What is the Exosome Diagnostics and Therapeutics Market Size?
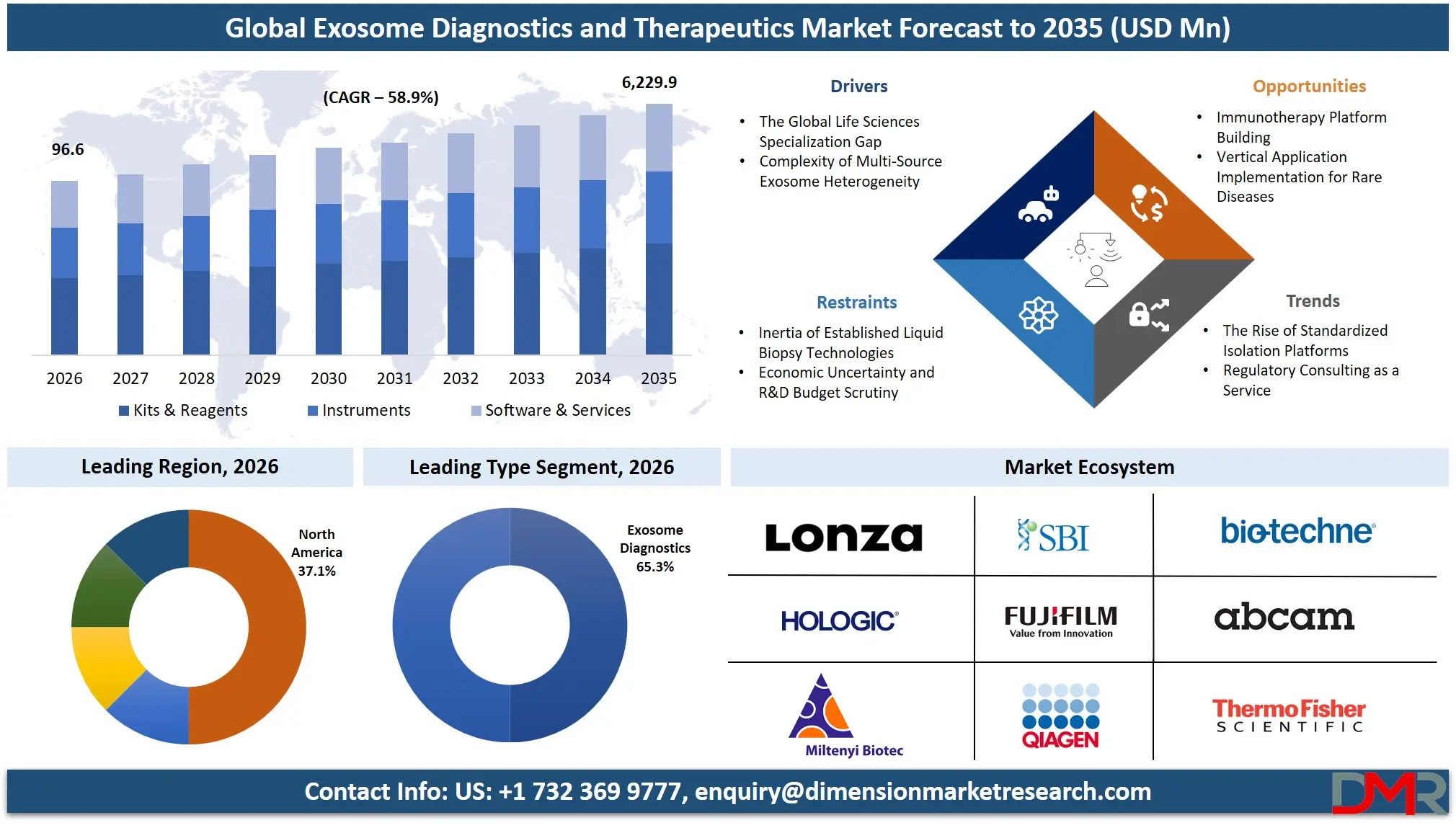
The Global Exosome Diagnostics and Therapeutics Market is expected to reach a value of USD 96.6 million in 2026, and it is further anticipated to reach USD 6,229.9 million by 2035, growing at a CAGR of 58.9% during the forecast period.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The exosome diagnostics and therapeutics market is increasing exponentially with the life sciences industry becoming increasingly focused on precision medicine and abandoning more traditional diagnostic and therapeutic approaches and moving to nanoscale, biologically-derived platforms.
