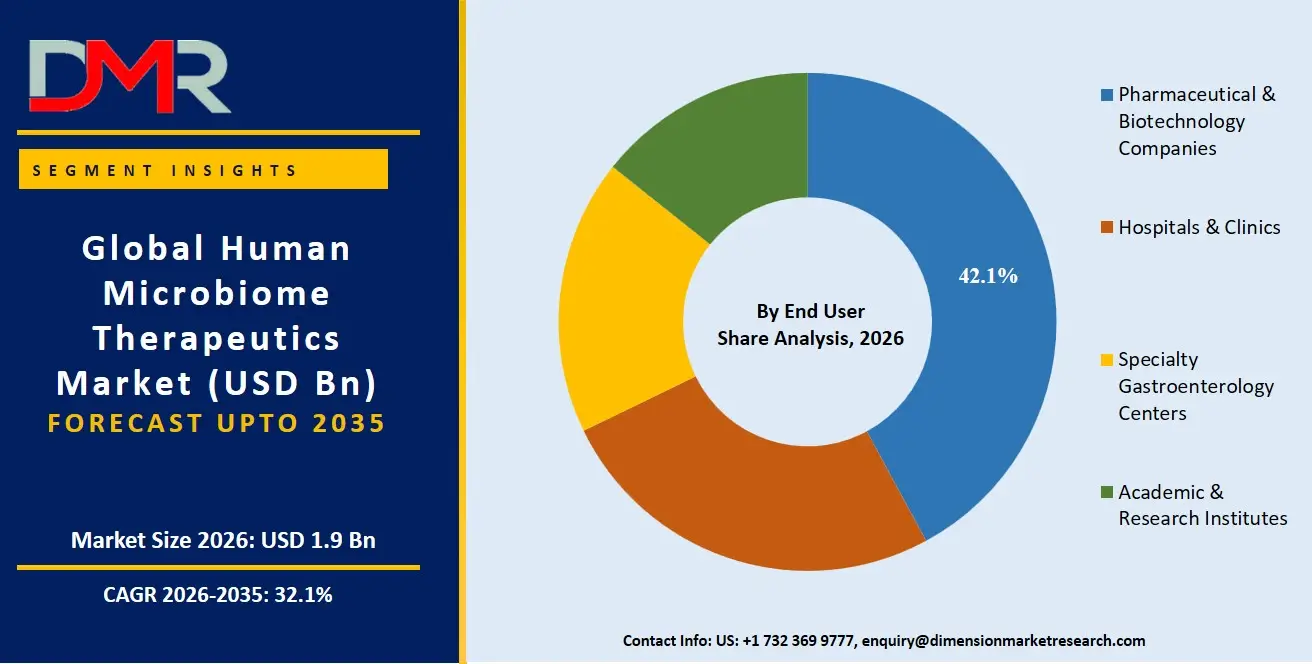
- By End User: The Pharmaceutical & Biotech Companies segment is anticipated to get the majority share of the Human Microbiome Therapeutics market in 2026.
