Market Overview
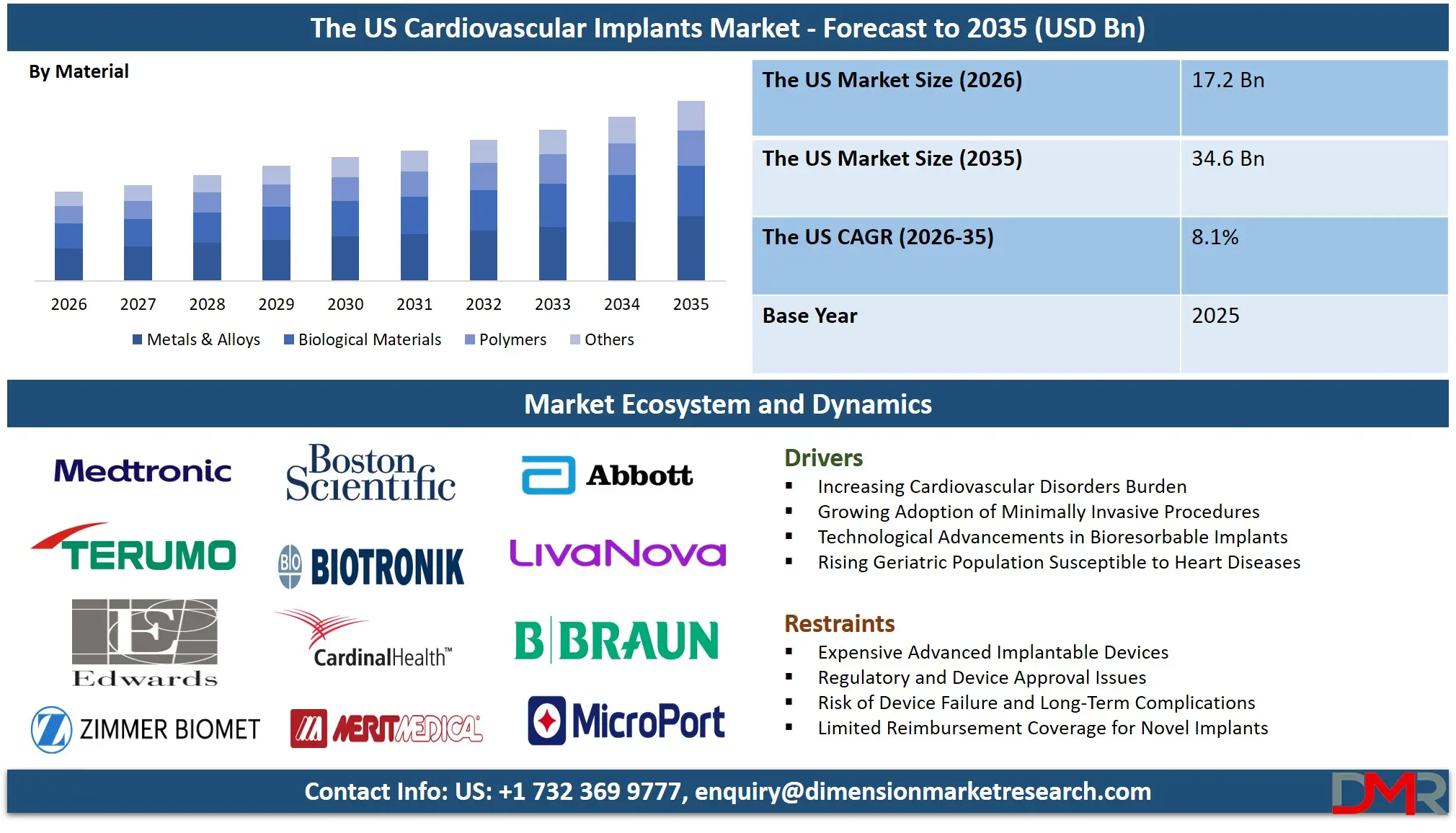
The US cardiovascular implants market size is expected to reach USD 17.2 billion in 2026 and is anticipated to expand at a CAGR of 8.1%, reaching approximately USD 34.6 billion by 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Cardiac implants can be termed as a fragment of cardiovascular apparatus that is inserted in the heart or blood vessels to cure a variety of heart and circulatory problems. Some of them include coronary stents, cardioverter ICD implants, heart valves, pacemakers, CRT stents and peripheral stents.