The market comprises of liquid biopsy-based diagnostics, drug delivery systems, isolation kits, instruments, and related software and services which will enable researchers and clinicians to harness exosomes to detect diseases and target therapy in a non-invasive manner. The need to have specialized products and services is being driven by the increasing demand to implement exosome-based Liquid biopsy-based diagnostics, biomarker discovery & disease monitoring, and novel drug delivery systems. The most common adopters are hospitals, diagnostic laboratories and pharmaceutical companies because of their high level of necessity in the research process. The areas of application of the cancer (oncology), neurological disorders, and cardiovascular diseases are some of the key drivers since they demand highly sensitive, specific, and biocompatible diagnostic and therapeutic solutions.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
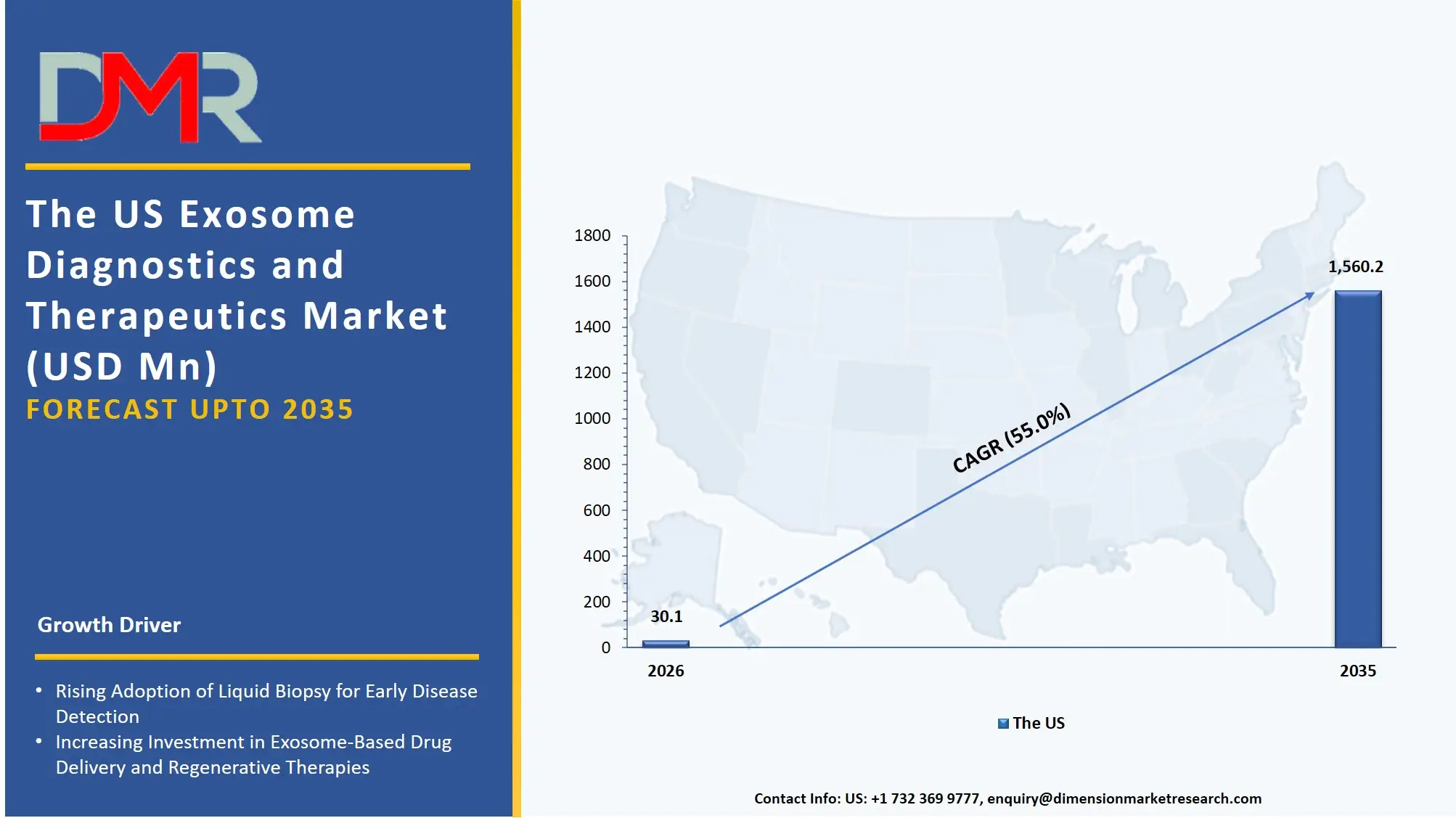
The US Exosome Diagnostics and Therapeutics Market
The US Exosome Diagnostics and Therapeutics Market is projected to reach USD 30.1 million in 2026 at a compound annual growth rate of 55.0% over its forecast period, which is further poised to be valued USD 1,560.2 million by 2035.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
The US remains the largest and most advanced market in exosome diagnostic and therapeutic due to the strong research infrastructure of the best academic institutes and the aggressive research and development programs of biotechnology companies. High demand of the liquid biopsy-based diagnostics services whereby organizations are targeted to identify circulating exosomal biomarkers of early cancer (oncology) screening has typified the market. In addition, the creation of engineered exosomes as Drug delivery systems is generating a similar requirement in bioinformatics tools and data analysis platforms to control cargo loading efficiency and therapeutic efficacy analytics.
The Europe Exosome Diagnostics and Therapeutics Market
The Europe market is estimated to be valued at USD 27.9 million in 2026 and is further anticipated to reach USD 1,753.7 million by 2035 at a CAGR of 58.1%.
The regulatory environment, such as the new In Vitro Diagnostic Regulation (IVDR) and the emerging cell-based therapy guidelines, have a pronounced influence on the European market and influence the necessity to use the vigorous detection and analysis reagents and standardized isolation kits. The region is also experiencing accelerated growth of exosome-based Regenerative medicine with both academic and commercial entities in Germany and Switzerland striving to balance the complexity of manufacturing with the scalability of the therapeutic process. In addition, initiatives such as the European Union's Horizon Europe are challenging service providers to create dedicated biomarker discovery & disease monitoring software and services to provide data interoperability and reproducibility across European research ecosystems.
The Japan Exosome Diagnostics and Therapeutics Market
The Japan market is projected to be valued at USD 10.2 million in 2026. It is further expected to witness robust growth, holding USD 579.7 million in 2035 at a CAGR of 56.4%.
The Japanese market is a distinct one, where nationally there is a drive to be first in introducing regenerative solutions due to the ageing population and the high levels of age related diseases. A significant portion of the expenditure is comprised of research in regenerative medicine and discovery and disease monitoring of biomarkers as universities and biotech companies carry exosome-based therapies into the bedside. There is also a strong need to integrate deeply with the local market to bridge the gaps between traditional cell culture techniques and advanced nanoparticle tracking analyzers, which forms a niche in ultracentrifuge and flow cytometry systems-based workflows.
Key Takeaways
- Market Size & Forecast: The Global Exosome Diagnostics and Therapeutics market is projected to reach USD 96.6 million in 2026, expanding dramatically to USD 6,229.9 million by 2035, due to the dual drivers of increased therapeutic pipeline development and the obligatory modernization of liquid biopsy cancer (oncology) screening.