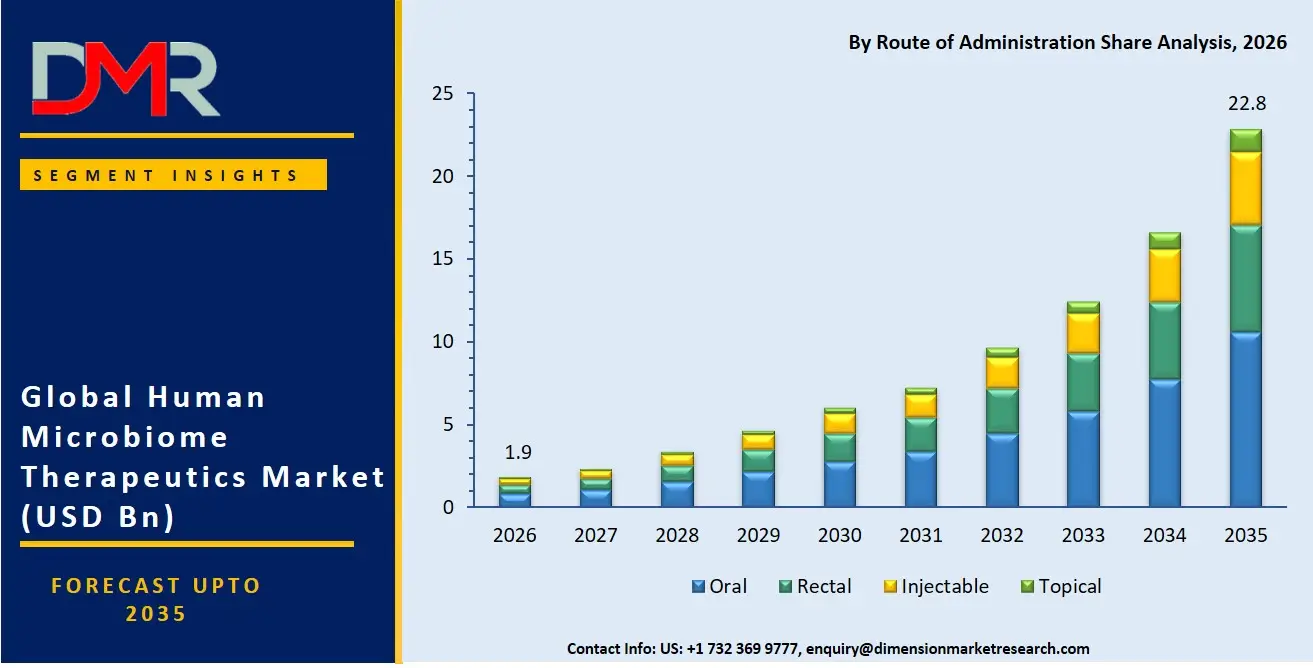
- By Route of Administration: The oral segment is expected to get the largest revenue share in 2026 in the Human Microbiome Therapeutics market.
- By Technology Platform: The Metageonomics segment is expected to get the largest revenue share in 2026 in the Human Microbiome Therapeutics market.
- Regional Leadership: North America is set to lead the Human Microbiome Therapeutics market with an estimated 41.6% share in 2026.
What is the Human Microbiome Therapeutics?
Human Microbiome Therapeutics refer to pharmaceutical products designed to affect the microbial life within the human body that includes not only bacteria but also fungi, viruses, and other organisms. The purpose of such medical solutions is to ensure normal microbial activity, optimize the body's immune function, control metabolism, and provide enhanced protection against diseases. Live microbial preparations, microbiota transplantation, engineered bacteria, postbiotics, synbiotics, and microbiome-derived metabolites represent several categories of such products currently available for the treatment of various diseases ranging from GI disorders to neurodegenerative disorders, cancer, skin diseases, and gynecological issues.
Use Cases
- Management of Gastrointestinal Diseases: The use of the microbiome-based therapy method is commonly used for the management of recurring infections from Clostridioides difficile, inflammatory bowel diseases, Crohn's disease, ulcerative colitis, and IBD. This therapy is useful in keeping the balance between microbes found within the digestive system healthy.
- Oncological Diseases Support: Over time, microbiome-based treatment methods have become increasingly important in improving the effectiveness of immunotherapy and reducing complications associated with chemotherapy in the management of oncological conditions such as colorectal cancer and melanomas.
- Metabolic Disorders Treatment: Microbiome therapies based on human health are helpful in the management of obesity, type 2 diabetes mellitus, and non-alcoholic fatty liver disease. The disorders result from metabolic disturbances, poor insulin sensitivity, and inflammation mediators.
- Promoting Neurological and Immunological Health: The application of microbiome-based therapies is increasing in the management of autism spectrum disorders, Parkinson's disease, psoriasis, and rheumatoid arthritis. More research studies have been done in recent years concerning gut-brain and gut-immune axes to create new microbiome-based therapies.
How AI Is Transforming the Human Microbiome Therapeutics Market
AI is greatly advancing discoveries related to the microbiome therapy by facilitating quick evaluation of complex datasets and biomarker discovery for diseases. AI technology also helps in selecting strains that can be used to develop precise medicines. Furthermore, it enables predictive diagnostics and personalized treatment and accelerates clinical trial analysis.
