What is the Rare Disease Gene Therapy Market Size?
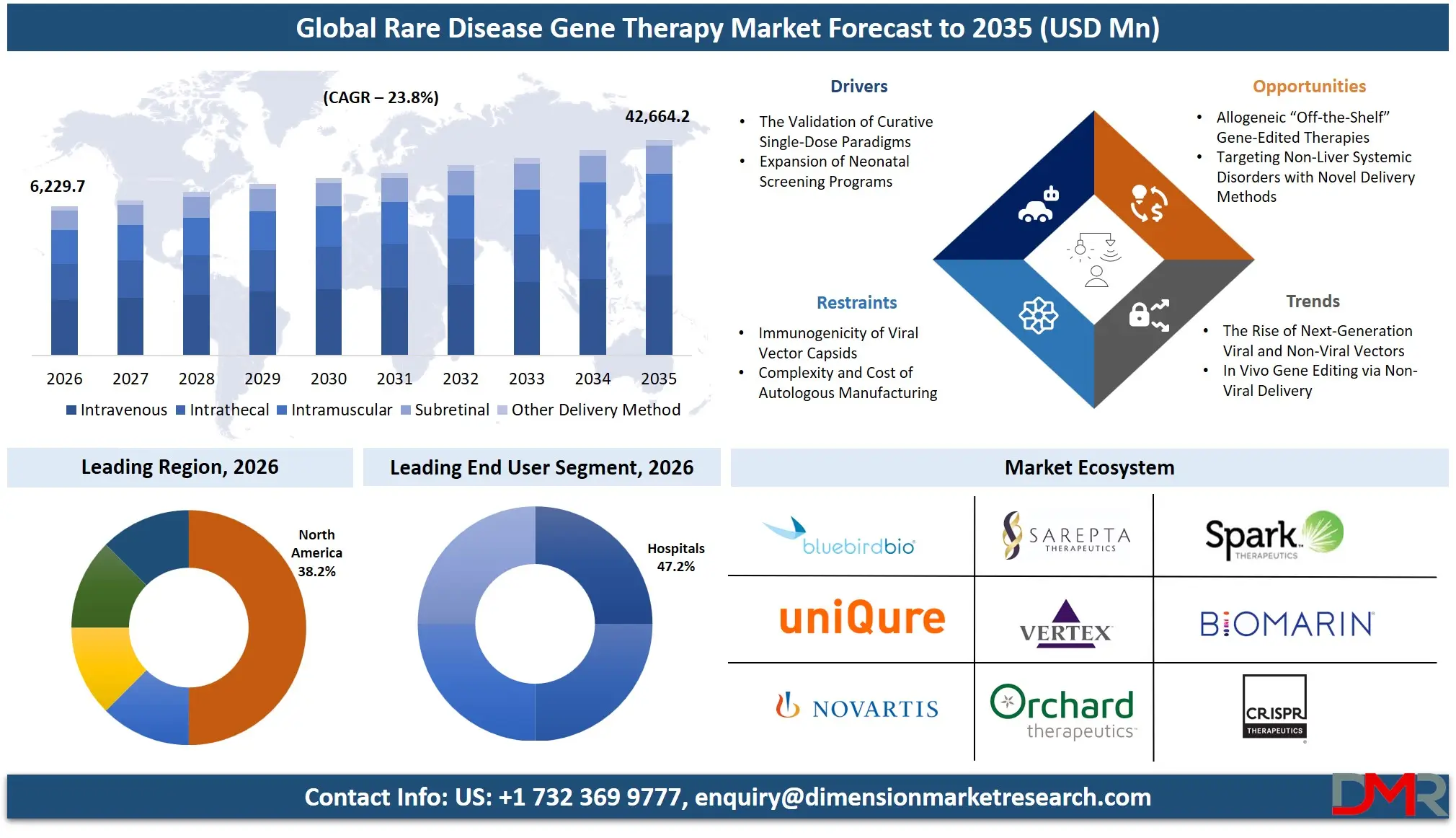
The Global Rare Disease Gene Therapy Market is expected to reach a value of USD 6,229.7 million in 2026, and it is further anticipated to reach USD 42,664.2 million by 2035, growing at a CAGR of 23.8% during the forecast period.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The rare disease gene therapy market has been expanding exponentially, moving from a theoretical scientific field into approved, paradigm shifting one-time treatments. The market spans in vivo, ex vivo, gene editing and RNA-based therapies, targeting the genetic origin of the disease underlying intractable conditions in the hematological, neuromuscular, metabolic and ophthalmic fields.
