Market Overview
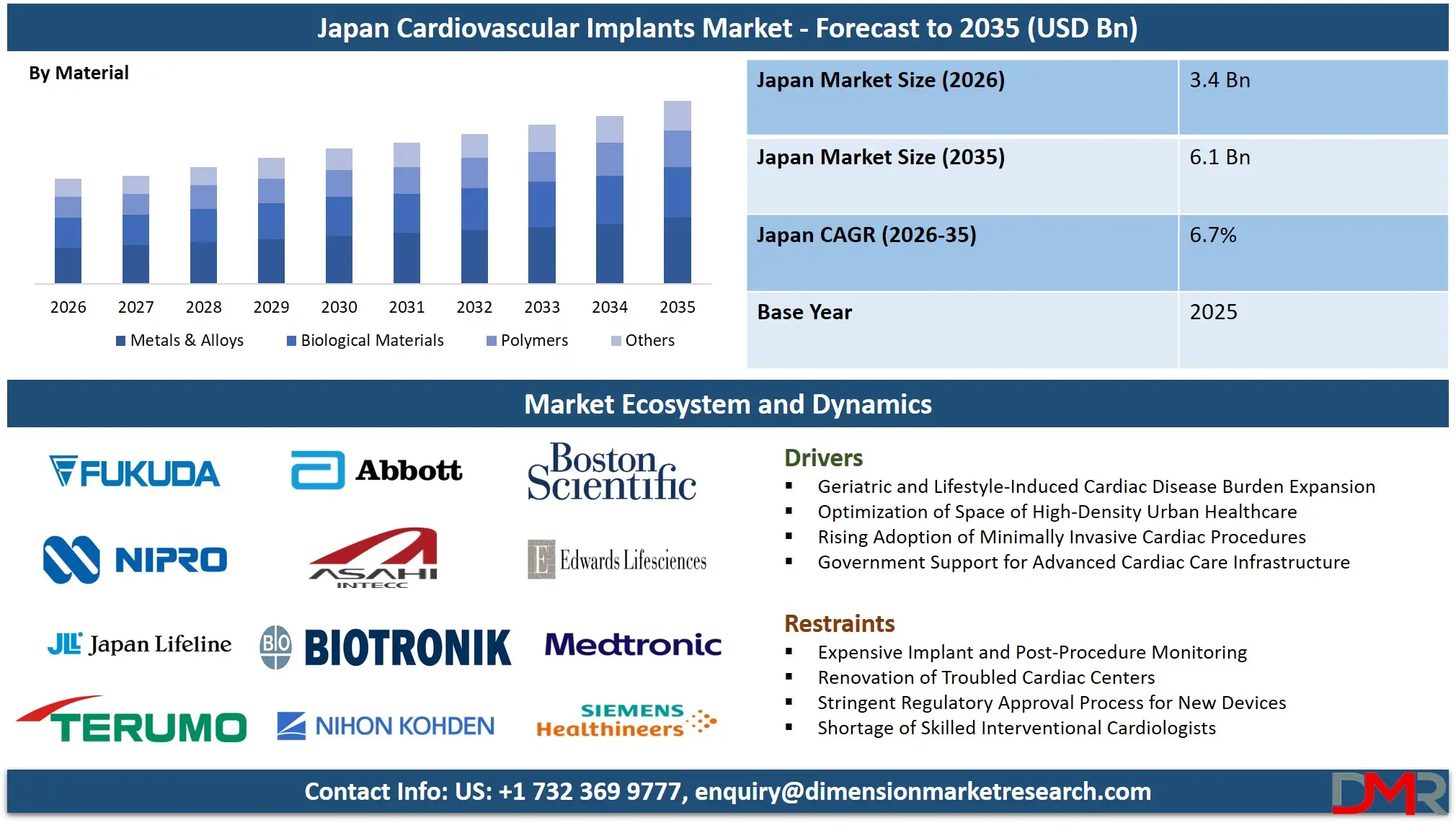
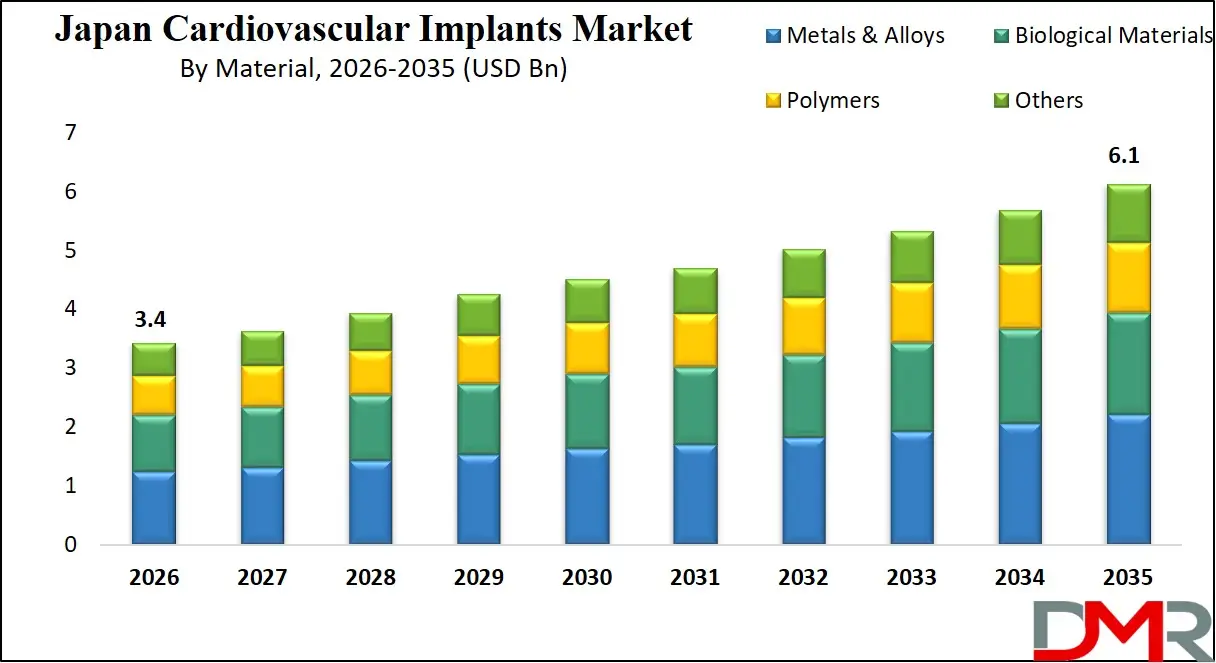
The Japan Cardiovascular Implants Market size is expected to grow from USD 3.4 billion in 2026 to USD 6.1 billion in 2035, with a compound annual growth rate (CAGR) of 6.7% during the forecast period from 2026 to 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Cardiovascular implants are medical equipment applied in surgery and interventional operations to treat cardiac diseases like arrhythmias, coronary artery disease, and heart failure. They are coronary stents, implantable cardioverter defibrillators (ICDs), heart valves and pacemakers as well as cardiac resynchronization therapy (CRT) devices. They improve patient survival, cardiac functioning, and decrease hospital readmission. These implants are commonly used in Japanese hospitals, specialty clinics and cardiac catheterization labs.
The Japanese cardiovascular implants market is highly mature and is led by an aging population, precision engineering and high-quality healthcare facilities. The demand of next-generation bioresorbable stents and leadless pacemakers is being fueled by urban medical centers. The use of robotics and smart implant is on the rise especially in complex angioplasty and open heart surgery. High levels of medical device research and development and growth of outpatient interventional cardiology are also typical of the industry.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Japan Cardiovascular Implants Market: Key Takeaways & Other Influencing Factors
- Market Size Insights: The Japan Cardiovascular Implants Market is projected to reach USD 6.1 billion by 2035, growing from an estimated USD 3.4 billion in 2026, witnessing growth at a CAGR of 6.7% during the forecasted period.