- Growth Rate & Outlook: Global market growth is expected at a CAGR of 58.9%, due to the critical lack of standardized isolation protocols, and the growing complexity of defining heterogeneous populations of exosomes in different sources.
- Primary Growth Drivers: Key forces include the widespread shift from invasive tissue biopsies to liquid biopsy-based diagnostics, the need for specialized isolation kits to avoid co-isolation of non-exosomal particles, and the engineering of immunotherapy platforms requiring specialized detection & analysis reagents.
- Key Market Trends: Major trends are the rise of therapeutic-specific engineering, the application of AI-powered tools in bioinformatics tools to predict exosome cargo function, and the transition to rigorous characterization as regulatory bodies prioritize consistency of drug delivery systems.
- By Type Analysis: Exosome diagnostics is projected to the dominant one because of its high clinical acceptance particularly in fluid biopsy to detect and monitor early. The increased revenue is due to commercial availability, regulatory progress, and the demand of non-invasive precision medicine solutions rather than therapeutics.
- By Product & Service Analysis: Kits and reagents is expected to dominate this segment as the key consumables that are reused in research and diagnostics. Isolation and detection product demand, ease of operation and research on the expanding exosome studies all assure consistent revenue, as opposed to the expensive instruments.
- By Source Analysis: Blood and plasma is anticipated to dominate this segment because they are easily collected with minimal invasiveness and have high biomarker concentrations. Their applicability to both liquid biopsy and standardized clinical guidelines makes them the most reliable and the most widely used source of exosome analysis.
- Regional Leadership: North America is poised to dominate this market with 37.1% of the market share in 2026 due to its well-developed life sciences ecosystem that utilizes this infrastructure to its fullest and makes it a leader in this market.
What is the Exosome Diagnostics and Therapeutics?
Exosome diagnostics and therapeutics are targeted nanoscale products and platforms that life science companies, diagnostic developers and research institutes use to help organizations navigate the entire exosome translational lifecycle. These platforms are associated with the biological complexity of exosome engineering and detection, unlike the traditional molecular assays or synthetic drug vectors. This includes diagnostics to develop a non-invasive disease signature to be used in early diagnosis, therapeutics to develop exosomes as targeted drug delivery systems in physical terms without toxicity to the organism, and software and services to ensure that heterogeneous exosome data can effectively communicate with existing clinical informatics tools. With numerous organizations running multi-omics discovery workflows, specialized services are needed to achieve analytical rigor, therapeutic potency, and regulatory compliance, making exosome investments translate into tangible clinical utility, as opposed to academic complexity.
Use Cases
- Early Cancer Screening in Oncology: Research and academic institutes seek diagnostic developers to use liquid biopsy-based diagnostics to identify exosomal signatures in blood and blood plasma, and re-write the paradigm of late-stage cancer detection into routine screening with non-invasive venipuncture.
- Targeted Drug Delivery in Neurological Disorders: Pharmaceutical and biotechnology companies apply exosome drug delivery systems and bioinformatics tools to designing stem cell-derived exosomes that cross the blood-brain barrier, migrating therapeutic cargoes to specific CNS sites and preparing the therapeutic approach to clinical trials.
- Regenerative Medicine in Cardiovascular Diseases: Hospital networks deploy therapeutic exosomes that reduce the formation of myocardial scars, using workflows based on exosome-based regenerative medicine, with GMP-compliant isolation kits and analyzed with nanoparticle tracking analyzers.
- Biomarker Monitoring in Infectious & Rare Diseases: Diagnostic laboratories use biomarker discovery and disease monitoring services to integrate exosome-based panels on urine samples with clinical data systems to provide real-time monitoring of disease progression and treatment response by non-invasive sampling.
How AI is Transforming the Exosome Diagnostics and Therapeutics Market?
The exosome diagnostics and therapeutics field is changing as AI can speed up the process of biomarker discovery, as well as increase the precision of the therapeutic design. In software and services, AI-based bioinformatics tools can potentially automatically deconvolve exosome cargo data, transforming intricate multi-omic datasets into clinically actionable biomarker panels, and vastly reduce manual data analysis time and project risk. In the meantime, AI-driven functions within drug delivery systems enable researchers to have a better control over loading payloads by predicting interactions between membranes, suggesting the best engineering solutions such as surface functionalization modifications to strengthen pharmacokinetic approaches.
The development of therapeutics and diagnostics is also centered around AI. In the area of detection & analysis reagents, intelligent image analysis agents are used to continuously monitor nanoparticle tracking analysis feeds and identify distinct exosome subpopulations, morphological patterns, and contaminants to keep research teams in line with experimental rigor. Additionally, bioinformatics tools are being complemented by generative AI assistants simulating exosome biodistribution and modeling future clinical trial results to provide stakeholders with a visualization of therapeutic success prior to investment of vast amounts of R&D resources.
Market Dynamics
Key Drivers in the Global Exosome Diagnostics and Therapeutics Market
The Global Life Sciences Specialization Gap
International institutions are struggling to get skilled professionals with the knowledge of nanoscale vesicle biology, advanced ultracentrifugation, exosome engineering, and multi-omics analysis. The skills are demanded at a rate higher than the rate at which the trained talents are available hence a structural shortage of the labor market. This is leading to a trend of pharmaceutical & biotechnology companies outsourcing specialized product and service providers of exosome tools rather than depending on in-house development. The companies help in the processes of isolation, characterization, engineering and data analysis that is continuously provided. Outsourcing such functions allows organizations to speed up translational research initiatives and minimize the chances of delays due to insufficient in-house capacities.