With the growing knowledge of the human genome, and the development of viral and non-viral vectors, the need for specialized therapeutic platforms is ever stronger. Academic medical centers and specialty treatment centers are the most common settings for the treatment, and adeno-associated virus (AAV) vectors and lentiviral vectors are the most popular vectors because of their demonstrated high transduction efficiency and tropism. The biotechnology and pharmaceutical industry is a major contributor, with the creation of safe, compliant, and highly scalable manufacturing ecosystems to cater to patient populations that are in dire need of services.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
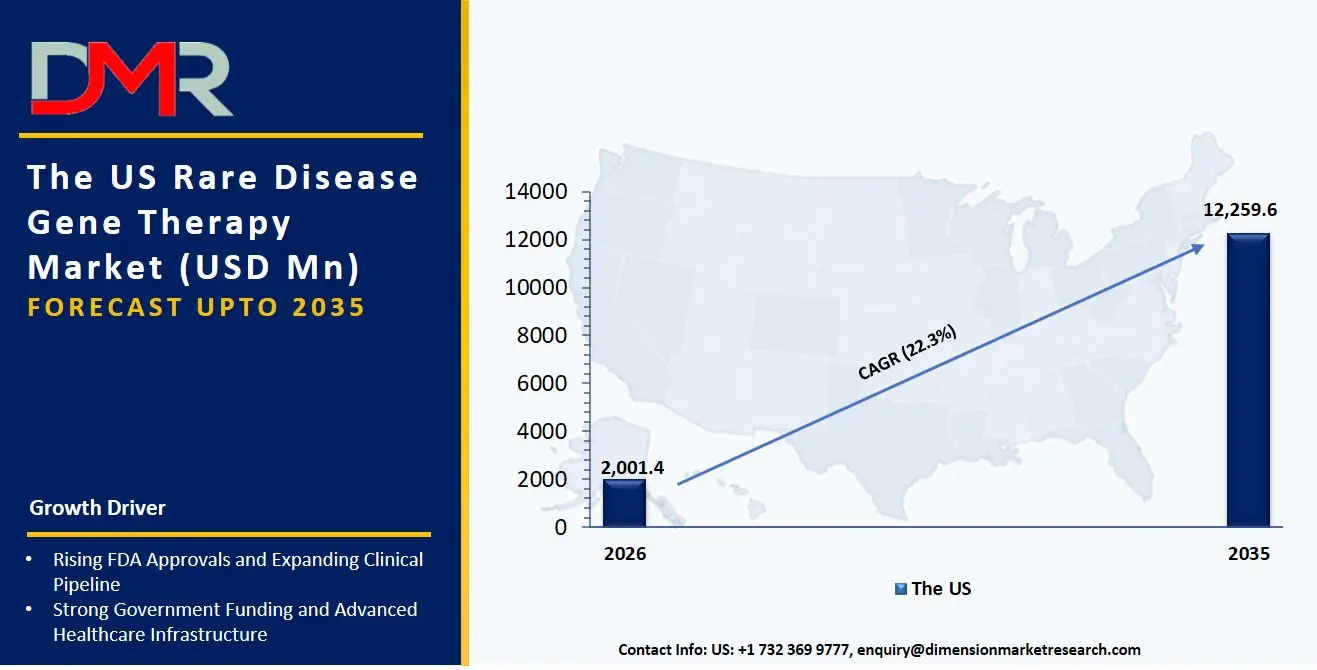
The US Rare Disease Gene Therapy Market
The US Rare Disease Gene Therapy Market is projected to reach USD 2,001.4 million in 2026 at a compound annual growth rate of 22.3% over its forecast period, culminating in a value of USD 12,259.6 million by 2035.
The U.S. remains the largest and most mature market for rare disease gene therapy because of the supportive regulatory environment for orphan drug designation, and the tight clustering of leading biotech companies. The market has been typified by high demand for In Vivo Gene Therapy, whereby organizations are aimed at delivering functional genes directly into target organs like the retina or central nervous system. Notably, the clinical translation of the Gene Editing Therapy based on CRISPR is creating a parallel demand for novel delivery strategies, such as Subretinal and Intrathecal delivery to ensure tissue-specific editing under the control of a therapeutic probiotic.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The Europe Rare Disease Gene Therapy Market
The Europe Rare Disease Gene Therapy Market is estimated to be valued at USD 1,847.2 million in 2026 and is further anticipated to reach USD 12,354.4 million by 2035 at a CAGR of 23.4%. The regulatory environment, such as the EMA's PRIME scheme and the national healthcare assessments that have advanced, has an important influence on the European market and creates a demand for sophisticated Health Economics and Outcomes Research that demonstrates high upfront costs. Accelerated growth of Ex Vivo Gene Therapy for hematological disorders is also being experienced in the region as treatment centers in Germany, France, and the UK are trying to strike a balance between complex autologous manufacturing logistics and centralized care delivery. Moreover, cross-border efforts are pushing therapy developers to establish patient-specific registries and follow-up plans to deliver long-term efficacy and safety data across European health care systems.
The Japan Rare Disease Gene Therapy Market
The Japan Rare Disease Gene Therapy Market is projected to be valued at USD 493.6 million in 2026. It is further expected to witness robust growth, holding USD 3,150.3 million in 2035 at a CAGR of 20.9%. The Japanese market is quite unique with a national policy aimed at becoming the first nation to develop regenerative medicine and gene therapies to address the country's aging population and prevalent population of specific inherited metabolic disorders. Much of the pioneering research falls under the umbrella of Gene Editing Therapy and Ex Vivo Gene Therapy, as new CRISPR-based tools in inherited metabolic diseases are developed by the leading universities and research institutes and brought to the clinic. There is also a critical need to tie in with the local market and fill the niche between traditional hospital systems and advanced therapy manufacturing suites for lentiviral vector processing and non-viral vector delivery systems.