- Coronary Stents dominance: Coronary stents dominate the product market with approximately 47.0% market share in 2026 because of the high prevalence of myocardial ischemia and large-scale usage in angioplasty interventions.
- Angioplasty as Primary Procedure Driver: Angioplasty leads procedures with almost a 35.0% share with the growing use of minimally invasive interventions and high patient throughput procedures in cardiac cath labs.
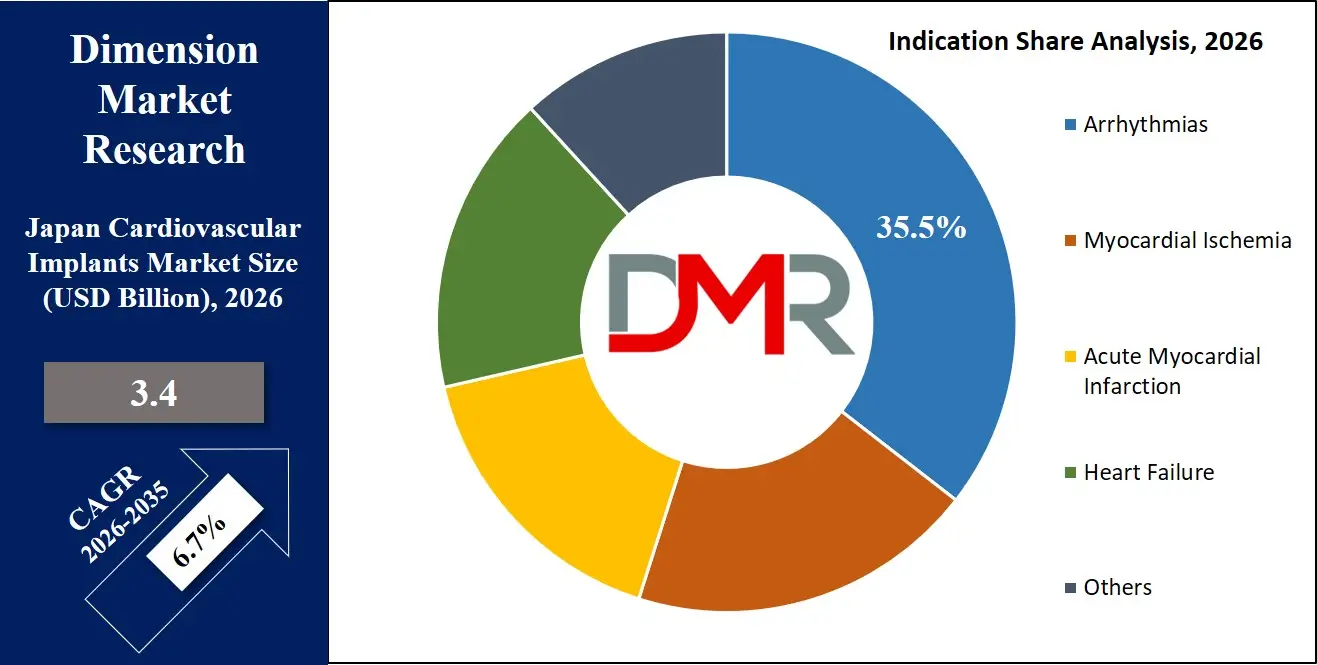
- Arrhythmias as Key Signal: Arrhythmias represent the highest share of implant procedures, with the rising support of ICD and pacemaker implantation.
- Automation & Smart Implant Acceleration: More than 60% of Japanese cardiac centers that have implemented an IoT-based remote monitoring are enhancing demand in advanced cardiovascular implants, which leads to better patient outcomes and device durability.
- Infrastructure Scale (MHLW): Japan has 1,245 hospitals that have interventional cardiology departments and an estimated 120,000 catheterization procedures per year, 13.2 million outpatient cardiology visits, and 4 new advanced cardiac centers scheduled by 2030, and cardiovascular care infrastructure is growing well.
- Digital Adoption (JETRO): 63% of cardiology clinics are equipped with remote monitoring systems, and 12.3% of cardiac follow-ups are done through telecardiology, which is a fast-growing trend towards smart cardiac implant management.