The US cardiovascular implants market is highly competitive and there has been the trend of high-tech and biocompatible and minimal invasive items that have been used in hospitals and also clinics and cascade surgical centers due to high prevalence of heart condition, and rising demand of interventional cardiology operation, hybrid catheterization laboratory, providing data analytics systems to make precise clinical decisions.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US Cardiovascular Implants Market: Key Takeaways and Other Influencing Factors
- Outlook of Market Growth Expansion: The US Cardiovascular Implants Market is projected to reach USD 17.2 billion in 2026 and nearly USD 34.6 billion by 2035, driven by structural heart disease interventions and device innovation.
- High Growth Rate Momentum: The market has the potential to grow at a rate of 8.1% CAGR in the period ranging from 2026 to 2035, owing to the increase in cardiovascular diseases and minimally invasive surgeries using the latest bioresorbable devices.
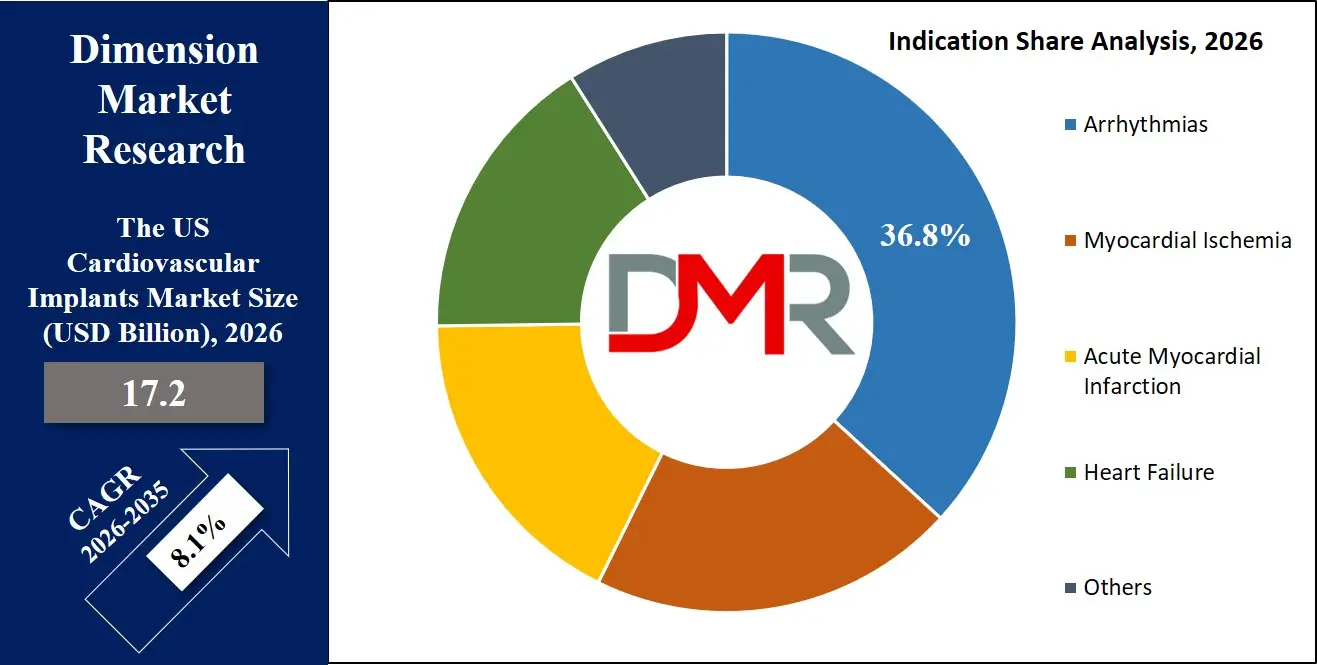
- Segment Leadership Structure: Coronary stents lead the product type segment with 67.0% share in 2026, while arrhythmias dominate the indication segment with 36.8% share and hospitals lead the end-user segment with 54.0% share, reflecting strong reliance on advanced cardiovascular implant solutions.
- Shift Towards Technology-based Care: This results in the gradual shift in the market of bioresorbable stents, leadless pacemakers, transcatheter heart valves, and AI-driven technology of remotely monitoring implanted devices to enhance patient outcomes without necessarily having surgery.