Complexity of Multi-Source Exosome Heterogeneity
Research enterprises of significant size are also inclined to analyze multiple exosome sources, largely blood and blood plasma, urine and stem cells, to ensure that they encompass a broad biological range of biomarkers. Nonetheless, controlling the heterogeneity of exosomes from different sources is very complicated. The companies have to standardize isolation procedures, characterization criteria, cargo examination and therapeutic capacity across a variety of platforms using a variety of tools and standards. This complexity can lead to inefficiencies, batch-to-batch variability and irreproducible data without the assistance of expert tools. And so there is an increasing requirement in the tools and software & services that can help enterprises to function in such a heterogeneous biological habitat.
Restraints in the Global Exosome Diagnostics and Therapeutics Market
Inertia of Established Liquid Biopsy Technologies
The vast majority of diagnostic laboratories continue to operate using systems that were developed around circulating tumor DNA (ctDNA) and circulating tumor cells (CTCs), which have become very standardized in their respective operations. The presence of these established technologies would be a major barrier to new exosome-based diagnostics, even though they hold potential avenues to more richly provide biological information. It can be costly and risky to shift complex clinical workflows, proprietary assay designs, and highly integrated regulatory filings to a new analyte class. Liquid biopsy transitions which involve hundreds of patient samples require much planning, analytical validation, and clinical evidence. The organizations fear a regulatory discontinuity, reimbursement uncertainty, and unexpected expenses in the transition. The adoption inertia slows the uptake of exosome diagnostics and is likely to delay or even prevent the uptake of larger scale exosome diagnostics.
Economic Uncertainty and R&D Budget Scrutiny
Unstable economies and uncertain funding conditions have made the pharmaceutical & biotechnology companies more cautious to invest in emerging technology projects. Although exosome therapeutics remains a strategic priority, research executives are under a pressure to justify each investment and offer a measurable pipeline value. The costly instrument platforms and long-term contract structures of software and services delivering bioinformatics solutions are more prone to fall under a greater amount of scrutiny. Companies have turned to short, discovery-based projects that yield rapid biomarker panels or proof-of-concept information. Larger therapeutic and regenerative medicine efforts over the long term are more likely to wait until the providers can show a near-term differentiation. This transformation is pushing the product and service companies to be more performance-oriented and value-driven.
Growth Opportunities in the Global Exosome Diagnostics and Therapeutics Market
Immunotherapy Platform Building
One of the significant growth opportunities in the exosome diagnostics and therapeutics market is assisting pharmaceutical & biotechnology companies to create secure, biologically-native immunotherapy platforms. Many companies now want to have their own platforms to exploit the inherent biocompatibility and targeting properties of exosomes. These advanced environments are developed with the professional expertise of engineering sources of stem cells, loading cargo with therapeutic content, and scaling of GMP-compliant isolation. The providers of exosome products and services can help enterprises to develop scalable, customized immunotherapy ecosystems that can aid targeted cancer (oncology) therapeutic interventions and immune modulation. The region can generate a strong demand in services of a highly specialized engineering and manufacturing services.
Vertical Application Implementation for Rare Diseases
The necessity to combine both technical expertise and knowledge of specific clinical domains is the driving force of the development of exosome diagnostics as researchers develop solutions to specific classes of diseases. These include exosome panels, centrally based on neurological disorders, biomarker assays of rare diseases, and cardiovascular disease monitoring workflows. Those companies that are interested in developing diagnostics in infectious and rare diseases, have to comply with severe clinical regulations and special operation requirements. Thus, they require partners that comprehend the exosome technology and disease-specific regulatory framework. To add value, product and service providers can consider adding new isolation kits to current diagnostic systems, in line with FDA guidelines, and adjusting analysis software.
Trends in the Global Exosome Diagnostics and Therapeutics Market
The Rise of Standardized Isolation Platforms
Standardisation of platforms is becoming very widely used in organisations as an alternative to the traditional and siloed home-brew centrifugation techniques. Labs Research & academic institutes are building Internal Reference Platforms, providing reproducible exosome preparations, instead of each lab developing its own protocol independently. These platforms make it easy to deploy isolation kits, to use instruments, and to automate workflow parameters. To combat this, vendors of exosome products are offering platform design, integration of ultracentrifuges with detection and analysis reagents, and automated analysis.
Regulatory Consulting as a Service
The changing regulatory landscape is also becoming a major factor when making exosomes therapeutic decisions as companies are under pressure to achieve CMC (Chemistry, Manufacturing, and Controls) consistency and minimize risk in clinical trials. Exosome strategies are currently of interest to biotech companies to assist in improving the therapeutic efficacy and also fulfill the FDA and EMA requirements. This has brought about the need to have specialized bioinformatics and regulatory services. Software and service providers help organizations to understand the analytical assay requirements, potency assay development, batch-to-batch consistency analysis, and quality control by advanced nanoparticle tracking analyzers.