AI technologies are contributing to operational efficiencies through fast tracking of drug discoveries and microbial engineering advancements. More and more pharmaceutical and biotechnology companies are using AI solutions to learn how to interact with the microbiome to provide treatment for specific conditions, including but not limited to oncological, gastrointestinal, metabolic, and neurological conditions.
Market Dynamic
Driving Factors in the Human Microbiome Therapeutics Market
Growing Occurrence of Chronic and GI Diseases
The growing number of gastrointestinal, metabolic, autoimmune, and neurological diseases is propelling the market forward. The demand for treatment options other than traditional drugs is rising as a result of antibiotic resistance and adverse effects. Microbiome therapies for humans provide precise treatment options by restoring the microbial population and modulating the immune system. The increasing amount of clinical data on the relationship between the microbiome and its host, along with increasing patient knowledge about gut health, is contributing to this trend.
Restraints in the Human Microbiome Therapeutics Market
Expensive Research and Weak Commercial Structure
The development of microbiome-based drugs necessitates the use of expensive equipment and complex manufacturing and testing procedures, which demand huge investments for research. The small-scale biotech companies find it challenging to commercialize their products owing to issues like lack of production scalability and reimbursement. Furthermore, there is poor knowledge about the therapy among health care practitioners, and no standard protocols exist for treatment.
Opportunities in the Human Microbiome Therapeutics Market
Increased Investment by Government and Private Sectors
Both government organizations, pharmaceutical giants, and venture capitalist investors are beginning to increase their funding in microbiome studies and the development of products from such studies. The growing funding in fields such as synthetic biology, genomics of microbes, and precision medicine is enabling start-up firms and innovations in clinical areas.
Trends in the Human Microbiome Therapeutics Market
Evolution of Engineered Live Biotherapeutics
There are rising instances of research efforts to develop genetically-engineered living microbes as treatment options that can provide therapeutic molecules specifically. The use of engineered bacteria for cancer therapy, inflammation, and metabolic diseases has gained momentum owing to their potential ability to regulate immunity and metabolic processes.
Research Scope and Analysis
In the Human Microbiome Therapeutics Market report, the focus is placed on different segments, such as product type, therapeutic area, mode of delivery, microbiome, technology platform, and end user. Furthermore, the research sheds light on the trends that may influence the regional market, along with the latest advancements in technology and regulations, as well as possible investment opportunities.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
By Product Type Analysis
Live biotherapeutic products are predicted to lead the market with a projected market share of 34.8% in 2026 due to their increasing clinical efficacy, high regulatory interest, and increased use in gastrointestinal and oncology treatments. The defined microbial consortia and genetically modified microbes are seeing rising acceptance due to their specificity and better safety profile than traditional medicines. The fecal microbiota transplantation is still the most popular choice for treating recurrent Clostridium difficile infection, while postbiotics and synbiotics are experiencing rapid growth driven by the need for safer and more stable microbiome-related treatment options.
By Therapeutic Area Analysis
The Gastrointestinal Disorders segment is projected to capture around 39.2% of the market share by 2026, given that the microbiome is known to play a major role in CDI, IBD, Crohn's disease, and IBS. Clinical research that confirms the efficacy of microbiome-based treatment on gastrointestinal health will continue to fuel the market growth in the segment. The oncology segment is predicted to become the fastest-growing market segment owing to the growing application of microbiome modulation to improve immunotherapy efficacy and mitigate side effects of chemotherapy.
By Route of Administration Analysis
Oral administration accounts for an estimated 46.5% of the market share by 2026 because of the convenience of use and easy administration, along with high acceptance rates of capsules, tablets, and powdered microbiome preparations. Oral delivery systems are favored for treating chronic conditions and providing preventive health care solutions. Rectal administration continues to be relevant for the treatment of serious digestive diseases and fecal microbiota transplantations. Microbiome treatments administered through injections have gained popularity for precise systemic delivery of treatment for cancers and immune system disorders.