- Cardiovascular Disease Burden: According to Centers for Disease Control and Prevention (CDC), the burden of cardiovascular disease was estimated to be about 702,880 deaths annually in the US, which is a high demand to effective and efficient cardiovascular implants.
- Event Frequency Insight: CDC statistics indicate every 34 seconds in the United States someone has a cardiovascular event which generates high dependency on potentially life sustaining implant devices like ICDs and coronary stents.
- Stroke Risk in AF Patients: The NIH states that stroke amongst atrial fibrillation patients is almost 5 times more prevalent, which proves the importance of implanted cardiac pacemakers and CRT devices in preventing and managing long-term rhythms.
Impact of the Iran conflict on the US Cardiovascular Implants Market
The tension between the US and Iran is disrupting the global supply chain of cardiovascular implants in the US market, oil prices are increasing, which results in disruption of the overall price of cardiovascular devices in the US market, shortage of specialty metals, polymers and biological materials may cause disruption in the overall supply of cardiovascular devices in the US market, and the conflict between the US and Iran could cause disruption in the supply.
The US Cardiovascular Implants Market: Use Cases
- Arrhythmia Management: It can be applied in the detection and treatment of irregular heartbeats such as atrial fibrillation by use of ICDs and implanted pacemaker to enhance accuracy in the control of rhythm at a younger age
- Remote Patient Monitoring: Provides telehealth and less frequent visits to the hospital by assisting in real-time data transmission of system devices at home care.
- Post-Procedure Monitoring: This aids in the process of following up on the recovery and detecting any complications that may arise after angioplasty or open heart surgery via implantable loop recorders and CRT devices.
- Preventive Cardiac Screening: This method enables bypassing high-risk patients by observing and assessing cardiac performance in the long run through implanted hemodynamic monitors.
The US Cardiovascular Implants Market: Market Dynamic
Driving Factors in the US Cardiovascular Implants Market
Increasing Cardiovascular Disorders Burden
The growing level of heart diseases, heart failure, and arrhythmias in the US is contributing to the pressure on the need to implement durable and effective cardiovascular implants. Coronary stents, ICDs and pacemakers can be done to perform early intervention enabling cardiac management in the long term to improve clinical outcomes and preventive cardiology.
Growing Adoption of Minimally Invasive Procedures
Minimally invasive cardiac procedures are more rapidly being adopted because the transcatheter valve interventions and catheter-based angioplasty are becoming more prevalent. Improvement of patient management and reduction of patient dependence on open heart surgery is being done by live imaging, advanced delivery systems and hybrid catheterization laboratories are being developed.
Restraints in the US Cardiovascular Implants Market
Expensive Advanced Implantable Devices
The affordability of the next-generation cardiovascular implants and the associated surgeon procedures is one of the factors that make these devices and services unavailable and utilized by the outpatient and ambulatory surgical centers. Healthcare centers that are sensitive to costs may not be willing to adopt the application of the advanced cardiac implant technology in a massive manner.
Regulatory and Device Approval Issues
FDA approvals, clinical trials, and post-market surveillance have strict compliance requirements that hamper the growth of the market. Some of the issues that determine the use of novel cardiovascular implants are biocompatibility, long-term safety as well as the longevity of the devices.
Opportunities in the US Cardiovascular Implants Market
Artificial Intelligence and Predictive Analytics
The adoption of artificial intelligence in implantable device monitoring will enable arrhythmia to be detected and cardiac insights to be automatically predicted. The remote control of the device will be more accurate due to the AI-based diagnostics, and the number of clinicians that will be hired will be lowered, and the long-term worth of cardiovascular implants will be enhanced.
Expansion of Home Healthcare Services
The increased preference of home-based patient care is posing a need in the remote-monitored implantable devices. ICDs and pacemakers powered by wireless allow managing patients in non-clinic conditions, which makes it possible to manage chronic heart disease and offer personal health care.
Trends in the US Cardiovascular Implants Market
Shifting towards Leadless and Bioresorbable Devices
The change towards leadless pacemaker and bioresorbable coronary stents with fewer long-term complications and higher patient adherence is clear as the conventional permanent implants are pushed out. The trend assists in maintaining cardiac functions in minimal lifetime burden of the device.