Key Takeaways
- Market Size & Forecast: The Global Rare Disease Gene Therapy market is projected to reach USD 6,229.7 million in 2026, expanding dramatically to USD 42,664.2 million by 2035, driven by both validated clinical proof-of-concept and required technology modernization of viral vector manufacturing.
- Growth Rate & Outlook: The global market is anticipated to grow at a CAGR of 23.8% owing to the urgent need for specialized GMP manufacturing capability as well as the increased complexity of handling long-term patient follow-up registries for durable gene expression.
- Primary Growth Drivers: There are key growth drivers such as the transition from chronic disease management to the potential single administration cure, novel AAV capsid engineering to ensure avoidance of pre-existing neutralizing antibodies and the integration of accelerated regulatory pathways, which require specialized regulatory consulting and clinical trial design.
- Key Market Trends: There are key growth drivers such as the transition from chronic disease management to the potential single administration cure, novel AAV capsid engineering to ensure avoidance of pre-existing neutralizing antibodies and the integration of accelerated regulatory pathways, which require specialized regulatory consulting and clinical trial design.
- By Delivery Method Analysis: Intravenous infusion is expected to dominate systemic disease applications due to its broad biodistribution profile. As more and more ex vivo therapies have been developed, seamless handling protocols are being developed, which requires professional manufacturing and administration knowledge.
- By Disease Type Analysis: Hematological Disorders and Neuromuscular Disorders are the most promising verticals with robust clinical validation and high unmet need. Metabolic Disorders is the fastest-growing area and in vivo gene therapy requires solid clinician training and newborn screening infrastructure integration.
- Regional Leadership: North America is poised to dominate this market with 38.2% of the market share in 2026 owing to its well-established biotechnology funding ecosystem and the utilization of this infrastructure to its full potential makes it a major player in this market.
What is the Rare Disease Gene Therapy?
Rare Disease Gene Therapies are specialized therapeutic products provided by innovation-driven biopharmaceutical manufacturers, contract development and manufacturing organizations (CDMOs), and specialty distributors that help healthcare systems find a solution for the underlying cause of genetic diseases. These treatments, unlike small molecule chronic therapies (the palliative management of symptoms), are related to the permanent correction of disease pathology. The three types of gene therapy include In Vivo Gene Therapy, which directly infuses a functional gene into a targeted organ, Ex Vivo Gene Therapy, which genetically modifies a patient's own cells outside their body before re-engrafting, and Gene Editing Therapy, which specifically edits a defective gene at its native site. There are more than 7,000 rare diseases that have been identified, and gene therapy is required to attain long-term clinical efficacy, immunologic tolerance and manufacturing scalability to realize the potential for a paradigm shift from disease to functional cure, rather than lifelong medical management.
Use Cases
- Hemoglobinopathy Correction in Hematological Disorders: In Hematological Disorders, specialized cell processing suites and logistics providers are contracted by treatment centers to support Ex Vivo Gene Therapy by transforming the patient's own HSC with lentiviral vectors containing functional beta-globin, resulting in transfusion independence for patients with beta-thalassemia and sickle cell disease.
- Motor Neuron Preservation in Neuromuscular Disorders: In Vivo Gene Therapy and Intrathecal Delivery Services at pediatric hospitals administer their AAV9 vectors containing the SMN1 gene to intercept the fatal course of spinal muscular atrophy (SMA) in newborns by a one-time infusion.
- Vision Restoration in Ophthalmic Disorders: In ophthalmic disorders, vision restoration, subretinal delivery is employed in ophthalmic surgical centres to introduce a functional RPE65 gene to retinal pigment epithelium (RPE) cells and restore functional vision to patients with inherited retinal dystrophy.
- Metabolic Pathway Reconstitution: Gene Editing Therapy platforms are being used by Research institutes to develop lipid nanoparticle-based delivery of CRISPR machinery to the liver to knock down a gene for a misfolded protein that causes TTR Amyloidosis and correct the metabolic pathway in a single treatment.
How AI is Transforming the Rare Disease Gene Therapy Market?
AI is changing the rare disease gene therapy landscape by accelerating the process of vector engineering, as well as enhancing operational efficiency in clinical trials. Within capsid design and optimization, AI can now be used to automatically create new AAV serotypes with improved tissue tropism and immune escape, significantly reducing the risk of liver toxicity in patients and increasing the number of patients that could benefit from treatment. Meanwhile, AI-powered features in clinical manufacturing allow biotech firms to better control production yields by predicting critical quality attributes, forecasting bioreactor performance, and suggesting optimal purification parameters to reinforce scalable manufacturing methods.
Patient identification and diagnosis projects are also revolving around AI. In the realm of diagnosis, AI-driven NLP agents are employed to roll the electronic health record and continuously screen for patients who have not received a diagnosis and whose clinical history matches the genetic characteristics of rare metabolic or neuromuscular disorders, thereby ending the diagnostic journey and hastening their participation in clinical trials.
Market Dynamics
Key Drivers in the Global Rare Disease Gene Therapy Market
The Validation of Curative Single-Dose Paradigms
Chronic rare diseases impose a high lifetime economic burden on global health systems, potentially costing millions of dollars for each patient in supportive care. The clinical results of approved therapies that prove multi-year efficacy from a single administration cycle provide a value proposition that fundamentally changes conventional pharmaco-economic models. That's driving a payers' shift toward provider annuity contracts on top of rejecting a product outright for the initial price tag. These innovative payment models help to speed up the journey of patients and reduce the budgetary burden on national health systems.