Impact of the Iran conflict on the Japan Cardiovascular Implants Market
The Iran war is further increasing the prices of the world supply chains and raw materials and indirectly the cost of the cardiovascular implants in Japan through the increased cost of medical-grade metals and logistics. Nevertheless, the long-term demand does not decrease as Japan is still on the way to implement next-generation bioresorbable stents and leadless pacemakers to improve patient outcomes and minimize reintervention costs.
Japan Cardiovascular Implants Market: Use Cases
- Emergency Angioplasty and STEMI Care: Coronary stents facilitate speedy revascularization of acute myocardial infarction, which allows the restoration of blood flow optimization and prompt recovery of patients within the emergency cardiology networks in Japan.
- Heart Failure & CRT Therapy: Cardiac resynchronization therapy implants help to synchronize ventricular contractions, which increases the efficiency of the cardiac output and survival of chronic heart failure patients.
- Arrhythmia Management and Sudden Cardiac Arrest Prevention: ICDs and pacemakers are used to detect and correct abnormal heart rhythm to offer effective long-term rhythm control and sudden cardiac death prevention.
- Outpatient and Ambulatory Surgical Centers: Outpatient and Ambulatory Surgical Centers provide minimal compatible implant systems to increase the capacity of the procedure, the patient throughput, and the efficiency of the overall cardiovascular care.
Japan Cardiovascular Implants Market: Market Dynamics
Driving Factors in the Japan Cardiovascular Implants Market
Geriatric and Lifestyle-Induced cardiac disease burden expansion
The increasing number of elderly people in Japan is placing the demand of advanced cardiovascular implants that can handle large numbers of complicated heart cases. To improve the success of the procedure, accuracy of diagnosis and long-term patient management, hospitals are investing in drug-eluting stents, MRI-compatible pacemakers, and implantable loop recorders. The trend towards outpatient interventional cardiology is also providing pressure on miniaturized and long-lasting implantable equipment.
Optimization of Space of High-Density Urban Healthcare
In Japan, cardiac centers are being compelled to use small, efficient cath labs with integrated implant storage facilities due to the lack of space to expand hospitals as well as high cost of real estates. The use of modular inventory and just-in-time delivery of implants is on the rise to achieve the maximum utilization of the available space and the efficient management of the devices.
Restraints in the Japan Cardiovascular Implants Market
Expensive Implant and Post Procedure Monitoring
Implementation of new technology in the cardiovascular implants, such as leadless pacemakers and CRT-D devices is capital intensive. The expenses related to the implantation of the devices, their long-term monitoring, and the replacement of batteries can restrict their use in smaller clinics and rural hospitals.
Renovation of Troubled Cardiac Centers
Most of the Japanese cardiac centers have old infrastructure that is not adequately fitted to accommodate modern implant systems and intra-operative room needs. Modernization of these facilities involves complicated redesign, unproductive time, and related high expenses, which can limit market growth.
Opportunities in the Japan Cardiovascular Implants Market
Cold Chain and Biologic Implant Logistics Development
The greater stress of temperature controlled transportation of biological heart valves and tissue-engineered implants is opening up prospects on special logistics solutions. Cardiac centers are also moving towards the use of inventory management systems and intelligent tracking solutions to improve the process of tracing devices and controlling inventory.
Expansion of Ambulatory Surgical Centers & Smart Cath Labs
An increasing demand is seen in scalable implant systems and computer-assisted procedure planning with the rise in the use of ambulatory surgical centers. The introduction of AI-driven image guidance, real-time device navigation, and intelligent cath lab integration is opening up new business opportunities in the Japanese cardiovascular infrastructure.
Trends in the Japan Cardiovascular Implants Market
Use of Leadless Implants and Boresorbable Implants
The movement towards leadless pacemakers and bioresorbable vascular scaffolds is growing in an attempt to enhance long-term patient outcomes. These equipments expand the flexibility of the procedures, minimise lead-associated problems, and facilitate quicker recovery of the procedure in cardiac centres of the present day era.