By Microbiome Type Analysis
Gut Microbiome Therapeutics Segment will dominate the industry with 58.7% of market share in 2026 due to the huge potential for research regarding gut bacteria and their involvement in various gastrointestinal disorders, metabolic disorders, disorders of the nervous system, and immune disorders. Increase in occurrence of gastrointestinal diseases and increase in awareness related to the health of the gut have contributed to the dominance of this segment. The skin and vaginal microbiome therapeutics segments are growing due to increased usage of microbiome-based dermatological and women's health drugs.
By Technology Platform Analysis
The metagenomic approach is expected to have a market share of about 31.4% by 2026 as it provides extensive microbial sequencing and biomarker discovery for developing therapeutic approaches. Metagenomics helps in precision medicine or personalized microbiome treatments with microbial ecosystem profiling. AI-driven microbiome discovery platforms are likely to show fast growth because of growing use of artificial intelligence technologies and predictive modeling in drug discovery. The CRISPR engineering platforms along with the synthetic biology approach are increasingly becoming popular for engineering microbial treatments.
By End User Analysis
Pharmaceutical & Biotech Companies are anticipated to be the key players in the market and capture around 42.1% market share by 2026 owing to growing investment in microbiome-based drug discovery, clinical trials, and business alliances. They have state-of-the-art R&D and regulatory skills needed for drug development on a large scale. Hospitals and specialist centers for Gastroenterology play a critical role in boosting the market for therapies based on microbiomes for GI disorders. Institutes involved in academics and research are anticipated to experience considerable growth in their contribution.
The Human Microbiome Therapeutics Market Report is segmented on the basis of the following:
By Product Type
- Live Biotherapeutic Products (LBPs)
- Single-strain therapeutics
- Multi-strain microbial consortia
- Genetically engineered microbes
- Fecal Microbiota Transplantation (FMT)
- Donor-derived FMT
- Oral capsule FMT
- Colonoscopic / enema-based FMT
- Postbiotics
- Microbial metabolites
- Cell-free microbial extracts
- Synbiotics
- Probiotic-prebiotic combinations
- Personalized synbiotic formulations
- Phage-based Therapeutics
- Bacteriophage therapies
- Precision pathogen-targeting phages
- Microbiome-derived Small Molecules
- Metabolite-derived drugs
- Immune-modulating compounds
By Therapeutic Area
- Gastrointestinal Disorders
- Clostridioides difficile infection (CDI)
- Inflammatory bowel disease (IBD)
- Crohn's disease
- Ulcerative colitis
- Irritable bowel syndrome (IBS)
- Oncology
- Immunotherapy response enhancement
- Chemotherapy support
- Metabolic Disorders
- Obesity
- Type 2 diabetes
- NAFLD / NASH
- Neurological Disorders
- Autism spectrum disorder
- Parkinson's disease
- Autoimmune & Immune Disorders