Expansion of Neonatal Screening Programs
A growing number of developed and developing countries are implementing mandatory newborn genomic sequencing programs. Rare disease diagnosis is now happening in days, instead of months or years, with the potential of diagnosing a patient with rare disease before irreversible pathological damage has occurred, effectively creating a new patient population that can be immediately helped. This complexity coupled with just-in-time therapy delivery at gene therapy centers is driving the need for specialized integrated care networks to handle the logistics of diagnostics, cell collection and vector preparation within a narrow therapeutic window, especially for metabolic and neuromuscular disorders.
Restraints in the Global Rare Disease Gene Therapy Market
Immunogenicity of Viral Vector Capsids
Many people in the human population have pre-existing, neutralizing antibodies against various natural AAV serotypes, as a direct result of exposure to common cold viruses. This pre-existing immunity is a major hurdle to treatment; even low levels of antibodies can make an intravenous or systemic gene therapy ineffective, or even produce harmful immune response against the liver. Excluding seropositive patients can affect the exclusion of up to 50% of the clinical trial population. A range of countermeasures, such as plasmapheresis or engineered immune-evasive capsids, are available to overcome this barrier, but are expensive, risky and still in their early stages, so that only a portion of the diagnosed population can access the current generation of therapies.
Complexity and Cost of Autologous Manufacturing
In contrast, ex vivo gene therapies rely on a vein-to-vein supply chain that is very fragile and expensive. It is a process of collecting cells at an apheresis center, sending the cells with cryopreservation to a centralized GMP cell modification facility and sending the living drug product back to the treatment center. One logistical mistake, temperature change or sterility violation could ruin the batch, and a patient receiving conditioning chemotherapy could end up with no therapeutic product. This complexity is decoupled from scaling economies, because the manufacturing of a therapy is a very expensive enterprise, with the raw materials and quality control testing for a single batch of therapy costing hundreds of thousands of dollars, thus limiting the growth in the market to very specialized tertiary centers.
Growth Opportunities in the Global Rare Disease Gene Therapy Market
Allogeneic "Off-the-Shelf" Gene-Edited Therapies
The rare disease gene therapy market is one of the important growth areas that will benefit the organizations to establish a secure universal donor cell platform for immunological and metabolic disorders. Many biotech firms have been developing autologous models, but now want to create CAR-T or hematopoietic stem cell products from healthy donors that are free of rejection risk. The specialized skills required in the development of these complex environments are Gene Editing Therapy, non-viral vector DNA repair templates and immune evasion engineering. Gene therapy companies can create scalable, cryopreserved Master Cell Banks that significantly cut costs of goods, shorten treatment timelines and increase access beyond academic hubs. The area has a tremendous capacity for a high demand of specific contract manufacturing and analytical development services.
Targeting Non-Liver Systemic Disorders with Novel Delivery Methods
The necessity to combine delivery technology skills with knowledge of particular anatomical barriers will lead to new therapeutic avenues due to the optimization of AAV serotypes for new targets. These include intrathecal (into the central nervous system), subretinal (into the eye) and intramuscular and systemic (into cardiac and skeletal muscle). Medical device manufacturers dealing with neuromuscular and ophthalmic diseases have to follow strict surgical procedures and precision of dosing in micro-dose levels. Therefore, they need channel partners and training solutions that understand the nuances of drug administration in addition to standard pharmacology that are operator dependent. To add value, therapy developers could create companion diagnostic catheters, as is done in neurosurgery, and develop dosing algorithms based on the dynamics of the CSF.
Trends in the Global Rare Disease Gene Therapy Market
The Rise of Next-Generation Viral and Non-Viral Vectors
Novel capsid engineering is becoming increasingly adopted in organizations as an alternative to traditional and wild-type AAV serotypes. Enterprises are constructing synthetic AAV capsids through directed evolution and machine learning that demonstrate superior cardiac or neurological transduction and escape liver sequestration, rather than relying on nature-derived vectors with poor target tissue specificity. These platforms enable the escape of pre-existing immunity, higher transgene expression from lower doses, and reduced toxicity. To counter this, gene therapy companies are providing knowledge in vector biology, manufacturing process development, and scalable synthesis. This trend is driving the development of therapies for previously unreachable dermatological disorders and muscular dystrophies.
In Vivo Gene Editing via Non-Viral Delivery
Another promising approach is enzymatic correction of mutations in the body itself, which are also an area of interest for gene therapy companies as they are becoming increasingly concerned with the need to overcome packaging constraints of viral vectors and immunogenicity. The business is now interested in lipid nanoparticle (LNP) formulations that are being developed to carry CRISPR base editors or prime editing machinery as mRNA cargo for a short period of time, reducing the risk of off-target DNA cutting. This has brought about the need for novel analytics and biodistribution modeling. Gene therapy developers help to design the best LNP formulation, to select the most targeted metabolic pathway in the liver and to lower immunogenicity from the editing enzyme derived from bacteria.