Research Scope and Analysis
The market is segmented by type into exosome diagnostics and therapeutics; by product and service including kits, reagents, instruments, software; by source such as blood, urine, stem cells; by application across diseases; and by end users including hospitals, laboratories, research institutes, and biopharma companies.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
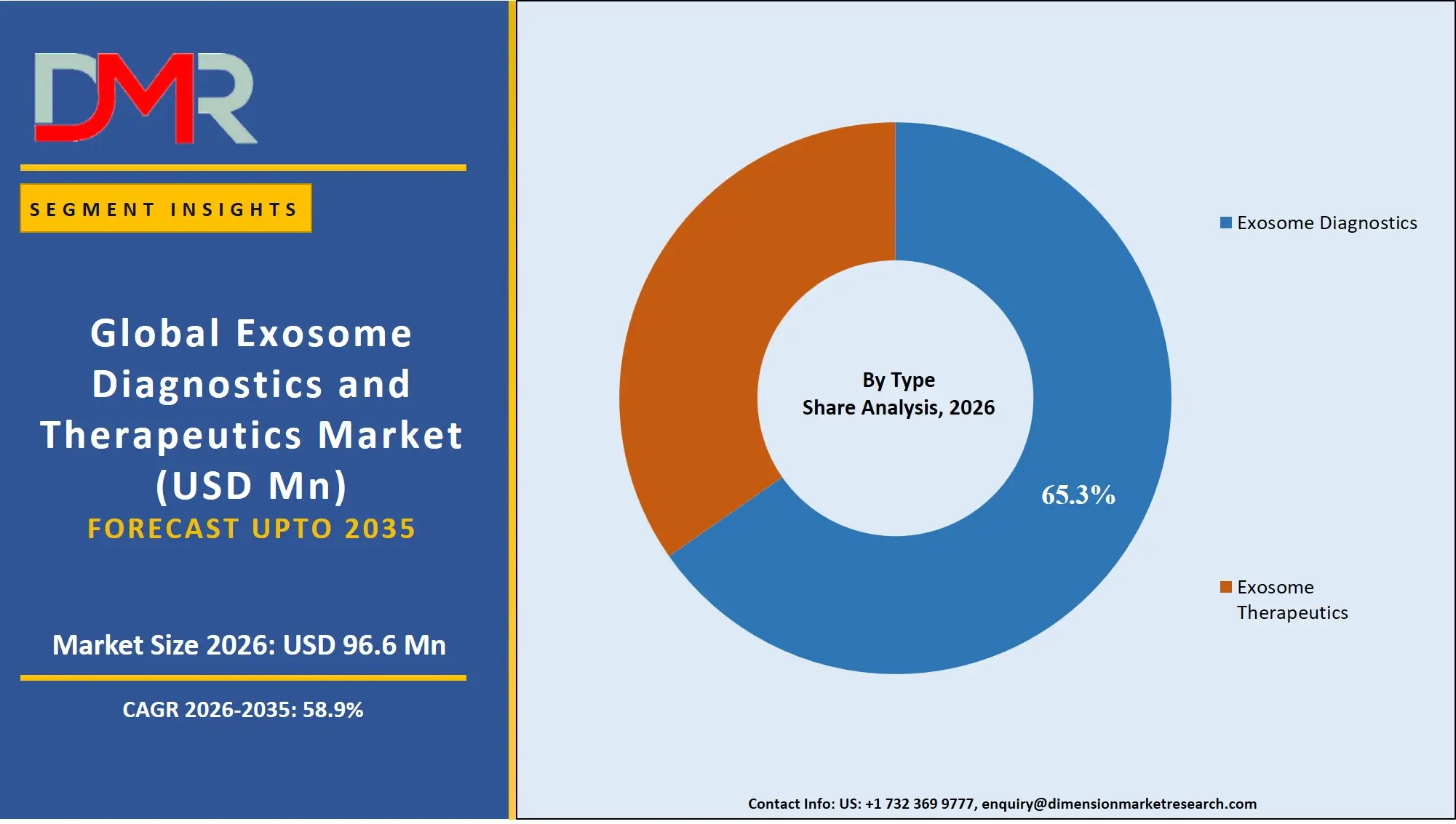
By Type Analysis
Exosome diagnostics is projected to dominate this market with highest market share in 2026 because of their immediate clinical utility and increasing use in non-invasive disease diagnosis. In particular, liquid biopsy-based diagnostics are showing a strong momentum since they are both able to detect early cancer, monitor in real time, and evaluate response to treatment without invasive procedures. In comparison to therapeutics, which are yet to attain commercialization particularly in the oncology area, diagnostic solutions have already reached commercialization, notably in the oncology sector. Also, the growing demand of precision medicine and personalized healthcare is driving the application of exosome-derived biomarkers. Some of the strongest regulatory developments and integration into clinical workflows further enhance the leadership of this segment and make diagnostics the main source of revenue and therapeutics a high-growth segment but still in its early stages.
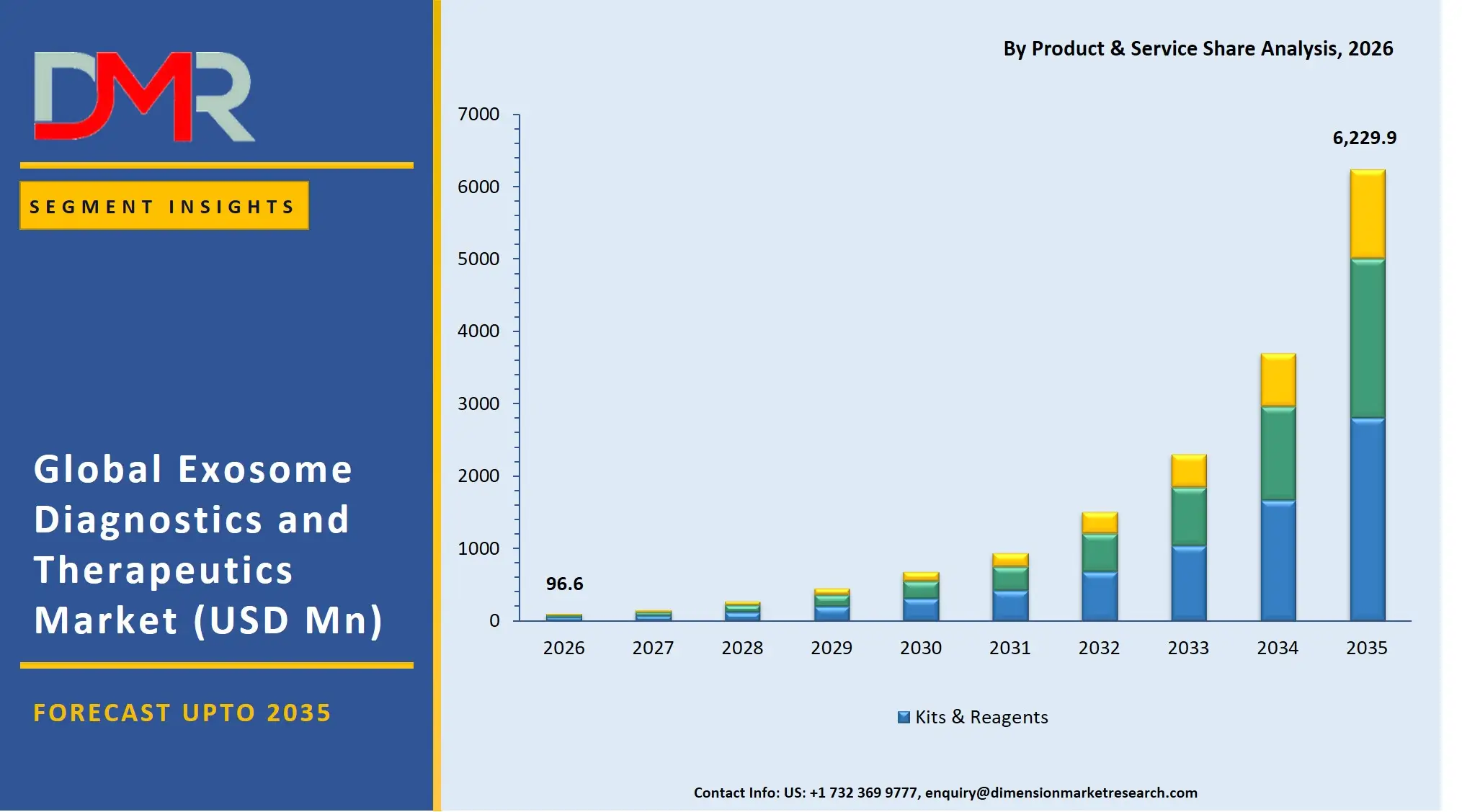
By Product & Service Analysis
Kits and reagents are poised to have the highest share of this segment because they are used repeatedly across both research and diagnostic uses. Isolation kits and detection reagents used in exosome isolation are important regular consumables in laboratories and therefore will always be in demand as opposed to purchasing an instrument once. The fast growth of exosome studies, in conjunction with the growing number of clinical validation studies, have greatly increased the consumption of these products. They are also extensively favored in academic and business environments because of their ease of use, standardization and compatibility with different types of samples. Instrumentation like ultracentrifuges and nanoparticle tracking analysers are essential but expensive necessities and thereby they reinforce the dominance of consumables in revenue generation.
By Source Analysis