- Rheumatoid arthritis
- Psoriasis
- Dermatological Disorders
- Women's Health
- Bacterial vaginosis
- Urinary tract infections
By Route of Administration
- Oral
- Rectal
- Enema delivery
- Colonoscopic delivery
- Injectable
- Topical
By Microbiome Type
- Gut Microbiome Therapeutics
- Skin Microbiome Therapeutics
- Oral Microbiome Therapeutics
- Vaginal / Urogenital Microbiome Therapeutics
By Technology Platform
- Metagenomics
- Metabolomics
- Synthetic Biology Platforms
- CRISPR-based Engineering
- AI-based Microbiome Discovery Platforms
By End User
- Hospitals & Clinics
- Specialty Gastroenterology Centers
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
Regional Analysis
Leading Region in the Human Microbiome Therapeutics Market
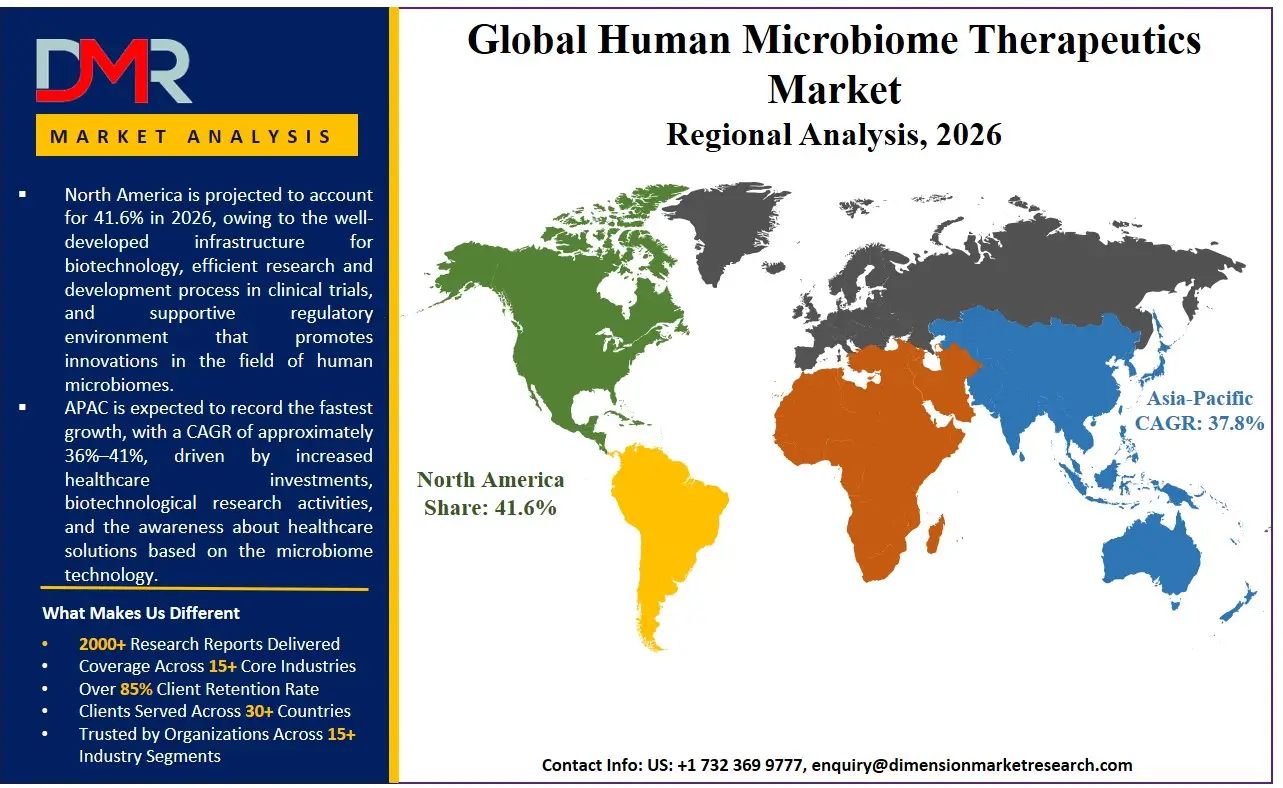
North America is expected to lead the Human Microbiome Therapeutics Market in terms of market share of 41.6% in 2026, owing to the well-developed infrastructure for biotechnology, efficient research and development process in clinical trials, and supportive regulatory environment that promotes innovations in the field of human microbiomes. North America holds the potential for growth due to increased approvals by the FDA for microbiome-based products, heavy spending on healthcare services, and significant presence of biotech firms engaged in developing live biotherapeutic products and artificial intelligence-based drug discovery systems.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Fastest Growing Region in the Human Microbiome Therapeutics Market
The Asia-Pacific region is forecasted to hold the most promising growth prospect for the Human Microbiome Therapeutics Market, considering the increased healthcare investments, biotechnological research activities, and the awareness about healthcare solutions based on the microbiome technology. There is significant development happening in terms of the genomic studies and precision medicine in countries like China, Japan, South Korea, and India. The prevalence of digestive and metabolic diseases, enhanced manufacturing capabilities in the pharmaceutical industry, and financial backing from the governments in their biotechnological innovations are aiding the growth of the regional market.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
Human Microbiome Therapeutics Market is highly competitive and innovative in nature and involves extensive clinical trials, partnerships, and significant investments made towards synthetic biology and artificial intelligence for discovering new microbes. Companies operating within the market have been making considerable efforts in increasing their clinical pipelines, getting approvals from regulatory bodies, and working on precision microbiome therapies against various disease conditions. Entry barriers within this market are quite steep owing to the complexity associated with regulation, clinical trials, and manufacturing processes. An increasing number of companies are preferring to enter into partnerships with research institutions, pharma companies, and AI platforms.