Research Scope and Analysis
The rare disease gene therapy market is witnessing strong growth driven by advancements in in vivo therapies, AAV vectors, hematological disease treatments, intravenous delivery methods, and expanding hospital-based infrastructure. Increasing regulatory approvals, technological innovation, and rising investment in curative therapies continue accelerating global market adoption.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
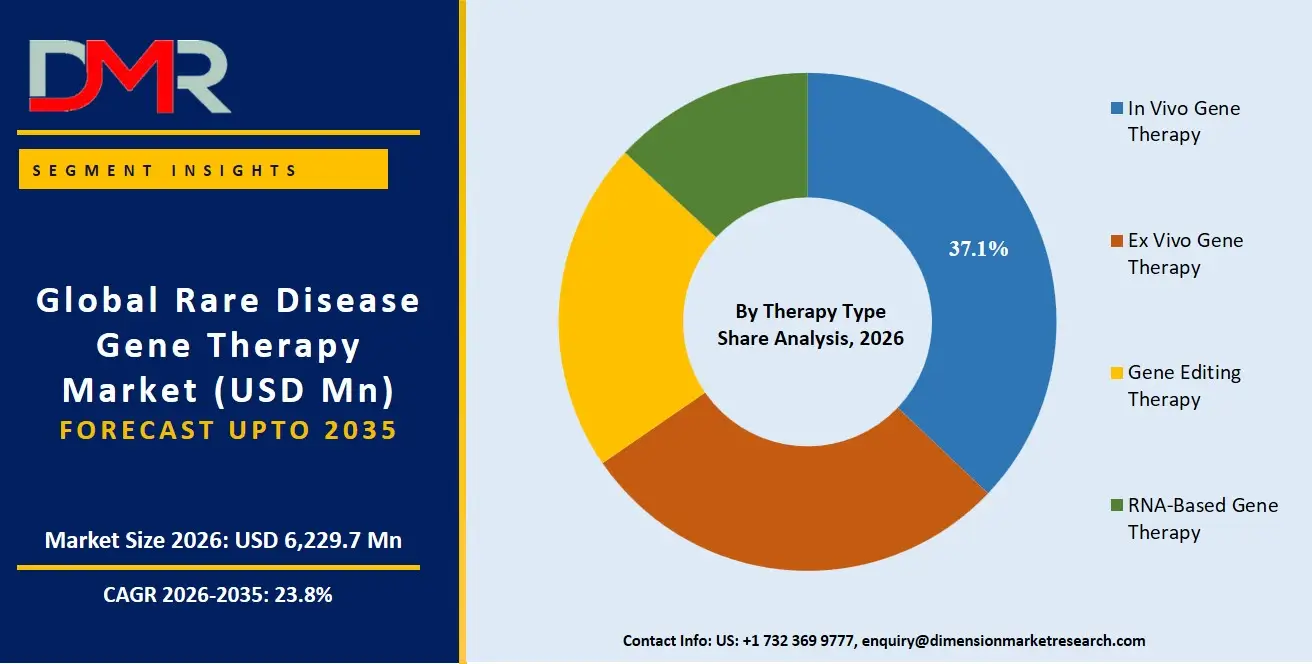
By Therapy Type Analysis
In vivo gene therapy is expected to dominate the therapy type segment because it enables direct delivery of therapeutic genetic material into a patient's body, reducing the need for complex cell extraction and laboratory manipulation. The approach has gained strong clinical and commercial momentum in rare diseases such as spinal muscular atrophy, inherited retinal disorders, and hemophilia. Pharmaceutical companies favor in vivo platforms due to their scalability, faster administration, and growing regulatory acceptance. Advances in viral vector engineering, targeted delivery technologies, and long-term gene expression capabilities further strengthen this segment's leadership. The increasing number of approved one-time curative therapies and expanding clinical pipelines across neurological and metabolic rare diseases continue to accelerate adoption globally, particularly in North America and Europe where advanced reimbursement systems support commercialization.