Implementation of Digital Cardiac Monitoring Technologies
Cardiologists are going digital, using remote patient monitoring systems, AI-based arrhythmia detection, and real-time device analytics. The technologies are combined with implantable devices to improve the management of patients, increase the duration of device life, and provide predictive maintenance in the changing cardiology environment in Japan.
Japan Cardiovascular Implants Market: Research Scope and Analysis
Product Type Analysis
The Japan cardiovascular implants market is dominated by coronary stents which control approximately 47.0% of the market in 2026 because of the extensive application in angioplasty and acute coronary syndromes. This is followed by ICDs and pacemakers owing to the increasing prevalence of arrhythmia. There is growing popularity of the use of heart valves and CRT devices in the management of heart failure and other products such as peripheral stents and others are used in niche vascular applications. The presence of constant innovation in the technologies of drug-eluting and bioresorbable stents also contributes to the superiority of coronary stents in the Japanese market.
By Material Analysis
Metals & alloys lead with approximately 41.0% share in 2026, favored for strength and biocompatibility in stents and pacemaker cans. Polymers are used in bioresorbable scaffolds and catheter components. Biological materials dominate tissue heart valves. Other materials include absorbable and composite variants. The shift toward hybrid materials combining metal strength with polymer flexibility is emerging as a key trend in next-generation cardiovascular implants.
By Procedure Type Analysis
Angioplasty is expected to have a share of approximately 35.1% in 2026 because of the adoption of minimal invasiveness and large volumes of procedures. Complex cases like multivessel disease and valve replacement that need direct surgical access still have large share in open heart surgery. The rising popularity of same-day discharge and outpatient angioplasty is also increasing the proportion of minimally invasive surgery in Japan.
By Indication Analysis
Arrhythmias represent the largest indication, driving ICD and pacemaker demand. Myocardial ischemia and acute myocardial infarction fuel coronary stent usage.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Heart failure patients benefit from CRT and ventricular assist devices. Other indications include congenital and valvular diseases. An aging Japanese population with multiple cardiac comorbidities is expected to sustain long-term demand across all major indication categories.
By End User Analysis
Hospitals are the end-users who are the most dominated with the largest amount of procedures and high-tech cath labs. Specialty clinics specialize in regular pacemaker follow-ups. Low-risk angioplasty is finding new homes in ambulatory surgical centers. Cardiac catheterization labs are still vital to interventional practices. Other ones are academic and research institutions. The growth of hybrid operating rooms and outpatient cath labs is slowly changing the end-user environment of the Japanese cardiovascular care industry.
The Japan Cardiovascular Implants Market Report is segmented on the basis of the following:
By Product Type
- Coronary Stents
- Implantable Cardioverter Defibrillators (ICDs)
- Heart Valves
- Implanted Cardiac Pacemakers
- Cardiac Resynchronization Therapy (CRT) Devices
- Peripheral Stents
- Others
By Material
- Metals & Alloys
- Polymers
- Biological Materials
- Others
By Procedure Type
- Angioplasty
- Open Heart Surgery
By Indication
- Arrhythmias
- Myocardial Ischemia
- Acute Myocardial Infarction
- Heart Failure
- Others
By End-User
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratories
- Others
Impact of Artificial Intelligence in the Japan Cardiovascular Implants Market
Artificial intelligence is changing the Japan cardiovascular implants market, as now it allows planning procedures smarter, choosing devices more optimally, and navigate during the process of implantation in real-time because of the AI-based technologies that minimize the use of contrasts and minimize the amount of time spent on fluoroscopy. Robotic-assisted interventions have also been used in combination with AI to enhance throughput, precision, and operational efficiency in highly precision-focused Japanese cath labs.
Moreover, AI supports predictive analysis of device failures and remote patient monitoring based on continuous telemetry data to reduce occurrences of cardiac accidents as well as improve the scheduling of battery replacement and the overall safety and scalability of cardiac implantable electronic devices.