Blood and blood plasma are expected to dominate the source segment due to its availability, quality, and extensive use in clinical diagnostics. The samples can be easily collected using minimally invasive procedures, which makes them very suitable to routine testing and large scale screening. The exosomes in blood are rich in molecular information (such as proteins, RNA and DNA) that can be very useful in detecting and monitoring diseases. Their compatibility with liquid biopsy technologies also contributes to their dominance and in particular in oncology applications. The blood presents better uniformity and standardized procedures as compared to other sources like urine or stem cells and thus the blood is the preferred choice in both clinical and research settings hence driving its dominant position in the market.
By Application Analysis
Cancer is expected to remain the dominant application segment because a large number of exosomes have been used to detect, predict, and monitor the progression of cancer. Exosomes have been found to be extremely significant in the area of cancer biology, with tumor-specific biomarkers (capable of being detected by liquid biopsy techniques) being transported by these exosomes. The growing cancer load in the world, and the mounting pressure on non-invasive diagnostic procedures are some of the major factors contributing to this dominance. Also, the fact that the research into exosome-based drug delivery systems to treat specific cancer types is ongoing, further fortifies this segment. Oncology has the advantage of a larger funding base, a shorter time to clinical adoption and high activity in its pipeline compared to other applications, such as cardiovascular or neurological disorders, making it the highest priority area within the exosome diagnostics and therapeutics market.
By End User Analysis
This segment is anticipated to largely dominated by research and academic institutes because they are the key players in the development of the science and technology of exosomes. Much of the exosome-relevant activity, such as the discovery of biomarkers, development of therapeutic agents and validation experiments, happens in these institutions. Extensive research projects are facilitated by high funding through governments and other organizations making exosome related products and services highly demanded. Moreover, the pre-clinical nature of the exosome therapeutics keeps most of the work in the research setting, but not in the clinical setting. Although the adoption of tools based on exosomes is increasingly taking place in hospitals and in diagnostic laboratories, much of the innovation and experimentation continues to be carried out in academic and research institutes, which continue to occupy a leading position in the market.