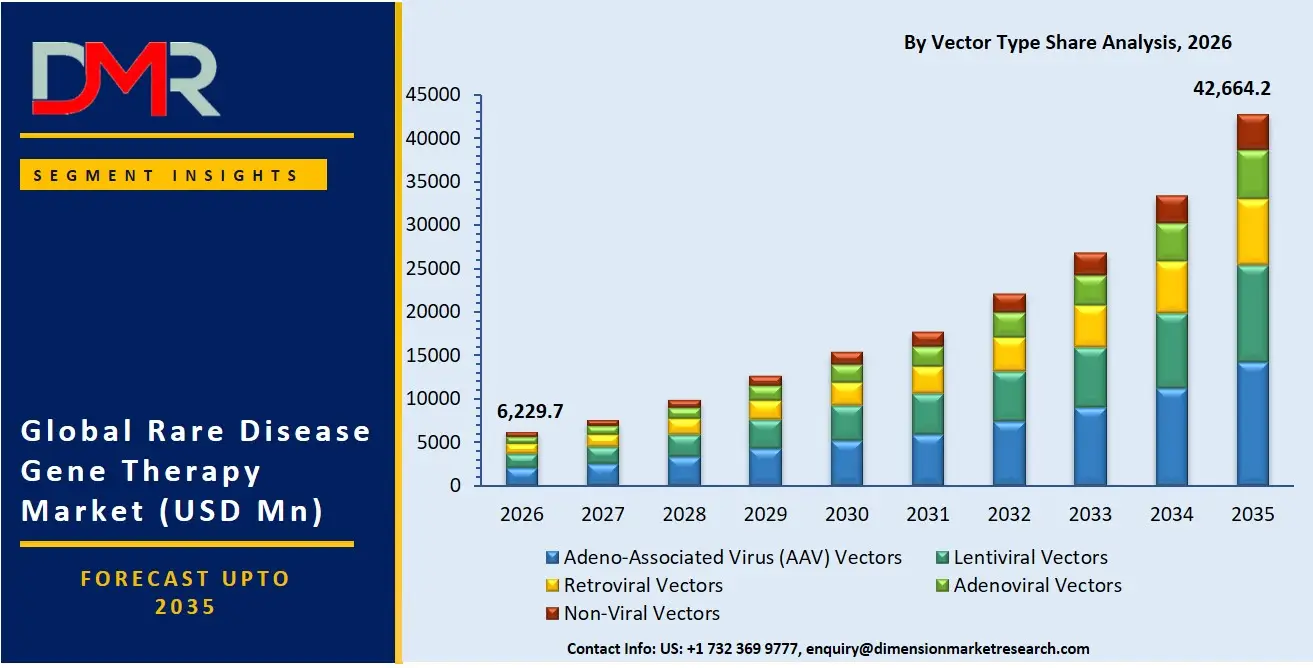
By Vector Type Analysis
Adeno-associated virus (AAV) vectors is poised to dominate the vector type segment owing to their strong safety profile, low immunogenicity, and efficient gene delivery capabilities. AAV vectors are widely used in rare disease therapies targeting neurological, ophthalmic, and muscular disorders because they can achieve long-term gene expression with relatively limited adverse effects. Their versatility across multiple tissue types and compatibility with systemic or localized administration make them highly attractive for developers. Regulatory approvals of several AAV-based therapies have reinforced industry confidence and encouraged significant investment into manufacturing infrastructure and vector optimization technologies. In addition, pharmaceutical and biotechnology firms increasingly prefer AAV platforms due to their proven clinical success, expanding intellectual property ecosystem, and ability to support scalable commercialization for high-value orphan disease therapies worldwide.
By Disease Type Analysis
Hematological disorders is anticipated to dominate the disease type segment due to the significant progress achieved in gene therapies for conditions such as sickle cell disease, beta-thalassemia, and hemophilia. These disorders have well-defined genetic causes, making them highly suitable for targeted gene correction and replacement strategies. Strong clinical outcomes demonstrating reduced transfusion dependence and long-term therapeutic benefits have accelerated regulatory approvals and commercial adoption. Pharmaceutical companies continue prioritizing hematological rare diseases because patient populations are identifiable, treatment efficacy can be clearly measured, and healthcare systems recognize the high unmet medical need. Additionally, increasing investments in CRISPR-based therapies, stem-cell-modified gene therapies, and advanced viral vector technologies further reinforce this segment's dominance, especially across developed healthcare markets with supportive reimbursement frameworks and specialized treatment infrastructure.
By Delivery Method Analysis
Intravenous delivery is expected to dominate the delivery method segment because it enables systemic distribution of gene therapies across multiple organs and tissues, which is essential for treating several rare genetic disorders. This method is particularly effective for metabolic, hematological, and neuromuscular diseases where widespread therapeutic reach is necessary. Intravenous administration also supports minimally invasive treatment compared to surgical delivery approaches, improving patient convenience and clinical workflow efficiency. Pharmaceutical developers prefer this route due to its compatibility with widely used viral vectors, including AAV and lentiviral systems. Growing approvals of intravenously administered one-time gene therapies and ongoing advancements in vector targeting technologies continue strengthening adoption. Moreover, healthcare providers increasingly rely on intravenous infusion infrastructure already established in hospitals and specialty centers, supporting large-scale treatment accessibility.
By End User Analysis
Hospitals is projected to dominate the end user segment because rare disease gene therapies require highly specialized clinical infrastructure, multidisciplinary expertise, and advanced patient monitoring capabilities. Most gene therapies involve complex administration protocols, genetic testing, immune response management, and long-term follow-up care, all of which are best supported within hospital settings. Large academic medical centers and tertiary hospitals also participate extensively in clinical trials, making them early adopters of newly approved therapies. In addition, hospitals maintain access to specialized laboratories, intensive care facilities, and trained genetic medicine professionals necessary for handling high-risk treatments. Reimbursement systems in major healthcare markets are also structured around hospital-based administration for expensive orphan therapies. The increasing establishment of dedicated gene therapy centers further strengthens hospital leadership in this segment globally.