Progress in Wireless and Remote Monitoring Systems
The implant data management is changing with the growing application of wireless connectivity, mobile applications, and cloud platforms. The future of cardiac management devices in the US is typified by smooth exchange of information, remote programming of the devices and alignment with electronic health records.
The US Cardiovascular Implants Market: Research Scope and Analysis
By Product Type Analysis
By product type, coronary stents are expected to capture the leading segment share of more than 67.0% in 2026 due to high prevalence of coronary artery disease and increasing adoption & application of drug eluting stent in angioplasty & acute myocardial infarction management in coming years. Increased use of these implants is due, in part, to their ability to restore blood flow and reduce restenosis. In contrast, though cost and longer approval times have stunted development compared to coronary stents, ICDs and implanted cardiac pacemakers are still very much candidates for chronic arrhythmia management therapy as well as heart failure therapy based on clinical effectiveness data quality combined with recommendations from guidelines.
By Material Analysis
The material segment is dominated by metals & alloys in 2026, mainly due to the extensive use of stainless steel, cobalt chromium, and titanium alloys in coronary stents, pacemakers, and ICDs due to their high strength, biocompatibility, and radiopacity. On the other hand, the use of polymers is rising in drug-eluting stents, bioresorbable stents, and insulation due to their flexibility and low inflammatory response. Similarly, the market share of various biological materials, including porcine and bovine heart valve replacement, is high due to their good hemodynamics and low anticoagulation therapy requirements. Finally, the 'others' category includes ceramic coatings and composite materials used in various implantable device components.
By Procedure Type Analysis
Alongside a substantial number of percutaneous coronary interventions carried out for coronary artery disease and acute myocardial infarction with laparoscopic or transcatheter delivery of drug-eluting balloons, angioplasty is projected to lead this portion of the market in 2026. This predominant status is due to the minimally invasive technique, shorter recovery durations and lower complication rates than open heart surgery. While open heart surgical procedures have lessened in many areas including coronary artery bypass grafting, they clearly remain essential for complex valve replacements, multi-vessel disease and congenital defects that cannot be managed with angioplasty.
By Indication Analysis
The arrhythmias segment accounts for the highest percentage of overall share by indication and is expected to dominate owing to rising need for early detection and treatment of the atrial fibrillation and ventricular tachycardia along with increasing adoption of ICDs and pacemakers in clinical practice, contributing around 36.8% share in 2026.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
This expansion is being driven with a focus on preventive electrophysiology as well. The acute myocardial infarction segment is anticipated to be driven by the increasing rate of emergency angioplasty & coronary stenting, where cardiovascular implants quickly restores coronary flow and enables real-time management of heart attacks in catheterization laboratories.
By End-User Analysis
In 2026, hospitals are anticipated to hold a significant 54.0% market share in the end-user segment due to large volume of patient inflows, advanced cardiac catheterization laboratories and strong inclination towards surgical-grade cardiovascular implants to ensure precision treatment for complex cardiac diseases. Coronary stents, ICDs, and heart valves are widely adopted in emergency and elective procedures in these settings. Specialty clinics have continued to grow with an emphasis on arrhythmias and heart failure. Outpatient-based angioplasty and pacemaker implantations are rapidly expanding at ambulatory surgical centers. Cardiac catheterization laboratories are important procedural nodes for diagnostic and interventional cases, while other sites include academic research centers and long-term care facilities.
The US Cardiovascular Implants Market Report is segmented on the basis of the following:
By Product Type
- Coronary Stents
- Implantable Cardioverter Defibrillators (ICDs)
- Heart Valves
- Implanted Cardiac Pacemakers
- Cardiac Resynchronization Therapy (CRT) Devices
- Peripheral Stents
- Others
By Material
- Metals & Alloys
- Polymers
- Biological Materials
- Others
By Procedure Type
- Angioplasty
- Open Heart Surgery
By Indication
- Arrhythmias
- Myocardial Ischemia
- Acute Myocardial Infarction
- Heart Failure
- Others
By End-User
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratories
- Others
Impact of Artificial Intelligence in the US Cardiovascular Implants Market
In the US cardiovascular implants market, artificial intelligence is providing significant improvements by allowing the automated detection of arrhythmias using implant devices. It is also allowing the timely processing of device telemetry information with increased precision. With the help of artificial intelligence-powered implant devices, real-time remote patient monitoring is being facilitated along with the early identification of cardiac conditions such as atrial fibrillation and ventricular tachycardia.