The Global Exosome Diagnostics and Therapeutics Market Report is segmented on the basis of the following:
By Type
- Exosome Diagnostics
- Liquid biopsy-based diagnostics
- Biomarker discovery & disease monitoring
- Exosome Therapeutics
- Drug delivery systems
- Regenerative medicine
- Immunotherapy platforms
By Product & Service
- Kits & Reagents
- Isolation kits
- Detection & analysis reagents
- Instruments
- Ultracentrifuges
- Flow cytometry systems
- Nanoparticle tracking analyzers
- Software & Services
- Bioinformatics tools
- Data analysis platforms
By Source
- Blood & Blood Plasma
- Urine
- Stem Cells
- Other Sources
By Application
- Cancer (Oncology)
- Cardiovascular Diseases (CVD)
- Neurological Disorders
- Musculoskeletal Disorders
- Dermatology
- Infectious & Rare Diseases
- Other Application
By End User
- Hospitals & Clinics
- Diagnostic Laboratories / Centers
- Research & Academic Institutes
- Pharmaceutical & Biotechnology Companies
Regional Analysis
Leading Region by Market Share
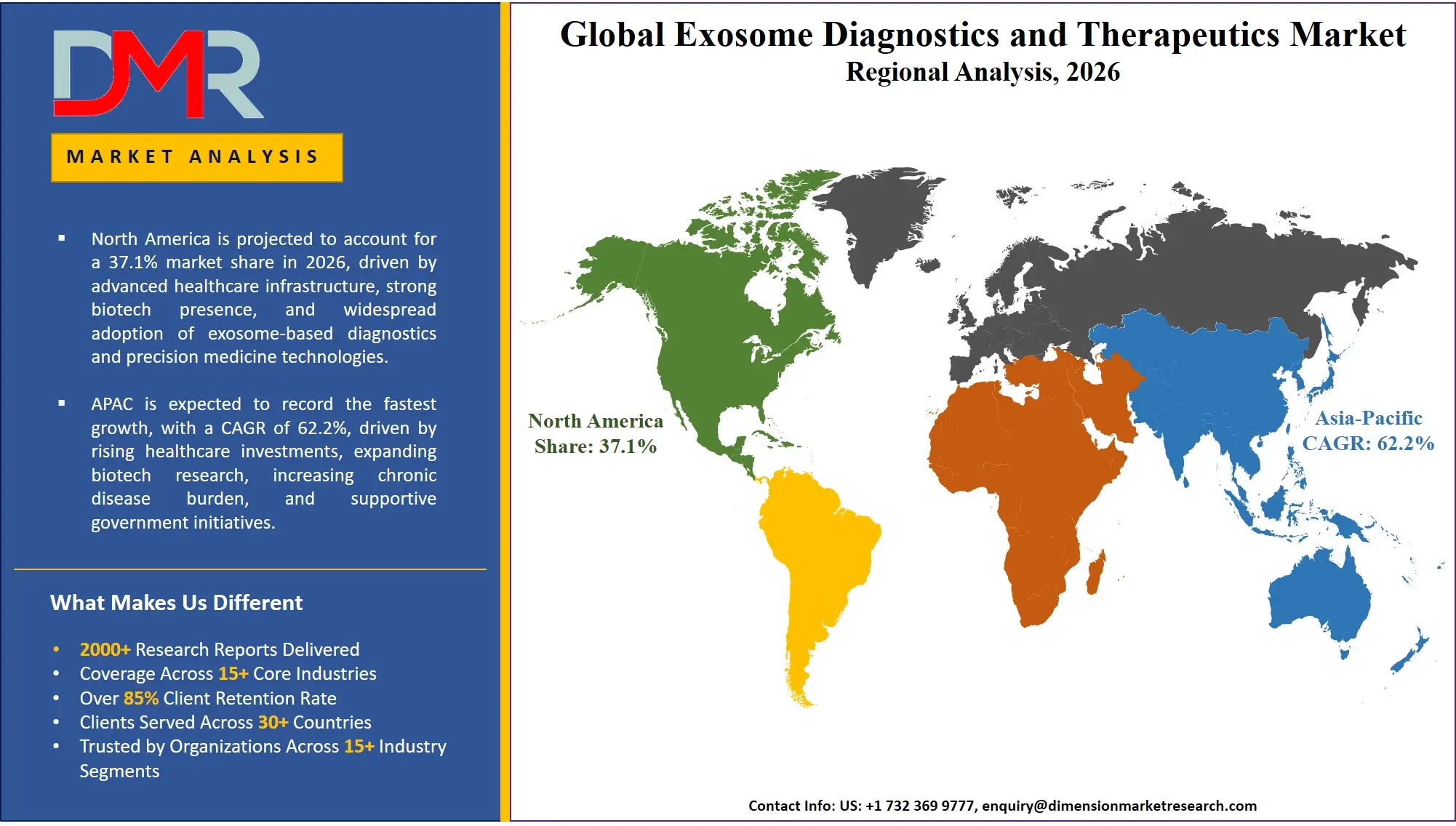
North America is poised to dominate the global exosome diagnostics and therapeutics market as it is projected to hold 37.1% of the market share by the end of 2026. The United States, which dominates North America, has the highest share in the exosome diagnostics and therapeutics market because of the unmatched concentration of leading research universities and the aggressive pipeline development agendas of biotechnology companies. The region boasts a well-developed ecosystem of global pharmaceutical companies, boutique exosome engineering companies, and rich pool of talent in exosome biology and bioinformatics. Cancer (oncology), neurological disorders, and overall retirement of inert nanocarrier systems enterprise investments contribute to the ongoing demand of immuno-oncology platforms development and liquid biopsy-based diagnostics as well as optimization of stem cell source. In addition, a strong climate of venture capital continues to fund future exosome-native enterprises that require expert instruments and software and services in order to translate therapeutic results expeditiously and comply with the FDA.
ℹ
To learn more about this report –
Download Your Free Sample Report Here
Fastest-Growing Regional Market
Asia-Pacific is projected to have the fastest growing exosome diagnostics and therapeutics market, which are being driven by sweeping biotech innovation initiatives in China, Japan, South Korea, and India, led by governments. The rapid economic development pace, the emergence of a significant patient base, and the active growth of the precision medicine economy are forcing existing pharmaceutical companies and research institutions to abandon unproductive old methods of diagnostic study. The discovery of biomarkers and disease monitoring are in high demand to assist these large organizations to move in the right direction towards exosome-native clinical models. An acute shortage of qualified exosome specialists in the region also exists, and it is necessary to outsource to products and services to isolate, characterize, and bioinformatics tools in order to cover the skills gap and allow faster investments in therapeutic and diagnostic projects.
By Region
North America
Europe
- Germany
- The U.K.