Some of the prominent players in the global Human Microbiome Therapeutics are:
- Seres Therapeutics
- Ferring Pharmaceuticals
- Vedanta Biosciences
- Finch Therapeutics
- MaaT Pharma
- BiomX
- Enterome
- 4D Pharma
- Assembly Biosciences
- Synlogic
- Second Genome
- Locus Biosciences
- Eligo Bioscience
- Rebiotix
- Seed Health
- Pendulum Therapeutics
- Yakult Honsha
- IFF
- Microbiotica
- BiomeBank
- Other Key Players
Recent Developments
- In April 2026, Nestlé became a member of Microbiome Therapeutics Innovation Group (MTIG), which is an independent body that strives to advance FDA-approved microbiome therapeutics and enhance access to cutting-edge microbiome drugs for patients. Speaking on behalf of Nestlé, Krys Araujo-Torres, MD, MPP, the Global Head of MSRO at Nestlé Health Science, will be the company's chief delegate in MTIG. According to Nestlé, the membership strengthens its dedication to innovation driven by science and collaboration towards safe and effective microbiome therapeutics.
- In February 2026, Positive data from the Phase 1b trial were reported by Microbiotica on the use of MB310 for treating patients with UC. In the First-in-Human COMPOSER-1 trial, it was established that the drug administered orally once daily had favorable safety, tolerability, and successful engraftment of bacteria. The novel live biotherapeutic product also demonstrated significant superiority over the placebo in terms of efficacy outcomes. More importantly, MB310 demonstrated clinical remission outcomes, suggesting its potential to modify the disease.
- In January 2026, ASM Health was introduced by the American Society for Microbiology, an initiative that aims to turn research into healthcare innovation. Four scientific innovations have been introduced under ASM Health to help advance resilient health through cooperation within academic, industrial, governmental, and nonprofit organizations. ASM Health is expected to accelerate research, clinical development, and regulatory advancement to develop innovative diagnostics, treatments, and prevention methods. According to Dr. Dev Mittar, Ph.D., ASM Health will be working towards turning rapid advances in microbiology into robust healthcare systems.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 1.9 Bn |
| Forecast Value (2035) |
USD 22.8 Bn |
| CAGR (2026-2035) |
32.1% |
| Historical Period |
2021 – 2025 |
| Forecast Period |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Segments Covered |
By Product Type, By Therapeutic Area, By Route of Administration, By Microbiome Type, By Technology Platform, By End User |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
Frequently Asked Questions
How big is the Human Microbiome Therapeutics Market?
▾ The Human Microbiome Therapeutics Market size is expected to reach USD 1.9 billion by 2026 and is projected to reach USD 22.8 billion by the end of 2035.
What is the CAGR of the Human Microbiome Therapeutics Market from 2026 to 2035?
▾ The market is growing at a CAGR of 32.1 percent over the forecasted period.
What factors are driving the growth of the Human Microbiome Therapeutics Market?
▾ Growing Occurrence of Chronic and GI Diseases, and more are the factors driving the growth of the Human Microbiome Therapeutics Market.
What are the major trends in the Human Microbiome Therapeutics Market?
▾ Evolution of Engineered Live Biotherapeutics, and more are some of the major trends in the market.
Who are the key players in the Human Microbiome Therapeutics Market?
▾ Some of the key players in the Human Microbiome Therapeutics Market include BiomX, MaaT Pharma, 4D Pharma, and more.
How is the Human Microbiome Therapeutics Market segmented?
▾ The Human Microbiome Therapeutics Market is segmented by product type, therapeutic area, route of administration, microbiome type, technology platform, end user.
Which region held the largest share of the Human Microbiome Therapeutics Market in 2026?
▾ North America is set to lead the Human Microbiome Therapeutics market with an estimated 41.6% share in 2026.
Which region is expected to grow the fastest in the Human Microbiome Therapeutics Market?
▾ Asia Pacific is the fastest-growing region in the Human Microbiome Therapeutics market during the forecast period.