The Global Rare Disease Gene Therapy Market Report is segmented on the basis of the following:
By Therapy Type
- In Vivo Gene Therapy
- Ex Vivo Gene Therapy
- Gene Editing Therapy
- RNA-Based Gene Therapy
By Vector Type
- Adeno-Associated Virus (AAV) Vectors
- Lentiviral Vectors
- Retroviral Vectors
- Adenoviral Vectors
- Non-Viral Vectors
By Disease Type
- Hematological Disorders
- Neuromuscular Disorders
- Metabolic Disorders
- Ophthalmic Disorders
- Immunological Disorders
- Dermatological Disorders
- Other Disease Type
By Delivery Method
- Intravenous
- Intrathecal
- Intramuscular
- Subretinal
- Other Delivery Method
By End User
- Hospitals
- Specialty Treatment Centers
- Research Institutes
- Ambulatory Surgical Centers
Regional Analysis
Leading Region by Market Share
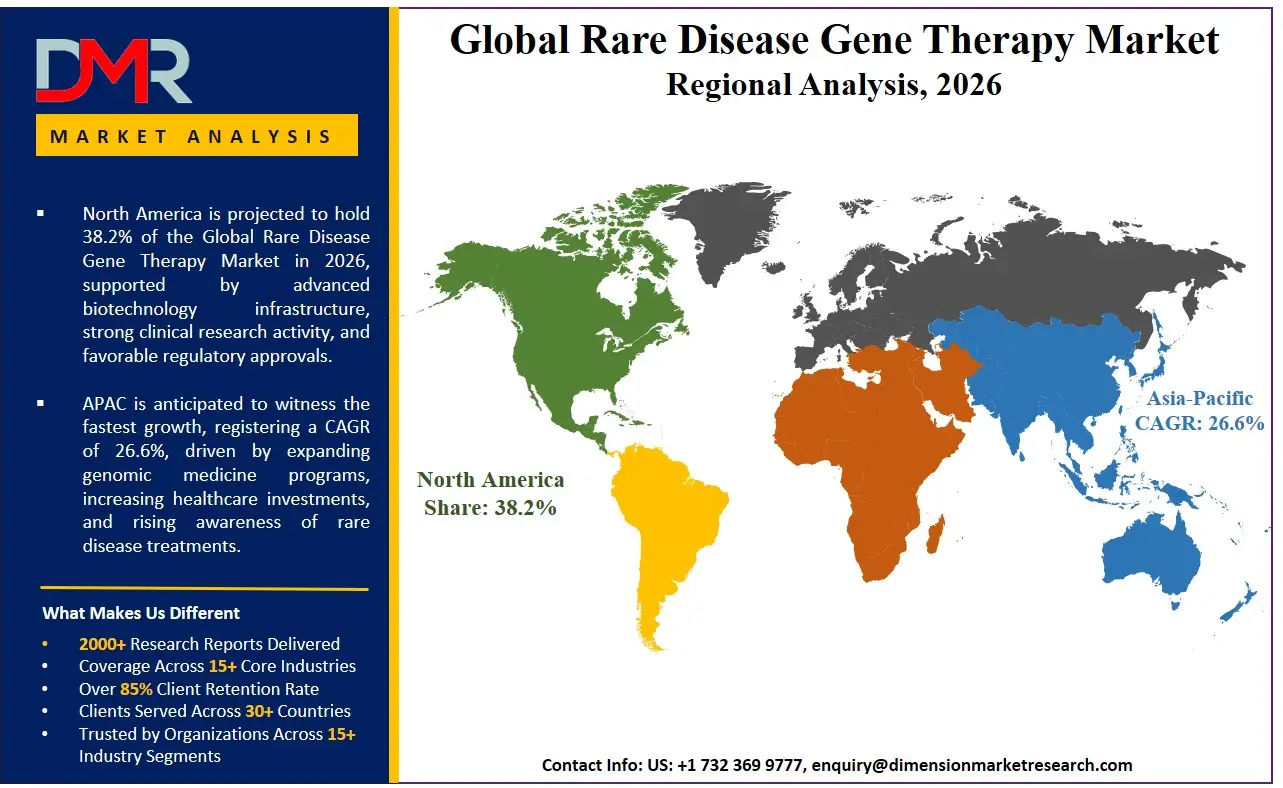
North America is poised to dominate the global rare disease gene therapy market as it is projected to hold 38.2% of the market share by the end of 2026. The United States, which dominates North America, has the highest share in the rare disease gene therapy market because of the unmatched concentration of venture capital-funded biotechnology hubs and the aggressive regulatory acceleration agendas of the FDA's Center for Biologics Evaluation and Research. The area has an established ecosystem of global CDMOs, boutique regulatory consultancies, and a rich pool of translational talent in genetic medicine. Institutional investment in CRISPR, base editing, and the overall expansion of newborn genomic screening initiatives contribute to the continued demand for adeno-associated virus vector manufacturing along with long-term clinical follow-up monitoring. Moreover, an optimistic capital market climate persistently finances the clinical trials of startup gene editing companies that need expert regulatory service providers to achieve expeditious IND approvals and Phase I/II data readouts.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Fastest-Growing Regional Market
Asia-Pacific is expected to be the most rapidly expanding rare disease gene therapy market, driven by government-led sweeping precision medicine initiatives in China, Japan, South Korea, and India. The fast-paced economic growth, the rise of a middle-income population, and the dynamic expansion of domestic biomanufacturing are compelling established pharmaceutical conglomerates and state research hospitals to invest heavily in genetic diagnostics and vector manufacturing infrastructure. Gene Editing Therapy is in high demand to help these large organizations pivot toward CRISPR-based therapeutics for diseases prevalent in the local population, such as high-incidence thalassemias. There is also a severe lack of qualified gene therapy manufacturing talent in the region, and it is necessary to outsource technology transfer and process development to global CDMOs to implement, scale, and secure lentiviral vector production to cover the skills gap and enable faster clinical entry for in-country therapeutic candidates.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The competitive environment of global rare disease gene therapy has become highly dynamic with a heterogeneous array of multinational pharmaceutical giants, pioneering biotechnology pure-plays, and niche viral vector contract manufacturers. The key to success will be the profound strategic platform ownership of proprietary capsid libraries or gene editing nucleases, because they will open the necessary freedom-to-operate positions and early access to new tissue-specific delivery capabilities. The movement towards market consolidation is rapidly progressing with large pharma acquiring clinical-stage gene therapy assets and manufacturing technology specialized boutiques to build out internal gene medicine franchises. Proprietary intellectual property, including optimized non-viral lipid nanoparticle formulations and muscle-specific AAV serotypes, is becoming a more important basis of competitive differentiation than just disease-area selection or clinical trial execution.