- France
- Italy
- Russia
- Spain
- Benelux
- Nordic
- Rest of Europe
Asia-Pacific
- China
- Japan
- South Korea
- India
- ANZ
- ASEAN
- Rest of Asia-Pacific
Latin America
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of Latin America
Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Israel
- Egypt
- Rest of MEA
Competitive Landscape
The competitive landscape of exosome diagnostics and therapeutics has become very dynamic with a heterogeneous array of multinational life science conglomerates, specialized exosome divisions of large diagnostics companies, and niche exosome-native biotech startups. The deep strategic partnerships with cancer (oncology) centers, neurological disorder research consortia, or pharmaceutical companies because they will provide access to the needed clinical co-development opportunities and access to samples of mature cells derived by the patient. The trend of market consolidation is rapidly gaining momentum with the conventional life science tool companies buying their isolation kit and engineering IP-specialized boutiques to survive. Proprietary intellectual property, such as patented exosome loading chemistries and disease-specific bioinformatic panels, is becoming an even more important source of competitive differentiation than generic ultracentrifuge sales, or basic development of assays.
Some of the prominent players in the Global Exosome Diagnostics and Therapeutics Market are:
- Thermo Fisher Scientific
- Danaher Corporation
- QIAGEN
- Bio-Techne Corporation
- Lonza Group
- Fujifilm Holdings Corporation
- Hologic Inc.
- Miltenyi Biotec
- Abcam plc
- RoosterBio Inc.
- System Biosciences LLC
- Aethlon Medical Inc.
- Capricor Therapeutics
- Evox Therapeutics
- ExoCoBio Inc.
- NanoSomix Inc.
- Malvern Panalytical
- JSR Corporation
- Aegle Therapeutics
- Aruna Bio
- Other Key Players
Recent Developments
- January 2026: Thermo Fisher Scientific announced a significant growth of their exosome services project to assist pharmaceutical and biotechnology customers in their oncology and neurological disease treatment. The company also seeks to establish proprietary drug delivery systems using a highly advanced engineering platform that is backed by skills in bioinformatics tools and data analytics.
- November 2025: QIAGEN N.V. enhanced its cooperation with Exosome Diagnostics and offered specific solutions in the field of biomarker discovery, disease monitoring, and software services. These services have been tailored to assist diagnostic laboratories change exosome-based panels into clinical practice and at the same time, comply with international IVDR regulations.
- October 2025: Bio-Techne Corporation purchased Exosome Diagnostics GmbH to develop its liquid biopsy-based diagnostics and to expand its kits and reagents portfolio to be used in blood and plasma-based applications. This action contributes to research and academic centers in order to overcome the difficulties associated with the reproducibility of biomarkers.
Report Details
| Report Characteristics |
| Market Size (2026) |
USD 96.6 Mn |
| Forecast Value (2035) |
USD 6,229.9 Mn |
| CAGR (2026–2035) |
58.9% |
| The US Market Size (2026) |
USD 30.1 Mn |
| Historical Data |
2021 – 2025 |
| Forecast Data |
2027 – 2035 |
| Base Year |
2025 |
| Estimate Year |
2026 |
| Segments Covered |
By Type, By Product & Service, By Source, By Application, and By End User |
| Regional Coverage |
North America – The US and Canada; Europe – Germany, The UK, France, Russia, Spain, Italy, Benelux, Nordic, & Rest of Europe; Asia-Pacific – China, Japan, South Korea, India, ANZ, ASEAN, Rest of APAC; Latin America – Brazil, Mexico, Argentina, Colombia, Rest of Latin America; Middle East & Africa – Saudi Arabia, UAE, South Africa, Turkey, Egypt, Israel, & Rest of MEA |
Frequently Asked Questions
How big is the Global Exosome Diagnostics and Therapeutics Market?
▾ The Global Exosome Diagnostics and Therapeutics market is poised to be valued at USD 96.6 million in 2026 and is projected to reach USD 6,229.9 million by 2035, driven by the universal need for specialized skills in exosome isolation, therapeutic engineering, and biomarker discovery.
What is the CAGR of the Global Exosome Diagnostics and Therapeutics Market from 2026 to 2035?
▾ The market is expected to grow at a CAGR of 58.9% from 2026 to 2035, reflecting the accelerating complexity of exosome-based platforms and the persistent shortage of internal nanovesicle biology talent.
What factors are driving the growth of the Global Exosome Diagnostics and Therapeutics Market?
▾ Key drivers include the global life sciences specialization gap, the imperative to replace invasive biopsies with liquid biopsy-based diagnostics, the engineering complexity of multi-source exosome heterogeneity, and the surge in demand for advanced detection & analysis reagents amid evolving regulatory frameworks.
Which region held the largest share of the Exosome Diagnostics and Therapeutics Market in 2026?
▾ North America is poised to dominate this market with 37.1% of the market share in 2026, driven by a mature biotech ecosystem and aggressive enterprise investment in cancer (oncology) and neurological disorder-focused exosome therapeutics and diagnostics.
Which region is expected to grow the fastest in the Exosome Diagnostics and Therapeutics Market?
▾ The Asia-Pacific region is expected to grow the fastest, fueled by rapid precision medicine adoption in China, Japan, and South Korea, where biomarker discovery & disease monitoring is critical for transitioning large pharmaceutical firms to exosome-native R&D.
What are the major trends in the Global Exosome Diagnostics and Therapeutics Market?
▾ Major trends include the integration of AI into bioinformatics workflows, the rise of GMP-standardized isolation kit platforms, the demand for disease-specific exosome panels, and the focus on reproducible characterization using nanoparticle tracking analyzers within complex multi-omics environments.
Who are the key players in the Global Exosome Diagnostics and Therapeutics Market?
▾ Key players include life science leaders like Thermo Fisher Scientific and QIAGEN, diagnostic innovators, as well as the therapeutic divisions of specialized biotechs like Evox Therapeutics and Codiak BioSciences, alongside exosome-native pure-play engineering firms.
How is the Global Exosome Diagnostics and Therapeutics Market segmented?
▾ The market is segmented by Type, By Product & Service, By Source, By Application, and By End User.