Additionally, machine learning is allowing the timely processing of large amounts of heart information obtained using implant devices. It is providing significant improvements in the US cardiovascular implants market.
The US Cardiovascular Implants Market: Competitive Landscape
The US cardiovascular implants market is very competitive with a high emphasis laid on innovation with respect to next-generation cardiovascular implant devices.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
There is an increased trend towards the development of advanced cardiovascular implant devices with high biocompatibility, battery life, and miniaturized devices. An increased trend is being observed with respect to minimally invasive procedures with the integration of artificial intelligence-powered remote patient monitoring.
Some of the prominent players in the US Cardiovascular Implants Market are:
- Medtronic plc
- Boston Scientific Corporation
- Abbott Laboratories
- Edwards Lifesciences Corporation
- Johnson & Johnson
- Terumo Corporation
- Biotronik SE & Co. KG
- LivaNova PLC
- B. Braun SE
- W. L. Gore & Associates, Inc.
- Teleflex Incorporated
- Getinge AB
- Cardinal Health, Inc.
- Merit Medical Systems, Inc.
- Cook Medical Inc.
- Artivion, Inc.
- MicroPort Scientific Corporation
- Abiomed, Inc.
- Penumbra, Inc.
- Siemens Healthineers
- Other Key Players
Recent Developments in the US Cardiovascular Implants Market
- October 2025: Abbott Laboratories introduced its AVEIR dual chamber leadless pacemaker system (DR), allowing wireless communication between two devices. This has further expanded the capabilities of leadless pacing.
- May 2024: Boston Scientific Corporation announced positive results for the MODULAR ATP clinical trial on the EMPOWER leadless pacemaker and subcutaneous ICD system. These results showed high levels of safety and efficacy.
- March 2024: Medtronic plc secured FDA clearance for its Evolut FX+ TAVR system, providing further treatment options for severe aortic stenosis.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 17.2 Bn |
| Forecast Value (2035) |
USD 34.6 Bn |
| CAGR (2026–2035) |
8.1% |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Report Coverage |
Market Revenue Estimation, Market Dynamics, Competitive Landscape, Growth Factors and etc. |
| Segments Covered |
By Product Type (Coronary Stents, Implantable Cardioverter Defibrillators (ICDs), Heart Valves, Implanted Cardiac Pacemakers, Cardiac Resynchronization Therapy (CRT) Devices, Peripheral Stents, and Others), By Material (Metals & Alloys, Polymers, Biological Materials, and Others), By Procedure Type (Angioplasty and Open Heart Surgery), By Indication (Arrhythmias, Myocardial Ischemia, Acute Myocardial Infarction, Heart Failure, and Others), By End-User (Hospitals, Specialty Clinics, Ambulatory Surgical Centers, Cardiac Catheterization Laboratories, and Others) |
| Country Coverage |
The US |
| Prominent Players |
Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, Edwards Lifesciences Corporation, Johnson & Johnson, Terumo Corporation, Biotronik SE & Co. KG, LivaNova PLC, B. Braun SE, W. L. Gore & Associates, Inc., Teleflex Incorporated, Getinge AB, Cardinal Health, Inc., Merit Medical Systems, Inc., Cook Medical Inc., Artivion, Inc., MicroPort Scientific Corporation, Abiomed, Inc., Penumbra, Inc., Siemens Healthineers, and Other Key Players |
| Purchase Options |
We have three licenses to opt for: Single User License (Limited to 1 user), Multi-User License (Up to 5 Users) and Corporate Use License (Unlimited User) along with free report customization equivalent to 0 analyst working days, 3 analysts working days and 5 analysts working days respectively. |
Frequently Asked Questions
How big is the US Cardiovascular Implants Market?
▾ The US Cardiovascular Implants Market is valued at USD 17.2 billion in 2026 and is projected to reach USD 34.6 billion by the end of 2035.
What is the growth rate for the US Cardiovascular Implants Market?
▾ The market is growing at a robust compound annual growth rate (CAGR) of 8.1% over the forecast period of 2026 to 2035.
Who are the key players in the US Cardiovascular Implants Market?
▾ Some of the major key players in the US Cardiovascular Implants Market are Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, Edwards Lifesciences Corporation, Terumo Corporation, Biotronik SE & Co. KG, and many others.