Some of the prominent players in the Global Rare Disease Gene Therapy Market are:
- Novartis
- Sarepta Therapeutics
- bluebird bio
- BioMarin Pharmaceutical
- Vertex Pharmaceuticals
- CRISPR Therapeutics
- Spark Therapeutics
- uniQure
- Orchard Therapeutics
- Krystal Biotech
- Pfizer
- Roche
- Biogen
- Amgen
- CSL Behring
- Editas Medicine
- Intellia Therapeutics
- Beam Therapeutics
- Adverum Biotechnologies
- Alnylam Pharmaceuticals
- Other Key Players
Recent Developments
- January 2026: The U.S. Food and Drug Administration declared a major expansion of its Accelerated Approval pathway guidance specific to gene therapies, a regulatory initiative intended to assist sponsors such as Sarepta Therapeutics, uniQure, and Rocket Pharmaceuticals in hematological and neuromuscular disorders to utilize surrogate biomarkers for early approval while committing to long-term extension studies as a condition of market authorization.
- November 2025: Beam Therapeutics, a Boston-based biotech company, strengthened its collaboration with Catalent and inaugurated a dedicated In Vivo CRISPR Delivery and Lipid Nanoparticle Integration practice aimed at supporting metabolic disorder pipelines in moving liver-targeted base-editing therapies into the clinic while maintaining compliance with emerging analytical method requirements.
- October 2025: Putnam Associates, a European regulatory consulting group, acquired Costello Medical to expand its Health Technology Assessment and long-term patient registry capabilities for ophthalmic disorder gene therapies, helping specialist hospital centers and pricing authorities validate real-world visual acuity outcomes.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 6,229.7 Mn |
| Forecast Value (2035) |
USD 42,664.2 Mn |
| CAGR (2026–2035) |
23.0% |
| The US Market Size (2026) |
USD 2,001.4 Mn |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Segments Covered |
By Therapy Type, By Vector Type, By Disease Type, and By Delivery Method |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
Frequently Asked Questions
How big is the Global Rare Disease Gene Therapy Market?
▾ The Global Rare Disease Gene Therapy market is poised to be valued at USD 6,229.7 million in 2026 and is projected to reach USD 42,664.2 million by 2035, driven by the universal need for durable, potentially curative interventions for previously untreatable genetic conditions.
What is the CAGR of the Global Rare Disease Gene Therapy Market from 2026 to 2035?
▾ The market is expected to grow at a CAGR of 23.8% from 2026 to 2035, reflecting the accelerating clinical validation of gene-editing platforms and the persistent breakthrough in engineering next-generation viral and non-viral vectors.
What factors are driving the growth of the Global Rare Disease Gene Therapy Market?
▾ Key drivers include the validation of single-dose curative paradigms, the expansion of mandatory newborn screening programs, the development of immune-evasive AAV capsids, and the surge in demand for streamlined regulatory frameworks amid evolving real-world evidence and durability data requirements.
Which region held the largest share of the Rare Disease Gene Therapy Market in 2026?
▾ North America, specifically the United States, is projected to hold 38.2% of the market share in 2026, driven by a mature biotechnology funding ecosystem and aggressive enterprise investment in In Vivo Gene Therapy and CRISPR-based gene editing capabilities for neurological and hematological disorders.
Which region is expected to grow the fastest in the Rare Disease Gene Therapy Market?
▾ The Asia-Pacific region is expected to grow the fastest, fueled by rapid precision medicine industrialization in China and Japan, where Gene Editing Therapy is critical for transitioning state-sponsored research hospitals to globally competitive genetic medicine trial sites.
What are the major trends in the Global Rare Disease Gene Therapy Market?
▾ Major trends include the integration of AI into novel capsid engineering, the rise of in vivo gene editing via non-viral lipid nanoparticle delivery, the demand for allogeneic off-the-shelf programmed cell therapies for immunological disorders, and the focus on scalable bioreactor-based vector manufacturing within a historically manual GMP environment.
Who are the key players in the Global Rare Disease Gene Therapy Market?
▾ Key players include pharmaceutical incumbents like Novartis and Vertex Pharmaceuticals, pioneering biotechs like Sarepta Therapeutics, CRISPR Therapeutics, and Bluebird bio, as well as the manufacturing divisions of global CDMOs and specialized pure-play vector engineering firms like Regenxbio.
How is the Global Rare Disease Gene Therapy Market segmented?
▾ The market is segmented by Therapy Type, Vector Type, Disease Type, Delivery Method and End Users