Japan Cardiovascular Implants Market: Competitive Landscape
The Japan cardiovascular implants market is moderate in terms of level of consolidation and competition with global and regional firms offering full range of cardiac equipment and interventional solutions. Competition is based on innovation, clinical evidence, and end-to-end implant systems, including leadless pacemakers and fully bioresorbable stents.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The greatest needs are found in high-volume urban cath labs where seismic compliance, engineering accuracy, and the long-term reliability of devices are the distinguishing factors. Other competencies that companies employ to respond to different patient and hospital needs are customization, cost efficiency and ease of implantation.
Some of the prominent players in the Japan Cardiovascular Implants Market are:
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- Edwards Lifesciences Corporation
- Terumo Corporation
- Johnson & Johnson
- Biotronik SE & Co. KG
- B. Braun Melsungen AG
- LivaNova PLC
- MicroPort Scientific Corporation
- W. L. Gore & Associates, Inc.
- Getinge AB
- Nipro Corporation
- Asahi Intecc Co., Ltd.
- Nihon Kohden Corporation
- Fukuda Denshi Co., Ltd.
- Japan Lifeline Co., Ltd.
- Canon Medical Systems Corporation
- Siemens Healthineers
- Koninklijke Philips N.V.
- Other Key Players
Recent Developments in the Japan Cardiovascular Implants Market
- July 2025: Abbott Laboratories received regulatory approval in Japan for its TriClip transcatheter tricuspid valve repair system, strengthening its structural heart portfolio and expanding minimally invasive treatment options in the country.
- March 2025: Edwards Lifesciences Corporation expanded its transcatheter mitral and tricuspid therapies (TMTT), including PASCAL and EVOQUE systems, supporting growth in structural heart interventions across developed markets such as Japan.
- February 2025: Medtronic plc reported steady growth in Japan driven by increasing adoption of its Avalus surgical aortic valve, reflecting rising demand for advanced heart valve replacement technologies.
- October 2024: Boston Scientific Corporation and Terumo Corporation advanced cardiac rhythm management and interventional cardiology solutions in Japan, focusing on minimally invasive devices and next-generation implant technologies.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 3.4 Bn |
| Forecast Value (2035) |
USD 6.1 Bn |
| CAGR (2026–2035) |
6.7% |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Product Type (Coronary Stents, Implantable Cardioverter Defibrillators (ICDs), Heart Valves, Implanted Cardiac Pacemakers, Cardiac Resynchronization Therapy (CRT) Devices, Peripheral Stents, and Others), By Material (Metals & Alloys, Polymers, Biological Materials, and Others), By Procedure Type (Angioplasty and Open Heart Surgery), By Indication (Arrhythmias, Myocardial Ischemia, Acute Myocardial Infarction, Heart Failure, and Others), By End-User (Hospitals, Specialty Clinics, Ambulatory Surgical Centers, Cardiac Catheterization Laboratories, and Others) |
| Country Coverage |
Japan |
| Prominent Players |
Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, Edwards Lifesciences Corporation, Terumo Corporation, Johnson & Johnson, BIOTRONIK SE & Co. KG, B. Braun Melsungen AG, LivaNova PLC, MicroPort Scientific Corporation, W. L. Gore & Associates, Inc., Getinge AB, Nipro Corporation, Asahi Intecc Co., Ltd., Nihon Kohden Corporation, Fukuda Denshi Co., Ltd., Japan Lifeline Co., Ltd., Canon Medical Systems Corporation, Siemens Healthineers, Koninklijke Philips N.V., and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users), and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the Japan Cardiovascular Implants Market?
▾ The Japan Cardiovascular Implants Market size is estimated to have a value of USD 3.4 billion in 2026 and is expected to reach USD 6.1 billion by the end of 2035.
What is the growth rate in the Japan Cardiovascular Implants Market in 2026?
▾ The market is growing at a CAGR of 6.7% over the forecasted period of 2026.
Who are the key players in the Japan Cardiovascular Implants Market?
▾ Some of the major key players in the Japan Cardiovascular Implants Market are Abbott Laboratories, Medtronic plc, Boston Scientific Corporation, Edwards Lifesciences Corporation, Terumo Corporation, and Nipro Corporation, and many others.